Advanced Light Microscopy magnifies scope of research

By Phillip Sitter | MU Bond Life Sciences Center
Microscopes have come a long way since Anton van Leeuwenhoek first looked at single-cell organisms in the 1600s.
Now, cutting-edge microscopes allow scientists a better look at how cells interact and work.
The results were easy to see Tuesday morning when a new digital light sheet illuminated all the cells in a zebrafish embryo of Anand Chandrasekhar, a Bond Life Sciences Center scientist and professor of biological sciences. The fish, with its two eyes, brain and spinal cord lit up like a green-colored digital ghost floating in invisible black waters of the monitor screen.
This new microscope joins an array in the Advanced Light Microscopy Core, or MCC, located at Bond LSC. The MCC is one of nine core facilities at MU that provide vital services across campus, from DNA sequencing to imaging.
Researchers and staff at the MCC showed on Tuesday how the new capabilities of the technology give them and MU a competitive edge in their research through better visualizations of their experiments.
The digital reconstruction produced by the light sheet combines “a whole bunch of images over an extended period of time,” Chandrasekhar said. Thousands of images of a cell or organism can be generated by the new equipment, at speeds of hundreds of frames per second, creating a picture that can easily take up a terabyte of hard drive space.
At those speeds, Chandrasekhar said he can “literally watch neurons in the brain light up” in real time. He observes how neuron changes occur in the fish’s brain as the animal goes about its different routine behaviors like avoiding possible predators and searching for food. With this capability, researchers like him can study how neuronal networks develop.
The new digital light sheet uses less damaging light than traditional microscopes and allows for clearer pictures, often with a 3-D look at their structure. More advanced imaging equipment also allow for faster, larger volume experiments that are gentler on the biological samples used in them.
Thomas Phillips, MCC Director and professor of biological sciences, explained how his center now can better serve scientists across campus.
In addition to the digital light sheet imaging system, the MCC also has two new super-resolution microscopy systems, what Phillips said researchers across campus expressed they needed most. The presence of these super-resolution systems in particular puts MU at the forefront of microscopic research.
“Whereas every school has a confocal, less than 10 percent have super-resolution capabilities,” Phillips said, and now MU has two super-resolution systems. “The new equipment adds totally new capabilities without interfering with the traditional confocal activities.”
Traditional confocal microscopy systems, while still useful, have limitations in their resolution which super-resolution systems overcome by how the specimens are illuminated.
In addition to having technology other institutions do not, MU has a higher quality version of super-resolution. Phillips explained that while systems at other institutions use two different lasers in the internal mechanisms of their imaging equipment, MU is one of less than 10 or 12 schools that has access to a super-resolution system that has a unique three laser combination that increases the resolution of the system.
“Super-resolution microscopy allows us to see how individual proteins are interacting inside cells in ways we haven’t been able to before,” he said.
Sergiy Sukhanov uses one of the super-resolution microscopes in cardiology research that looks at failure of organ systems. Sukhanov studies heart failure and atherosclerosis — the chronic, dangerous build-up of plaque in arteries that can create blockages that lead to heart attacks, strokes and death.
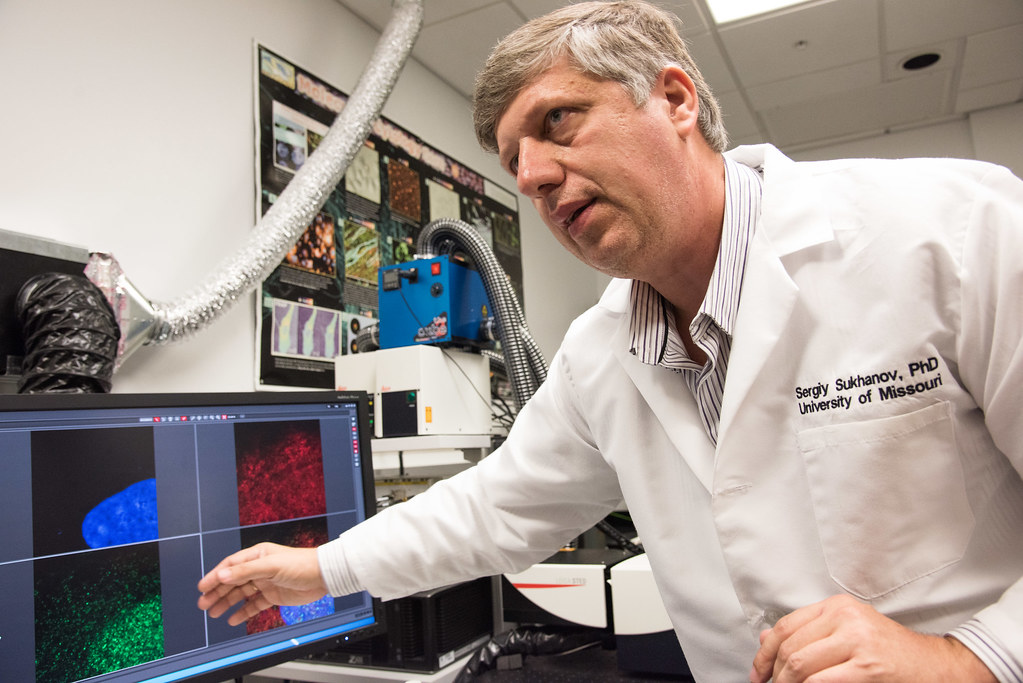
The associate research professor at MU’s School of Medicine showed on Tuesday images of protein interactions in the smooth muscle cells that line arteries. The goal of understanding these interactions is to help keep these smooth muscles in the healthy condition they need to be in to prevent catastrophic blockages.
With its capability of a resolution of up to 30 nanometers, Sukhanov said that the new equipment’s advantage for him is that he can actually see the cell he’s working on. Soon, he hopes to be able to work with live cells so he can observe changes in their protein structures in response to changes in their environment in real time.
Availability of the new equipment lured Sukhanov and his research team to MU over other institutions in 2014. He explained that he struggled to find the equipment he needed for his experiments at Tulane University in New Orleans or elsewhere in Louisiana. Other systems at other institutions usually only have a resolution of up to 50 nanometers.
Sukhanov’s decision is an example of the growth that the new imaging capabilities at the MCC can promote. As director Phillips explained, “you don’t plan experiments ahead of time if you don’t have the apparatus for it.”
MCC not only provides services across campus, but can give scientists insight into how to better look at their specimens.
First-year graduate student Jennifer Wolf displayed a super-resolution image of cancerous liver cells infected with Hepatitis C that been treated with a drug thought to prevent the virus from spreading. The drug aggregates viral capsid molecules – the outer part of a virus – within the infected cells to effectively contain them.
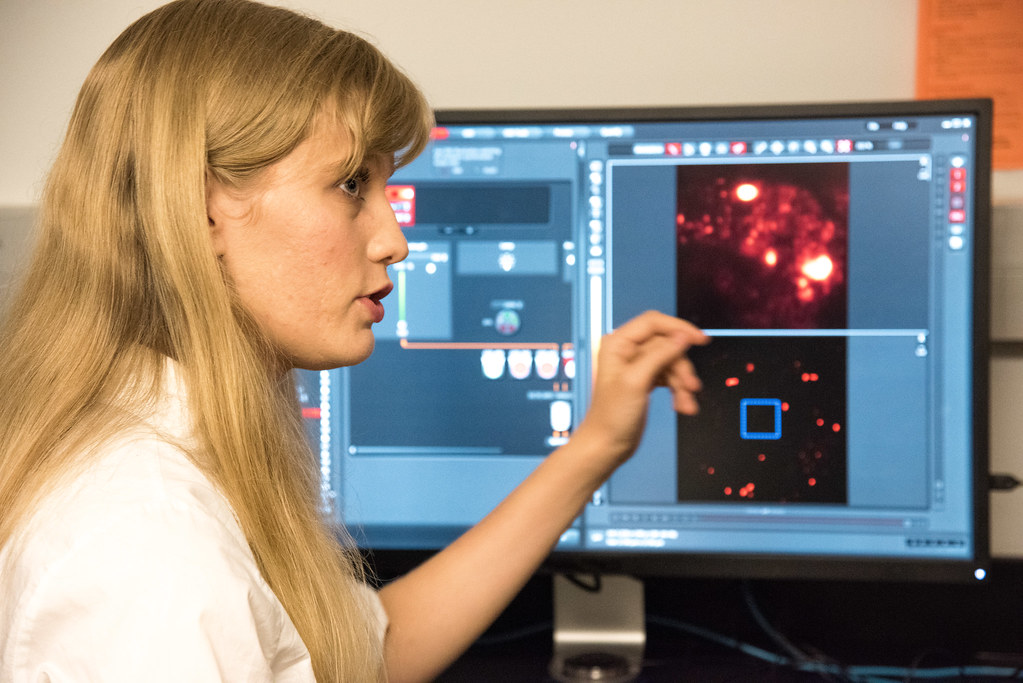
Wolf said with the new equipment she is now able to see an individual molecule, and using that see the overlap of proteins, RNA and DNA fragments, which can help determine the effectiveness of drugs in treatment.
Associate director of the MCC Alexander Jurkevich explained that the super-resolution equipment also allows for the compilation of separate images into an even more detailed 3-D projection.
Wolf pulled up an image of green-colored mitochondria surrounded by red micro-tubules, green hubs of cellular activity connected by red highways that looked almost like a city from space at night. Wolf used the 3-D capability and rotated the image. She turned the biological intricacy on its side until it looked something like a galaxy on a cosmic horizon, only this view that maybe even fewer people have witnessed is microscopic.
“It’s important to use new technology like this to help the University of Missouri to stay on top,” Wolf said.


