By Cara Penquite | Bond LSC
Gene-editing is the pinnacle of a biologist’s toolbox, but often left unexplained it seems more magic than science.
Growing rice from a small cluster of cells to 4-foot stalks can take six-months or more of planning and careful nurture. But how do scientists change the intricate genetic material in each cell of the plants?
The CRISPR-Cas9 gene editing tool changes a plant’s DNA. As Ph.D. student Ajay Gupta knows firsthand through work altering plants for the Bing Yang lab.
“CRISPR is relatively new. It’s like 10 years old only and still we are working to modify it and improve it in plants,” Gupta said.
The process starts with a few cells from seeds capable of maturing into any type of cell to form a cell cluster, known as a callus.
But how do scientists insert instructions into a cell?
In the Yang lab, they use particle bombardment to insert DNA, fundamental instructions used to build everything within a cell. These inserted DNA strands tell the cells how to build the CRISPR tools to change their own DNA.
To shoot the DNA molecules into the cells, researchers use high pressure helium gas operated gene gun to bombard the cells with DNA coated gold particles.
Once the DNA instructions are in the cell, they build the CRISPR-Cas9 tool using the plant’s genetic machinery.
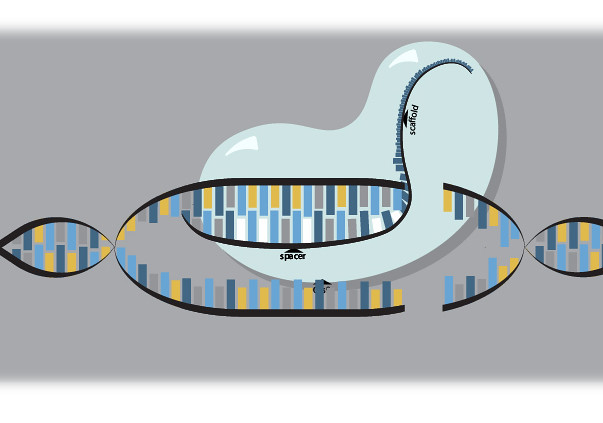
CRISPR-Cas9 has two parts: the Cas9 protein and an RNA molecule that only binds to the DNA section scientists want to change. This RNA strand guides the Cas9 to the correct location.
But the parts of the guide RNA do two things. First, the spacer part — usually about 20 base pairs only binds to the section of DNA researchers want it to. Second, a part called the scaffold — nearly four times the size of the spacer is required for attaching to the Cas9.

When the spacer of guide RNA binds to the DNA, the Cas9 enzyme acts like a pair of scissors, cutting that section of DNA.


Now we have DNA broken in two, and the plant’s built-in repair system starts its job. However, this repair system is flawed, and it usually messes up while rebuilding the DNA’s bases, or building blocks. Its errors vary every time, so many different mutations are possible.
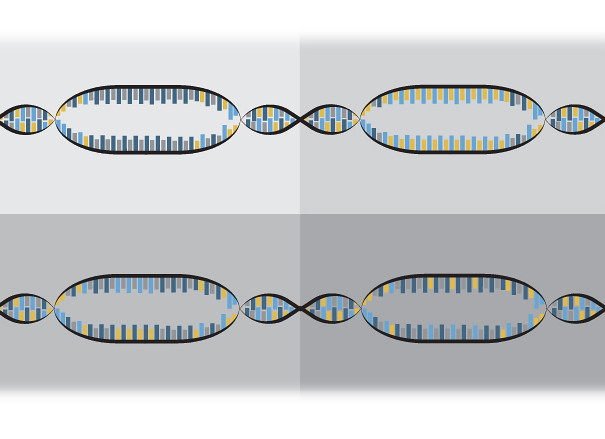
“The repair machinery which is employed is not perfect,” Gupta said. “When it repairs those double-strand breaks, it causes some errors. The error could be a single base, and it could be 10 bases or it could be a thousand bases. So that is unpredictable.”
Think of it this way: imagine you have a ten-page instruction manual, and you rip out pages two and three. You could replace them with blank, yellow or red pieces of paper and the book would have ten pages again, but the instruction manual can’t be used again. Just like that, it doesn’t matter which error happens, because any mutated change in the gene results in it not working.
“This is how the normal CRISPR-Cas9 works, when you just want to shut that gene down,” Gupta said.
This puts the gene’s instructions out of commission and is commonly called a knockout gene. That helps scientists determine what the particular gene does. They then compare “normal” plants to plants with the knocked out gene to determine what happens differently.
Now the waiting happens. Researchers grow the plants using different hormones to mature the callus cells into roots and stems.


“Once they have shoots and roots then you transfer them to soil and grow the complete plants out of that,” Gupta said.
When the plants become adults, the researchers check the knocked out changes by extracting its DNA in a process known as genotyping.
While traditional CRISPR-Cas9 is used by researchers to learn more about plant genomes, Gupta and others in the Yang lab also work to edit parts of DNA and replace it with specific mutations. This process is known as prime editing.
“The exciting part about this [prime editing] is that theoretically it can do anything,” Gupta said.
Prime editing takes traditional CRISPR-Cas9 editing a step further, and the Yang lab’s research into it opens the door for future collaborations with labs around the world in applied and basic sciences.



