Scientists find how nematodes use key hormones to take over root cells
Roger Meissen | Bond Life Sciences Center
This is a story about spit.
Not just any spit, but the saliva of cyst nematodes, a parasite that literally sucks away billions in profits from soybean and other crops every year.
Researchers are working to uncover exactly how these tiny worms trick plant root cells into feeding them for life.
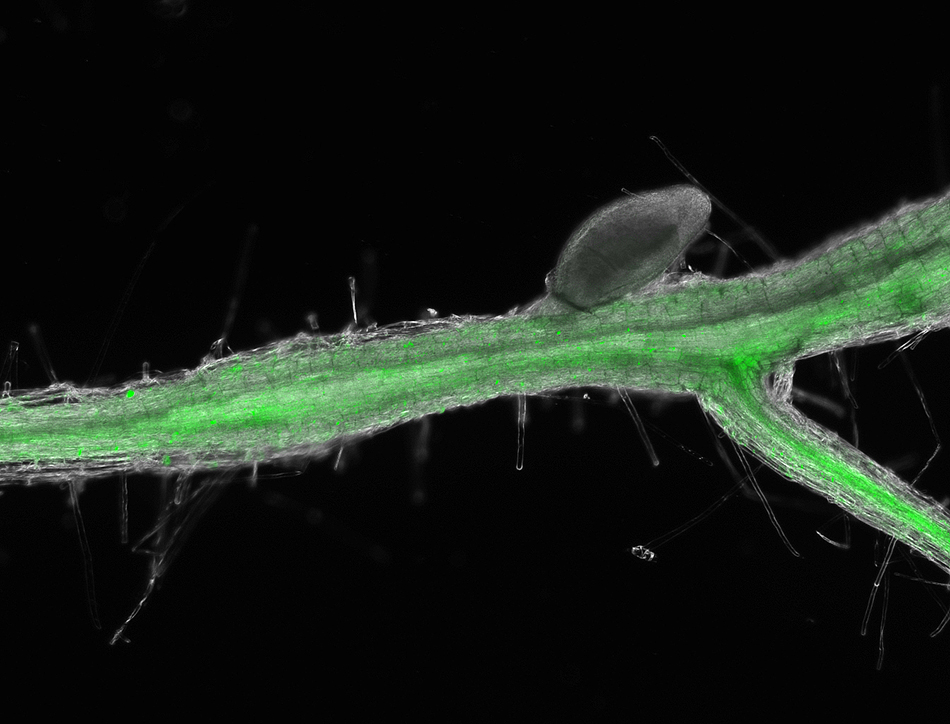
A team at the University of Missouri Bond Life Sciences Center collaborated with scientists at the University of Bonn in Germany to discover genetic evidence that the parasite uses its own version of a key plant hormone and that of the plants to make root cells vulnerable to feeding. Their research recently appeared in Proceedings of the National Academy of Sciences.

Cytokinin is normally produced in plants, but these researchers determined that this growth hormone is also produced by nematode parasites that use it to take over plant root cells.
“While it’s well-known that certain bacteria and some fungi can produce and secrete cytokinin to cause disease, it’s not normal for an animal to do this,” said Melissa Mitchum, an MU plant scientist and co-author on the study. “This is the first study to demonstrate the ability of an animal to synthesize and secrete cytokinin for parasitism.”
Not Science Fiction
Reprogramming another organism might sound like a far out concept, but it’s a reality for plants susceptible to nematodes.
Cyst nematodes hatch from eggs laid in fields and quickly migrate to the roots of nearby plants. They inject nematode spit into a single host cell of soybean, beet and other crop roots.

“Imagine a hollow needle at the head of the nematode that the parasite uses to penetrate into the plant cell wall and secrete pathogenic proteins and hormone mimics,” said Carola De La Torre, a co-author of the study and plant sciences PhD student with Mitchum’s lab. “Nematodes use the spit to transform the host cell into a nutrient sink from which they feed on during their entire life cycle. This de novo differentiation process greatly depends on nematode–derived plant hormone mimics or manipulation of plant hormonal pathways caused by effector proteins present in the nematode spit.”
These effector proteins and other small molecules in their spit cause the root cell to forego normal processes and create a huge feeding site called a syncytium. In a short period of time, this causes hundreds of root cells to combine into a large nutrient storage unit that the nematode feeds from for its entire life.
Being able to convince a root cell to do the nematode’s bidding starts with a takeover of the plant host cell cycle — which regulates DNA replication and division. This implies that a plant hormone like cytokinin is involved, says Mitchum. Cytokinin normally regulates a plant’s shoot growth, leaf aging, and other cell processes.
Proving the relationship
While Mitchum’s lab had a hunch that cytokinin was key to this takeover, proving it took some creative science.
De La Torre and Demosthenis Chronis, a postdoctoral fellow MU at the Bond LSC depended on mutant Arabidopsis plants to explore the relationship. “One of the great things about using Arabidopsis as our host plant is the vast genetic resources of cytokinin and hormone mutants that are available through the scientific community,” De La Torre said.
She infected Arabidopsis that contained a reporter gene called TCSn/GFP with nematodes. This gene is associated with cytokinin responses within the plant cells and is fused with a jellyfish protein that glows green when turned on. So, De La Torre saw nematodes activated cytokinin responses in the plant early after infection when her plants emitted a green fluorescent glow under the microscope.
Next, she infected plants missing the majority of their cytokinin receptors with nematodes. Then she started counting nematodes present.
“After a careful evaluation of nematode infection, we observed less female nematodes developing in the receptor mutants compared to the wild type” De La Torre said. “The nematodes could not infect well, and that was a clear piece of evidence suggesting that cytokinin plays a main role in plant–nematode interactions.”
Another experiment looked at Arabidopsis containing a reporter gene called GUS that was fused to the regulatory sequences of the cytokinin receptor genes. All three cytokinin receptor genes were activated where the nematode was feeding.
A final experiment used a mutant that created an excess of an enzyme that degrades cytokinin, finding that a base level of plant cytokinin was also necessary for nematode growth.
“The simple statement is that the cytokinin receptors were activated in response to nematode infection and the mutants did not support growth and development of the nematodes,” Mitchum said. “This shows that if you take away the ability of the plant to recognize cytokinin the worms are unable to fully develop.”
An international collaboration
Mitchum’s team did not work alone.
The lab of Florian Grundler at Rheinische Friedrich-Wilhelms-University of Bonn, Germany, was also on a mission to uncover if genes in the nematode controlled cytokinin activation. They had identified a key gene in the beet cyst nematode that makes the cytokinin hormone. When they took away the ability of the nematode to secrete cytokinin certain cell cycle genes were not activated at the feeding site and the nematodes did not develop. Now we know that the nematode is also secreting cytokinin to modulate the pathways.
De La Torre took that information and found the same gene in the soybean cyst nematode.
Now, Mitchum’s team is trying to find how this key gene might work differently in other nematode types, like root-knot nematode as part of a new National Science Foundation grant. They hope this will help lead to better resistance in future crops.
“Understanding how the nematode modulates its host is going to help us exploit new technologies to engineer plants with enhanced resistance to this terribly devastating pathogen,” Mitchum said. “Technology is changing all the time, we’re gaining new tools constantly, so you never know when something new is going to allow us to do something specific at the site of nematode feeding that will lead to a breakthrough.”
Mitchum is a Bond LSC investigator and an associate professor of Plant Sciences in the College of Agriculture, Food and Natural Resources. The study “A Plant Parasitic Nematode Releases Cytokinin that Control Cell Division and Orchestrate Feeding-Site Formation in Host Plants” recently was published by the Proceedings of the National Academy of Sciences and was supported by the National Science Foundation (Grant #IOS-1456047 to Mitchum). The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding agencies.


binance bonus za registráciu
February 16, 2024 @ 3:55 pm
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me? https://www.binance.com/sk/register?ref=PORL8W0Z
binance
February 18, 2024 @ 4:14 am
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me. https://www.binance.com/ka-GE/register?ref=W0BCQMF1
anm"ala dig till binance
February 19, 2024 @ 6:04 pm
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me. https://www.binance.com/sv/register?ref=GJY4VW8W
Register
February 20, 2024 @ 3:30 am
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me. https://www.binance.com/en/join?ref=FIHEGIZ8
Μπνου εγγραφ Binance
March 2, 2024 @ 8:09 am
Your point of view caught my eye and was very interesting. Thanks. I have a question for you. https://accounts.binance.com/el/register?ref=B4EPR6J0
crie uma conta na binance
March 29, 2024 @ 11:13 am
Thanks for sharing. I read many of your blog posts, cool, your blog is very good.
高級 リアル ドール
May 4, 2024 @ 5:41 pm
ラブドール販売 憧れの人形に深い友情を感じても大丈夫ですか?
Bonus Pendaftaran di Binance
May 8, 2024 @ 7:40 pm
Your article helped me a lot, is there any more related content? Thanks!
binance registracija
May 11, 2024 @ 5:12 am
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
Friday Night Funkin
June 21, 2024 @ 1:45 am
Thanks to this Friday Night Funkin by ninjamuffin99. I just want to say that!
Binance创建账户
June 21, 2024 @ 11:40 am
Your article helped me a lot, is there any more related content? Thanks!
najlepsze escape roomy
June 28, 2024 @ 1:19 pm
Good day! Do you know if they make any plugins to help with Search Engine Optimization?
I’m trying to get my site to rank for some targeted keywords but I’m not seeing very good results.
If you know of any please share. Appreciate it! I
saw similar article here: Escape rooms hub
Stephenced
June 29, 2024 @ 3:59 am
mexico drug stores pharmacies: mexican pharmacy – best online pharmacies in mexico
Stephenced
June 29, 2024 @ 6:09 am
mexican online pharmacies prescription drugs: cmq pharma mexican pharmacy – mexican online pharmacies prescription drugs
Stephenced
June 29, 2024 @ 9:49 am
mexican online pharmacies prescription drugs: mexican pharmacy online – pharmacies in mexico that ship to usa
DonaldPoild
June 29, 2024 @ 9:52 am
medicine in mexico pharmacies
http://cmqpharma.com/# medication from mexico pharmacy
mexico drug stores pharmacies
DonaldPoild
June 29, 2024 @ 12:27 pm
buying prescription drugs in mexico online
https://cmqpharma.online/# medicine in mexico pharmacies
mexico pharmacies prescription drugs
DonaldPoild
June 29, 2024 @ 1:50 pm
reputable mexican pharmacies online
https://cmqpharma.online/# mexican mail order pharmacies
mexican online pharmacies prescription drugs
DonaldPoild
June 29, 2024 @ 10:07 pm
mexican border pharmacies shipping to usa
http://cmqpharma.com/# reputable mexican pharmacies online
best online pharmacies in mexico
ร้านปะยางรถยนต์ใกล้ฉันในสงขลา
July 6, 2024 @ 2:30 am
I know this if off topic but I’m looking into starting
my own blog and was curious what all is required to get set
up? I’m assuming having a blog like yours would cost a pretty penny?
I’m not very internet smart so I’m not 100% sure.
Any suggestions or advice would be greatly appreciated. Kudos
Margareta N
July 6, 2024 @ 5:05 pm
I like this site very much, Its a real nice office to read and
find information.?
Rodolfoexete
July 9, 2024 @ 12:18 am
mexico drug stores pharmacies
https://cmqpharma.com/# mexico pharmacies prescription drugs
mexico drug stores pharmacies
IrvinDag
July 9, 2024 @ 12:43 am
mexican pharmacy: cmqpharma.com – medication from mexico pharmacy
Michaelnix
July 10, 2024 @ 9:27 pm
mexico drug stores pharmacies
http://cmqpharma.com/# best online pharmacies in mexico
medication from mexico pharmacy
HenryElory
July 11, 2024 @ 1:09 am
mexican mail order pharmacies: cmqpharma.com – purple pharmacy mexico price list
russkoeporno365.pro
July 14, 2024 @ 1:00 am
I reckon something truly special in this website.
My website: chastnoeporno
Charlesskimi
July 19, 2024 @ 8:51 pm
top 10 online pharmacy in india buy medicines online in india indian pharmacy paypal
Edwardbus
July 19, 2024 @ 9:21 pm
http://canadapharmast.com/# canadian pharmacy no scripts
Davidgrigh
July 19, 2024 @ 10:29 pm
canadian medications: canadianpharmacyworld com – canadian medications
Charlesskimi
July 20, 2024 @ 1:23 am
india pharmacy mail order online pharmacy india indian pharmacy
Michaeleduch
July 20, 2024 @ 2:28 am
online canadian pharmacy: canadianpharmacyworld com – canada drug pharmacy
Michaeleduch
July 20, 2024 @ 3:56 am
reputable mexican pharmacies online: mexico drug stores pharmacies – mexico drug stores pharmacies
Edwardbus
July 20, 2024 @ 4:16 am
http://indiapharmast.com/# top 10 online pharmacy in india
Edwardbus
July 20, 2024 @ 7:51 am
https://foruspharma.com/# mexican border pharmacies shipping to usa
Charlesskimi
July 20, 2024 @ 10:13 am
canadian pharmacy cheap best canadian pharmacy canadian pharmacy price checker
Davidgrigh
July 20, 2024 @ 10:52 am
best canadian pharmacy online: legitimate canadian pharmacies – canadian pharmacy reviews
Michaeleduch
July 20, 2024 @ 11:48 am
medicine in mexico pharmacies: buying prescription drugs in mexico – mexican online pharmacies prescription drugs
Michaeleduch
July 20, 2024 @ 1:30 pm
Online medicine home delivery: indian pharmacies safe – indian pharmacies safe
Charlesskimi
July 20, 2024 @ 1:30 pm
п»їlegitimate online pharmacies india Online medicine home delivery top 10 pharmacies in india
Davidgrigh
July 20, 2024 @ 4:36 pm
online shopping pharmacy india: india online pharmacy – indian pharmacy paypal
Edwardbus
July 20, 2024 @ 5:10 pm
http://canadapharmast.com/# pharmacy canadian
Edwardbus
July 20, 2024 @ 7:01 pm
http://foruspharma.com/# mexican rx online
Michaeleduch
July 20, 2024 @ 8:47 pm
canada pharmacy world: safe reliable canadian pharmacy – canadian pharmacy meds
Davidgrigh
July 20, 2024 @ 9:37 pm
pharmacy website india: online shopping pharmacy india – india pharmacy
Michaeleduch
July 20, 2024 @ 10:23 pm
mexico drug stores pharmacies: mexican mail order pharmacies – purple pharmacy mexico price list
house not selling
July 21, 2024 @ 4:52 am
Good article! We are linking to this great content on our website. Keep up the great writing.
Michaeleduch
July 21, 2024 @ 6:05 am
mail order pharmacy india: best india pharmacy – buy medicines online in india
Edwardbus
July 21, 2024 @ 6:53 am
http://foruspharma.com/# purple pharmacy mexico price list
Michaeleduch
July 21, 2024 @ 7:51 am
reputable indian online pharmacy: pharmacy website india – pharmacy website india
Jamesbug
July 21, 2024 @ 10:50 am
doxycycline tablets in india: doxycycline iv – antibiotic doxycycline
ThomasVer
July 21, 2024 @ 1:03 pm
http://doxycyclinedelivery.pro/# doxycycline 40 mg india
ThomasVer
July 21, 2024 @ 4:45 pm
http://clomiddelivery.pro/# can i order generic clomid without insurance
Jamesbug
July 21, 2024 @ 8:52 pm
amoxicillin 250 mg price in india: amoxicillin 500 mg – where can i get amoxicillin
ThomasVer
July 21, 2024 @ 9:10 pm
https://amoxildelivery.pro/# where to buy amoxicillin 500mg without prescription
Jamesbug
July 21, 2024 @ 11:42 pm
vibramycin 100 mg: doxycycline coupon – doxycycline capsules
ThomasVer
July 22, 2024 @ 12:55 am
http://doxycyclinedelivery.pro/# cost of doxycycline
ThomasVer
July 22, 2024 @ 5:20 am
https://paxloviddelivery.pro/# paxlovid india
ThomasVer
July 22, 2024 @ 8:46 am
https://clomiddelivery.pro/# can you buy cheap clomid now
Jamesbug
July 22, 2024 @ 9:28 am
can you buy cheap clomid without insurance: how to buy clomid without a prescription – can you buy cheap clomid without insurance
Jamesbug
July 22, 2024 @ 12:17 pm
how to buy generic clomid: can i order cheap clomid tablets – can i purchase cheap clomid
ThomasVer
July 22, 2024 @ 1:07 pm
http://paxloviddelivery.pro/# paxlovid pharmacy
ThomasVer
July 22, 2024 @ 4:42 pm
https://doxycyclinedelivery.pro/# doxycycline for sale
ThomasVer
July 22, 2024 @ 9:06 pm
http://doxycyclinedelivery.pro/# where can i get doxycycline
Jamesbug
July 22, 2024 @ 10:07 pm
purchase cipro: buy cipro without rx – ciprofloxacin 500mg buy online
ThomasVer
July 23, 2024 @ 12:36 am
http://clomiddelivery.pro/# can you get generic clomid for sale
ThomasVer
July 23, 2024 @ 4:43 am
http://amoxildelivery.pro/# amoxicillin 500 mg without a prescription
ThomasVer
July 23, 2024 @ 7:55 am
https://ciprodelivery.pro/# cipro ciprofloxacin
Jamesbug
July 23, 2024 @ 10:47 am
doxycycline uk cost: doxycycline 100mg tablet price – doxycycline for dogs
Jamesbug
July 23, 2024 @ 1:37 pm
clomid without insurance: can i purchase generic clomid tablets – can you get clomid now
ThomasVer
July 23, 2024 @ 3:09 pm
https://clomiddelivery.pro/# can i buy cheap clomid without prescription
ThomasVer
July 23, 2024 @ 7:48 pm
http://amoxildelivery.pro/# buy amoxicillin 500mg canada
Jamesbug
July 23, 2024 @ 11:30 pm
amoxicillin 500mg over the counter: where to buy amoxicillin pharmacy – price of amoxicillin without insurance
ThomasVer
July 23, 2024 @ 11:31 pm
https://doxycyclinedelivery.pro/# doxycycline 100mg price south africa
Jamesbug
July 24, 2024 @ 2:27 am
paxlovid cost without insurance: paxlovid cost without insurance – paxlovid generic
Jamesbug
July 24, 2024 @ 12:12 pm
paxlovid buy: paxlovid covid – Paxlovid over the counter
Jamesbug
July 24, 2024 @ 3:07 pm
antibiotics cipro: where to buy cipro online – purchase cipro
Jamesbug
July 25, 2024 @ 12:45 am
buy paxlovid online: Paxlovid over the counter – buy paxlovid online
Jamesbug
July 25, 2024 @ 3:36 am
can you buy doxycycline over the counter uk: doxycycline 150 mg price – doxycycline tablets online
Русское Порно 365
July 26, 2024 @ 5:53 am
My website: частное порно
Robertlense
July 27, 2024 @ 2:01 am
ciprofloxacin 500 mg tablet price: buy cipro online canada – cipro
RichardBup
July 29, 2024 @ 7:48 am
https://mexicandeliverypharma.com/# п»їbest mexican online pharmacies
Dominictix
July 29, 2024 @ 9:23 am
best online pharmacies in mexico: mexican drugstore online – buying from online mexican pharmacy
Wayneren
July 29, 2024 @ 9:23 am
mexican rx online: mexican border pharmacies shipping to usa – purple pharmacy mexico price list
ร้านปะยางรถสิบล้อ
July 29, 2024 @ 9:41 am
I’ve learn a few excellent stuff here. Certainly
value bookmarking for revisiting. I surprise how much effort you set to create any such great informative website.
Wayneren
July 29, 2024 @ 2:43 pm
buying prescription drugs in mexico online: mexican border pharmacies shipping to usa – buying prescription drugs in mexico online
Arnoldstota
July 29, 2024 @ 8:45 pm
purple pharmacy mexico price list buying prescription drugs in mexico online mexican pharmacy
ร้านปะยางรถบรรทุก24ชมใกล้ฉัน
July 30, 2024 @ 2:54 am
Hey very interesting blog!
boy789
July 30, 2024 @ 3:19 am
These are actually wonderful ideas in concerning blogging.
You have touched some good factors here. Any way keep up
wrinting.
Dominictix
July 31, 2024 @ 6:02 am
mexican border pharmacies shipping to usa: purple pharmacy mexico price list – buying from online mexican pharmacy
Dominictix
July 31, 2024 @ 3:53 pm
medicine in mexico pharmacies: mexican online pharmacies prescription drugs – medicine in mexico pharmacies
Arnoldstota
July 31, 2024 @ 4:03 pm
mexican online pharmacies prescription drugs purple pharmacy mexico price list mexican pharmacy
NelsonbiC
August 1, 2024 @ 3:55 am
purple pharmacy mexico price list: mexico drug stores pharmacies – buying prescription drugs in mexico online
Wayneren
August 1, 2024 @ 11:11 pm
п»їbest mexican online pharmacies: reputable mexican pharmacies online – buying prescription drugs in mexico
Wayneren
August 2, 2024 @ 9:10 pm
buying prescription drugs in mexico: reputable mexican pharmacies online – mexican pharmaceuticals online
Dominictix
August 3, 2024 @ 3:05 am
mexican pharmaceuticals online: purple pharmacy mexico price list – mexican rx online
Wayneren
August 3, 2024 @ 6:29 am
best online pharmacies in mexico: mexican pharmaceuticals online – pharmacies in mexico that ship to usa
NelsonbiC
August 3, 2024 @ 1:22 pm
mexico drug stores pharmacies: purple pharmacy mexico price list – best online pharmacies in mexico
ร้านปะยางที่ใกล้ฉัน
August 3, 2024 @ 1:49 pm
There is definately a great deal to learn about this subject.
I like all the points you have made.
ร้านปะยางมอเตอร์ไซค์24ชมใกล้ฉัน
August 3, 2024 @ 2:02 pm
Howdy! Do you use Twitter? I’d like to follow
you if that would be ok. I’m definitely enjoying your blog and look forward to new
updates.
RobertKeync
August 4, 2024 @ 12:37 am
tamoxifen pill cost of tamoxifen tamoxifen joint pain
Matthewadact
August 4, 2024 @ 12:37 am
get generic propecia: get generic propecia tablets – buy propecia
Patrickirowl
August 4, 2024 @ 12:39 am
https://nolvadexbestprice.pro/# tamoxifen and depression
Lewiscoata
August 4, 2024 @ 12:49 am
http://prednisonebestprice.pro/# prednisone pills cost
RobertKeync
August 4, 2024 @ 5:43 am
buy cytotec online buy misoprostol over the counter purchase cytotec
Matthewadact
August 4, 2024 @ 6:03 am
zithromax 500 without prescription: zithromax over the counter – zithromax cost canada
Patrickirowl
August 4, 2024 @ 6:12 am
http://propeciabestprice.pro/# cost of cheap propecia price
Matthewadact
August 4, 2024 @ 7:35 am
cytotec buy online usa: Abortion pills online – buy cytotec over the counter
Patrickirowl
August 4, 2024 @ 7:48 am
https://zithromaxbestprice.pro/# zithromax 1000 mg online
RobertKeync
August 4, 2024 @ 12:22 pm
prednisone 2 mg buy prednisone from india buy prednisone online canada
Matthewadact
August 4, 2024 @ 1:08 pm
buy cytotec online fast delivery: order cytotec online – cytotec online
Patrickirowl
August 4, 2024 @ 1:25 pm
https://prednisonebestprice.pro/# prednisone 2 mg
RobertKeync
August 4, 2024 @ 1:51 pm
zithromax for sale 500 mg zithromax price canada zithromax for sale 500 mg
Matthewadact
August 4, 2024 @ 2:42 pm
order propecia pills: propecia cheap – buying generic propecia without insurance
Patrickirowl
August 4, 2024 @ 3:00 pm
http://nolvadexbestprice.pro/# tamoxifen hair loss
ปะยางมอเตอร์ไซค์นอกสถานที่
August 4, 2024 @ 3:42 pm
Everything is very open with a precise explanation of the issues.
It was really informative. Your website is very useful.
Thank you for sharing!
Lewiscoata
August 4, 2024 @ 3:46 pm
http://prednisonebestprice.pro/# 5mg prednisone
ร้านประยาง
August 4, 2024 @ 6:08 pm
payang-pp.com ให้บริการปะยางนอกสถานที่ใกล้ฉันครอบคลุมทั่วกรุงเทพและปริมณฑล ไม่ว่าคุณจะเจอปัญหายางแตกหรือรั่วที่ไหน เพียงโทรเรียกใช้บริการ ทีมช่างของเราพร้อมเดินทางไปเปลี่ยนยางนอกสถานที่ให้คุณอย่างรวดเร็ว ด้วยอุปกรณ์และเครื่องมือที่ทันสมัย มาตรฐาน เรามั่นใจว่าจะสามารถปะยางหรือเปลี่ยนยางให้คุณได้อย่างมีประสิทธิภาพ
โดยไม่ต้องเสียเวลานำรถเข้าอู่ให้ยุ่งยาก
RobertKeync
August 4, 2024 @ 6:55 pm
where to buy zithromax in canada zithromax capsules 250mg zithromax cost uk
Matthewadact
August 4, 2024 @ 8:09 pm
zithromax 250 mg: where to buy zithromax in canada – zithromax 250 mg australia
Matthewadact
August 4, 2024 @ 9:48 pm
nolvadex side effects: how to get nolvadex – should i take tamoxifen
Kevinraf
August 4, 2024 @ 11:43 pm
cost of generic zithromax: buy zithromax 1000 mg online – zithromax price canada
Matthewadact
August 5, 2024 @ 4:14 am
generic propecia without a prescription: propecia online – cost propecia no prescription
Matthewadact
August 5, 2024 @ 5:50 am
prednisone 50 mg price: where can i buy prednisone without a prescription – how much is prednisone 10mg
NathanGriva
August 5, 2024 @ 7:04 am
Link pyramid, tier 1, tier 2, tier 3
Primary – 500 references with integration contained in pieces on writing portals
Secondary – 3000 web address Forwarded links
Lower – 20000 hyperlinks assortment, remarks, writings
Implementing a link network is useful for web crawlers.
Require:
One link to the site.
Key Phrases.
Valid when 1 key phrase from the content topic.
Observe the extra functionality!
Essential! Primary links do not intersect with Tier 2 and 3rd-level hyperlinks
A link network is a instrument for increasing the circulation and backlink portfolio of a internet domain or online community
binance us registrace
August 5, 2024 @ 8:25 am
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me?
Matthewadact
August 5, 2024 @ 11:26 am
cost of generic propecia pills: buying cheap propecia price – get generic propecia tablets
Matthewadact
August 5, 2024 @ 1:03 pm
tamoxifen men: tamoxifen citrate – tamoxifen and grapefruit
Matthewadact
August 5, 2024 @ 6:39 pm
buy cytotec over the counter: Misoprostol 200 mg buy online – Abortion pills online
Lewiscoata
August 5, 2024 @ 7:48 pm
http://zithromaxbestprice.pro/# can you buy zithromax over the counter
Matthewadact
August 5, 2024 @ 8:17 pm
cortisol prednisone: buy prednisone online uk – prednisone for sale online
Carlosses
August 6, 2024 @ 1:01 am
does tamoxifen cause weight loss: tamoxifen – how does tamoxifen work
Carlosses
August 6, 2024 @ 1:26 am
nolvadex half life: tamoxifenworld – cost of tamoxifen
Matthewadact
August 6, 2024 @ 1:49 am
cost of propecia pills: generic propecia pill – buy generic propecia
Matthewadact
August 6, 2024 @ 3:26 am
zithromax capsules: zithromax cost uk – buy zithromax online
StevenDak
August 6, 2024 @ 3:34 am
prednisone 20 mg purchase http://prednisonebestprice.pro/# prednisone 54899
prednisone nz
Carlosses
August 6, 2024 @ 3:38 am
arimidex vs tamoxifen bodybuilding: tamoxifen benefits – aromatase inhibitors tamoxifen
StevenDak
August 6, 2024 @ 4:27 am
5 mg prednisone daily http://prednisonebestprice.pro/# buy prednisone online usa
20mg prednisone
StevenDak
August 6, 2024 @ 4:30 am
prednisone 10 mg coupon https://prednisonebestprice.pro/# prednisone 20mg for sale
can you buy prednisone in canada
Matthewadact
August 6, 2024 @ 9:04 am
п»їcytotec pills online: order cytotec online – cytotec pills buy online
payang24hr.com
August 6, 2024 @ 10:31 am
For most up-to-date news you have to go to see world wide web and on internet I found
this web site as a finest web page for hottest
updates.
Matthewadact
August 6, 2024 @ 10:41 am
order cytotec online: buy cytotec online – п»їcytotec pills online
payang24hr
August 6, 2024 @ 10:59 am
Write more, thats all I have to say. Literally, it seems as though you relied on the video
to make your point. You definitely know what youre talking about, why throw away
your intelligence on just posting videos to your blog when you could be giving us something informative
to read?
payang24hr.com
August 6, 2024 @ 11:01 am
Awesome issues here. I’m very glad to look your article. Thank you a lot and I
am looking ahead to contact you. Will you please drop me a mail?
payang24hr.com
August 6, 2024 @ 12:12 pm
Just want to say your article is as amazing. The clarity on your post is just spectacular and i can suppose you are
an expert in this subject. Fine along with your permission let me to
clutch your feed to stay updated with imminent post. Thank you a million and please continue the enjoyable work.
payang24hr.com
August 6, 2024 @ 12:17 pm
Appreciating the dedication you put into your website and detailed information you provide.
It’s great to come across a blog every once in a while that isn’t the same old rehashed material.
Excellent read! I’ve bookmarked your site and
I’m including your RSS feeds to my Google account.
payang24hr.com
August 6, 2024 @ 12:30 pm
certainly like your web-site however you have to check the spelling
on several of your posts. Many of them are rife with
spelling issues and I to find it very bothersome to tell
the truth however I will surely come back again.
ปะยางนอกสถานที่
August 6, 2024 @ 12:52 pm
Howdy! This is my 1st comment here so I just wanted to give a quick shout out and say I truly enjoy reading your posts.
Can you suggest any other blogs/websites/forums that cover the same topics?
Thanks a lot!
ปะยาง
August 6, 2024 @ 1:14 pm
It’s difficult to find knowledgeable people about this subject, however, you seem like
you know what you’re talking about! Thanks
Matthewadact
August 6, 2024 @ 4:10 pm
buy cheap propecia without a prescription: order generic propecia pill – buying generic propecia pills
Matthewadact
August 6, 2024 @ 5:45 pm
cost propecia without a prescription: propecia online – how cЙ‘n i get cheap propecia pills
payang24hr.com
August 6, 2024 @ 9:57 pm
For newest news you have to pay a visit world wide web and on world-wide-web I found
this website as a finest website for most recent updates.
payang24hr
August 6, 2024 @ 10:31 pm
Hi to all, it’s really a pleasant for me to visit this
web site, it consists of important Information.
ปะยาง
August 6, 2024 @ 10:33 pm
Hey there just wanted to give you a brief heads up and let you know a few
of the images aren’t loading correctly. I’m not sure
why but I think its a linking issue. I’ve tried it in two different
web browsers and both show the same outcome.
Tomasstync
August 7, 2024 @ 10:57 am
comprare farmaci online all’estero: avanafil generico – п»їFarmacia online migliore
TimothyLew
August 7, 2024 @ 12:01 pm
https://cialisgenerico.life/# Farmacie online sicure
RandyDox
August 7, 2024 @ 2:03 pm
farmacia online senza ricetta: Avanafil prezzo – farmacie online autorizzate elenco
RandyDox
August 7, 2024 @ 2:07 pm
viagra cosa serve: viagra farmacia – viagra online in 2 giorni
Tomasstync
August 7, 2024 @ 3:34 pm
farmacia online: Avanafil 50 mg – farmaci senza ricetta elenco
TimothyLew
August 7, 2024 @ 4:55 pm
http://avanafil.pro/# farmacia online
Tomasstync
August 7, 2024 @ 6:51 pm
acquisto farmaci con ricetta: kamagra gel – farmacia online piГ№ conveniente
RandyDox
August 7, 2024 @ 8:59 pm
farmacie online affidabili: Cialis generico 20 mg 8 compresse prezzo – Farmacie online sicure
Tomasstync
August 7, 2024 @ 11:16 pm
Farmacia online piГ№ conveniente: Cialis generico prezzo – farmaci senza ricetta elenco
ปะยางนอกสถานที่หาดใหญ่
August 8, 2024 @ 12:10 am
These are really great ideas in about blogging. You have touched some good points here.
Any way keep up wrinting.
Tomasstync
August 8, 2024 @ 2:37 am
acquistare farmaci senza ricetta: Farmacie online sicure – farmaci senza ricetta elenco
RandyDox
August 8, 2024 @ 4:11 am
farmacie online autorizzate elenco: Cialis generico recensioni – comprare farmaci online con ricetta
Tomasstync
August 8, 2024 @ 7:21 am
miglior sito per comprare viagra online: viagra farmacia – viagra ordine telefonico
Tomasstync
August 8, 2024 @ 10:47 am
п»їFarmacia online migliore: Cialis generico controindicazioni – farmacia online piГ№ conveniente
Tomasstync
August 8, 2024 @ 3:35 pm
Farmacie on line spedizione gratuita: avanafil senza ricetta – migliori farmacie online 2024
Tomasstync
August 8, 2024 @ 7:02 pm
farmacie online autorizzate elenco: Cialis generico farmacia – top farmacia online
RandyDox
August 9, 2024 @ 2:25 am
farmacia senza ricetta recensioni: acquisto viagra – viagra generico sandoz
Tomasstync
August 9, 2024 @ 3:36 am
comprare farmaci online all’estero: Avanafil 50 mg – farmacia online senza ricetta
Tomasstync
August 9, 2024 @ 7:07 am
viagra naturale: acquisto viagra – alternativa al viagra senza ricetta in farmacia
GeorgeZed
August 9, 2024 @ 2:05 pm
viagra coupon: Viagra online price – 100 mg viagra lowest price
GeorgeZed
August 9, 2024 @ 2:09 pm
viagra cialis canada: Buy Tadalafil 20mg – cialis online from canada
SamuelGop
August 9, 2024 @ 4:51 pm
http://sildenafil.llc/# viagra side effects
SamuelGop
August 9, 2024 @ 9:10 pm
https://tadalafil.auction/# cialis with dapoxetine without prescription mastercard
GeorgeZed
August 9, 2024 @ 10:12 pm
viagra generic: Buy Viagra online in USA – viagra for sale
GeorgeZed
August 9, 2024 @ 10:16 pm
viagra professional: Viagra without a doctor prescription – blue pill viagra
SamuelGop
August 10, 2024 @ 6:46 am
http://sildenafil.llc/# online viagra
GeorgeZed
August 10, 2024 @ 7:34 am
100mg viagra without a doctor prescription: Viagra without a doctor prescription – viagra generic
SamuelGop
August 10, 2024 @ 12:12 pm
https://tadalafil.auction/# cialis usa
GeorgeZed
August 10, 2024 @ 4:57 pm
cialis vs viagra: Viagra without a doctor prescription – canadian viagra
GeorgeZed
August 10, 2024 @ 5:02 pm
cialis canadian pharmacy: Generic Cialis without a doctor prescription – where to buy generic cialis
Charlescaulk
August 10, 2024 @ 11:13 pm
http://mexicopharmacy.win/# mexico drug stores pharmacies
where can i get ed pills
Douglasidomb
August 11, 2024 @ 3:14 am
online ed pharmacy: get ed meds online – low cost ed pills
Douglasidomb
August 11, 2024 @ 3:17 am
Online medicine order: Indian pharmacy international shipping – top 10 pharmacies in india
Charlescaulk
August 11, 2024 @ 4:02 am
http://mexicopharmacy.win/# mexican drugstore online
ed pills for sale
Charlescaulk
August 11, 2024 @ 6:46 am
https://indiapharmacy.shop/# top 10 pharmacies in india
online erectile dysfunction pills
Charlescaulk
August 11, 2024 @ 10:34 am
https://mexicopharmacy.win/# medication from mexico pharmacy
low cost ed meds
Douglasidomb
August 11, 2024 @ 11:21 am
Online medicine home delivery: Top mail order pharmacies – world pharmacy india
Douglasidomb
August 11, 2024 @ 11:25 am
mail order pharmacy india: Online pharmacy – online pharmacy india
Charlescaulk
August 11, 2024 @ 1:22 pm
http://edpillpharmacy.store/# low cost ed medication
erectile dysfunction meds online
Charlescaulk
August 11, 2024 @ 5:10 pm
http://edpillpharmacy.store/# cheapest ed pills
best ed meds online
Douglasidomb
August 11, 2024 @ 7:19 pm
ed treatments online: Cheapest online ED treatment – best online ed pills
Douglasidomb
August 11, 2024 @ 7:23 pm
indian pharmacies safe: Online pharmacy USA – cheapest online pharmacy india
Charlescaulk
August 11, 2024 @ 7:54 pm
http://edpillpharmacy.store/# best online ed meds
п»їed pills online
Douglasidomb
August 12, 2024 @ 3:25 am
buying prescription drugs in mexico online: Medicines Mexico – mexico drug stores pharmacies
Douglasidomb
August 12, 2024 @ 3:29 am
cheapest online pharmacy india: Online pharmacy USA – world pharmacy india
Leonardginge
August 12, 2024 @ 5:25 am
https://edpillpharmacy.store/# online ed drugs
Douglasidomb
August 12, 2024 @ 11:54 am
edmeds: ED meds online with insurance – cheap ed meds
Douglasidomb
August 12, 2024 @ 11:58 am
top online pharmacy india: Online medicine home delivery – п»їlegitimate online pharmacies india
Douglasidomb
August 12, 2024 @ 7:34 pm
mexico pharmacies prescription drugs: Purple pharmacy online ordering – reputable mexican pharmacies online
Douglasidomb
August 12, 2024 @ 7:37 pm
erectile dysfunction online prescription: Cheapest online ED treatment – erectile dysfunction online prescription
Douglasidomb
August 13, 2024 @ 2:52 am
mexico drug stores pharmacies: mexico pharmacy win – mexican drugstore online
Douglasidomb
August 13, 2024 @ 2:55 am
buy medicines online in india: Indian pharmacy international shipping – best india pharmacy
Douglasidomb
August 13, 2024 @ 10:55 am
best ed meds online: cheap ed medicine – ed treatments online
Leonardginge
August 13, 2024 @ 2:11 pm
https://edpillpharmacy.store/# ed online prescription
Douglasidomb
August 13, 2024 @ 7:07 pm
buy medicines online in india: Online pharmacy – india pharmacy mail order
Douglasidomb
August 13, 2024 @ 7:12 pm
pharmacies in mexico that ship to usa: Best pharmacy in Mexico – mexican pharmaceuticals online
Douglasidomb
August 14, 2024 @ 2:48 am
buy ed meds: ed pills online – where can i buy ed pills
Leonardginge
August 14, 2024 @ 9:48 am
https://mexicopharmacy.win/# mexico pharmacies prescription drugs
Douglasidomb
August 14, 2024 @ 10:24 am
mexican pharmaceuticals online: Best pharmacy in Mexico – mexico pharmacies prescription drugs
Douglasidomb
August 14, 2024 @ 10:28 am
mexican mail order pharmacies: mexican pharmacy – mexico drug stores pharmacies
Douglasidomb
August 14, 2024 @ 6:05 pm
indian pharmacy: Cheapest online pharmacy – india pharmacy
Software Klinik
August 15, 2024 @ 1:52 am
With Assist id, make your health a top priority without the hassle of long queues at the clinic.
Kampus Digital
August 15, 2024 @ 1:56 am
Being a digital marketing freelancer requires considerable skills to convince prospective clien, you can explore your digital marketing skills more deeply together Argiaacademy
Leonardginge
August 15, 2024 @ 8:28 am
http://mexicopharmacy.win/# mexican rx online
Douglasidomb
August 15, 2024 @ 9:26 am
ed pills for sale: ED meds online with insurance – low cost ed meds
Douglasidomb
August 15, 2024 @ 9:30 am
pharmacy website india: Online India pharmacy – top 10 pharmacies in india
Douglasidomb
August 15, 2024 @ 4:46 pm
medicine in mexico pharmacies: mexico pharmacy win – п»їbest mexican online pharmacies
Douglasidomb
August 15, 2024 @ 4:49 pm
world pharmacy india: Top online pharmacy in India – buy prescription drugs from india
travel blog
August 16, 2024 @ 12:17 pm
Very interesting details you have mentioned, thanks for posting.
Euro travel
Douglasidomb
August 16, 2024 @ 12:42 pm
reputable indian online pharmacy: Best Indian pharmacy – online pharmacy india
Douglasidomb
August 16, 2024 @ 12:45 pm
mexico drug stores pharmacies: Best online Mexican pharmacy – pharmacies in mexico that ship to usa
Douglasidomb
August 16, 2024 @ 7:27 pm
mexican border pharmacies shipping to usa: Certified Mexican pharmacy – reputable mexican pharmacies online
DanielTam
August 18, 2024 @ 8:55 pm
purchase cytotec http://furosemide.win/# lasix 100mg
lasix 20 mg
Stanleyquota
August 18, 2024 @ 10:24 pm
nolvadex during cycle buy tamoxifen online tamoxifen bone pain
StephenTip
August 19, 2024 @ 3:47 am
https://furosemide.win/# furosemide 100mg
Stanleyquota
August 19, 2024 @ 6:39 am
buy generic lipitor Atorvastatin 20 mg buy online lipitor 10mg price australia
DanielTam
August 19, 2024 @ 6:43 am
buy cytotec online https://lipitor.guru/# lipitor 20 mg daily
lasix side effects
StephenTip
August 19, 2024 @ 8:00 am
https://tamoxifen.bid/# tamoxifen and ovarian cancer
JamesTah
August 19, 2024 @ 9:02 am
lasix 40 mg: cheap lasix – lasix furosemide
StephenTip
August 19, 2024 @ 3:27 pm
https://tamoxifen.bid/# aromatase inhibitors tamoxifen
Stanleyquota
August 19, 2024 @ 3:31 pm
tamoxifen adverse effects buy tamoxifen citrate tamoxifen for gynecomastia reviews
DanielTam
August 19, 2024 @ 4:33 pm
buy cytotec in usa https://lisinopril.guru/# zestril drug
lasix uses
JamesTah
August 19, 2024 @ 4:49 pm
50 mg lisinopril: buy lisinopril – lisinopril tabs 20mg
DanielTam
August 19, 2024 @ 6:40 pm
Cytotec 200mcg price https://lipitor.guru/# lipitor drug prices
lasix generic name
StephenTip
August 19, 2024 @ 7:31 pm
https://cytotec.pro/# п»їcytotec pills online
Stanleyquota
August 19, 2024 @ 9:48 pm
lisinopril 2.5 tablet Lisinopril refill online lisinopril for sale uk
DanielTam
August 20, 2024 @ 3:05 am
buy cytotec over the counter https://tamoxifen.bid/# hysterectomy after breast cancer tamoxifen
lasix uses
StephenTip
August 20, 2024 @ 5:23 am
https://lisinopril.guru/# lisinopril 20 mg for sale
DanielTam
August 20, 2024 @ 6:29 am
cytotec online https://furosemide.win/# furosemide 40 mg
lasix 20 mg
StephenTip
August 20, 2024 @ 10:40 am
http://lipitor.guru/# lipitor lowest price
Stanleyquota
August 20, 2024 @ 3:17 pm
furosemide 100 mg furosemide online lasix generic
sex
August 20, 2024 @ 4:19 pm
Thank you very much for sharing, I learned a lot from your article. Very cool. Thanks.
DanielTam
August 20, 2024 @ 5:09 pm
Misoprostol 200 mg buy online http://furosemide.win/# lasix 40mg
buy lasix online
MatthewInalf
August 20, 2024 @ 5:15 pm
apple сервис центр
StephenTip
August 20, 2024 @ 7:58 pm
http://lisinopril.guru/# lisinopril 15mg
StephenTip
August 21, 2024 @ 1:18 am
https://cytotec.pro/# cytotec buy online usa
DanielTam
August 21, 2024 @ 7:46 am
cytotec abortion pill http://tamoxifen.bid/# tamoxifen effectiveness
furosemide 100mg
StephenTip
August 21, 2024 @ 10:34 am
https://lisinopril.guru/# lisinopril australia
StephenTip
August 21, 2024 @ 3:39 pm
https://cytotec.pro/# Cytotec 200mcg price
JamesTah
August 21, 2024 @ 4:38 pm
order lipitor: cheapest ace inhibitor – how much is lipitor
StephenTip
August 22, 2024 @ 12:18 am
https://furosemide.win/# furosemide 40 mg
JamesTah
August 22, 2024 @ 8:09 am
lasix 40mg: furosemida 40 mg – furosemide 40mg
MatthewInalf
August 22, 2024 @ 8:43 am
мастер айфон москва
doramasmp4
August 22, 2024 @ 9:37 am
https://doramasmp4.com.mx/ _It is a good and great article.
MatthewInalf
August 22, 2024 @ 5:25 pm
сервис по ремонту телефонов
JamesTah
August 23, 2024 @ 4:40 am
purchase cytotec: Misoprostol price in pharmacy – Misoprostol 200 mg buy online
JamesTah
August 23, 2024 @ 11:30 am
does tamoxifen make you tired: buy tamoxifen citrate – aromatase inhibitor tamoxifen
MatthewInalf
August 23, 2024 @ 1:11 pm
сервисный ремонт apple
MatthewInalf
August 24, 2024 @ 5:42 am
ремонт телефонов ближайший ко мне
Rickynairm
August 24, 2024 @ 11:27 pm
mexican rx online here buying prescription drugs in mexico online
reputable mexican pharmacies online
Rickynairm
August 24, 2024 @ 11:33 pm
mexican rx online more mexican online pharmacies prescription drugs
mexican pharmaceuticals online
Rickynairm
August 24, 2024 @ 11:49 pm
mexico pharmacies prescription drugs MexStar Pharma п»їbest mexican online pharmacies
buying prescription drugs in mexico
Jamesmalge
August 24, 2024 @ 11:54 pm
top 10 online pharmacy in india here buy medicines online in india
world pharmacy india
Rickynairm
August 25, 2024 @ 12:08 am
best online pharmacies in mexico here mexico pharmacies prescription drugs
reputable mexican pharmacies online
BryantRaics
August 25, 2024 @ 12:10 am
canadian king pharmacy rx canada my canadian pharmacy reviews
is canadian pharmacy legit
Rickynairm
August 25, 2024 @ 12:22 am
mexico drug stores pharmacies Mex Star Pharma mexican pharmaceuticals online
buying prescription drugs in mexico
Rickynairm
August 25, 2024 @ 12:37 am
buying prescription drugs in mexico online MexStar Pharma buying from online mexican pharmacy
mexican rx online
Rickynairm
August 25, 2024 @ 1:10 am
п»їbest mexican online pharmacies Mex Star Pharma mexican mail order pharmacies
medication from mexico pharmacy
BryantRaics
August 25, 2024 @ 1:32 am
northwest canadian pharmacy EasyRxCanada best canadian pharmacy to buy from
my canadian pharmacy rx
BryantRaics
August 25, 2024 @ 1:39 am
cheap canadian pharmacy online website canadian pharmacies compare
canadian family pharmacy
Rickynairm
August 25, 2024 @ 1:54 am
reputable mexican pharmacies online MexStarPharma mexican border pharmacies shipping to usa
mexican pharmaceuticals online
BryantRaics
August 25, 2024 @ 1:58 am
canadian pharmacy ratings read canadian pharmacy checker
canadian pharmacy mall
Rickynairm
August 25, 2024 @ 2:03 am
medication from mexico pharmacy more mexican drugstore online
best online pharmacies in mexico
Rickynairm
August 25, 2024 @ 2:12 am
mexican mail order pharmacies mexstarpharma.com mexican drugstore online
medication from mexico pharmacy
Rickynairm
August 25, 2024 @ 2:15 am
mexican rx online view buying from online mexican pharmacy
mexican online pharmacies prescription drugs
Rickynairm
August 25, 2024 @ 2:18 am
mexican drugstore online view mexican pharmaceuticals online
pharmacies in mexico that ship to usa
MelvinBog
August 25, 2024 @ 3:02 am
buying drugs from canada https://easyrxcanada.online/ reputable canadian online pharmacies
canada pharmacy
MelvinBog
August 25, 2024 @ 3:16 am
recommended canadian pharmacies https://easyrxcanada.online/ 77 canadian pharmacy
canadian pharmacy store
MelvinNop
August 25, 2024 @ 3:20 am
mexico pharmacies prescription drugs https://mexstarpharma.com/ medicine in mexico pharmacies
reputable mexican pharmacies online
MelvinNop
August 25, 2024 @ 3:31 am
buying from online mexican pharmacy https://mexstarpharma.com/ mexican drugstore online
mexican drugstore online
MelvinNop
August 25, 2024 @ 3:36 am
best online pharmacies in mexico https://mexstarpharma.online/ buying from online mexican pharmacy
mexico drug stores pharmacies
MelvinNop
August 25, 2024 @ 3:45 am
pharmacies in mexico that ship to usa https://mexstarpharma.com/ mexico pharmacies prescription drugs
mexican rx online
MelvinBog
August 25, 2024 @ 3:47 am
canadian pharmacy cheap https://easyrxcanada.com/ canadian pharmacy online reviews
canadian pharmacy
MelvinNop
August 25, 2024 @ 3:59 am
mexico drug stores pharmacies https://mexstarpharma.com/ mexican rx online
buying from online mexican pharmacy
MelvinBog
August 25, 2024 @ 4:08 am
canada pharmacy online https://easyrxcanada.online/ legitimate canadian online pharmacies
best canadian pharmacy
MelvinBog
August 25, 2024 @ 4:21 am
best canadian pharmacy to order from http://easyrxcanada.com/ canadian online pharmacy
canadian valley pharmacy
MelvinNop
August 25, 2024 @ 4:36 am
buying from online mexican pharmacy https://mexstarpharma.online/ purple pharmacy mexico price list
mexican online pharmacies prescription drugs
MelvinNop
August 25, 2024 @ 4:40 am
best online pharmacies in mexico http://mexstarpharma.com/ medication from mexico pharmacy
buying prescription drugs in mexico
MelvinNop
August 25, 2024 @ 4:50 am
medication from mexico pharmacy http://mexstarpharma.com/ pharmacies in mexico that ship to usa
mexico drug stores pharmacies
MelvinNop
August 25, 2024 @ 5:20 am
mexican pharmaceuticals online https://mexstarpharma.com/ medicine in mexico pharmacies
mexico drug stores pharmacies
MelvinBog
August 25, 2024 @ 5:26 am
canadian pharmacy victoza https://easyrxcanada.online/ canadian family pharmacy
reliable canadian pharmacy
PeterMeeft
August 25, 2024 @ 5:36 am
online pharmacy india http://easyrxindia.com/ best online pharmacy india
buy prescription drugs from india
MelvinBog
August 25, 2024 @ 5:45 am
the canadian drugstore https://easyrxcanada.com/ canadian pharmacy scam
pharmacy in canada
MelvinBog
August 25, 2024 @ 6:06 am
canadian pharmacy meds https://easyrxcanada.online/ best online canadian pharmacy
canada rx pharmacy world
MelvinBog
August 25, 2024 @ 6:12 am
canadian pharmacy prices https://easyrxcanada.com/ safe canadian pharmacies
best online canadian pharmacy
MelvinBog
August 25, 2024 @ 6:20 am
canadian discount pharmacy https://easyrxcanada.com/ pharmacy in canada
best canadian pharmacy
MelvinNop
August 25, 2024 @ 6:30 am
reputable mexican pharmacies online https://mexstarpharma.com/ mexican mail order pharmacies
purple pharmacy mexico price list
MelvinNop
August 25, 2024 @ 6:33 am
п»їbest mexican online pharmacies http://mexstarpharma.com/ purple pharmacy mexico price list
buying prescription drugs in mexico
MelvinBog
August 25, 2024 @ 6:39 am
buying from canadian pharmacies https://easyrxcanada.online/ ed drugs online from canada
canadian pharmacy service
MelvinNop
August 25, 2024 @ 6:47 am
medicine in mexico pharmacies https://mexstarpharma.online/ buying prescription drugs in mexico online
buying from online mexican pharmacy
MelvinBog
August 25, 2024 @ 7:06 am
canadian pharmacy in canada https://easyrxcanada.online/ rate canadian pharmacies
canadian discount pharmacy
AnthonyGep
August 25, 2024 @ 8:10 am
canadian drug prices canadian discount pharmacy canadian pharmacy prices
AnthonyGep
August 25, 2024 @ 10:02 am
п»їbest mexican online pharmacies buying prescription drugs in mexico mexican pharmaceuticals online
Hermanlor
August 25, 2024 @ 3:17 pm
top 10 pharmacies in india: best online pharmacy india – indian pharmacy online
Jeremytop
August 25, 2024 @ 6:10 pm
https://easyrxindia.com/# online shopping pharmacy india
AnthonyGep
August 25, 2024 @ 11:48 pm
my canadian pharmacy rx canadian online drugstore onlinecanadianpharmacy 24
Hermanlor
August 26, 2024 @ 2:49 am
Online medicine order: india pharmacy mail order – best india pharmacy
Hermanlor
August 26, 2024 @ 7:57 am
top 10 online pharmacy in india: online shopping pharmacy india – buy medicines online in india
PeterMeeft
August 26, 2024 @ 6:25 pm
mail order pharmacy india: india online pharmacy – best online pharmacy india
ремонт телевизоров
August 26, 2024 @ 6:27 pm
ремонт телевизора
Hermanlor
August 26, 2024 @ 7:28 pm
indian pharmacy: top online pharmacy india – top online pharmacy india
binance account creation
August 26, 2024 @ 8:17 pm
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me. https://www.binance.com/bn/register?ref=UM6SMJM3
Ремонт смартфонов
August 26, 2024 @ 9:44 pm
Профессиональный сервисный центр по ремонту сотовых телефонов, смартфонов и мобильных устройств.
Мы предлагаем: ремонт мобильных телефонов в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Ремонт телефонов
August 26, 2024 @ 11:19 pm
Профессиональный сервисный центр по ремонту сотовых телефонов, смартфонов и мобильных устройств.
Мы предлагаем: ремонт сотовых телефонов в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Hermanlor
August 27, 2024 @ 12:14 am
pharmacy website india: top online pharmacy india – mail order pharmacy india
ремонт macbook в москве
August 27, 2024 @ 5:32 am
Профессиональный сервисный центр по ремонту ноутбуков, макбуков и другой компьютерной техники.
Мы предлагаем:ремонт ноутбуков мак
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Darrelnog
August 27, 2024 @ 9:36 am
en yeni slot siteleri: bonus veren casino slot siteleri – en yeni slot siteleri
Ремонт смартфонов
August 27, 2024 @ 10:54 am
Профессиональный сервисный центр по ремонту сотовых телефонов, смартфонов и мобильных устройств.
Мы предлагаем: сервис по ремонту смартфонов
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Darrelnog
August 27, 2024 @ 11:43 am
slot siteleri: deneme bonusu veren siteler – yasal slot siteleri
ремонт квадрокоптеров москва
August 27, 2024 @ 4:35 pm
Профессиональный сервисный центр по ремонту квадрокоптеров и радиоуправляемых дронов.
Мы предлагаем:ремонт квадрокоптеров в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Kevinnut
August 27, 2024 @ 5:04 pm
https://sweetbonanza.network/# guncel sweet bonanza
Jesusdop
August 27, 2024 @ 5:12 pm
slot kumar siteleri: slot casino siteleri – slot kumar siteleri
Darrelnog
August 27, 2024 @ 6:38 pm
slot kumar siteleri: slot oyun siteleri – deneme bonusu veren siteler
Jesusdop
August 27, 2024 @ 10:22 pm
bonus veren siteler: deneme bonusu – deneme bonusu
Kevinnut
August 28, 2024 @ 5:51 am
https://denemebonusuverensiteler.win/# bonus veren siteler
Darrelnog
August 28, 2024 @ 8:09 am
en iyi slot siteler: slot oyun siteleri – en yeni slot siteleri
Jesusdop
August 28, 2024 @ 8:35 am
bahis siteleri: deneme bonusu veren siteler – deneme bonusu veren siteler
telemundo
August 28, 2024 @ 11:53 pm
A good blog always comes-up with new and exciting information and while reading I have feel that this blog is really have all those quality that qualify a blog to be a one. tusnovelas
JK Anime
August 28, 2024 @ 11:55 pm
Good Day. I recommend this website more than anyone else. wish you luck Anime flv
gogoanime
August 28, 2024 @ 11:56 pm
This is the new website address. I hope it will be a lot of energy and lucky site, 9Anime
gogoanime
August 28, 2024 @ 11:59 pm
This is the new website address. I hope it will be a lot of energy and lucky site gogo anime
ремонт imac в москве
August 29, 2024 @ 4:45 am
Профессиональный сервисный центр по ремонту ноутбуков, imac и другой компьютерной техники.
Мы предлагаем:срочный ремонт аймака
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
expibre
August 30, 2024 @ 3:43 am
priligy price PMID 35069437 Free PMC article
ремонт ноутбуков
August 30, 2024 @ 7:16 am
Профессиональный сервисный центр по ремонту ноутбуков и компьютеров.дронов.
Мы предлагаем:ремонт ноутбуков москва центр
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
expibre
August 30, 2024 @ 11:29 am
EYE DROPS N A CIBA VISION AG SWITZERLAND priligy buy By reducing the availability of poorly liganded Fe II and Fe III, which are needed to catalyze the Haber Weiss and Fenton equations in the generation of reactive oxygen species, NGAL appears to have an important role in limiting oxidative damage in both acute and chronic diseases
ремонту iPhone
August 30, 2024 @ 4:51 pm
центры ремонта iphone
outlookindiasportsm
August 31, 2024 @ 10:04 pm
This layout is amazing! You most surely understand how to engage readers. I was almost inspired to start my own blog because of your humor and videos. Excellent work.
사설 토토사이트
outlookindiapowerballm
August 31, 2024 @ 10:04 pm
You are a highly skilled blogger, and this is very attention-grabbing. I subscribed to your rss feed and am looking forward to reading more of your fantastic content. Additionally, I shared your website on my social media accounts!
파워볼사이트
Kevinnut
September 1, 2024 @ 2:17 am
https://sweetbonanza.network/# sweet bonanza bahis
sightcare official
September 1, 2024 @ 3:52 am
Sight Care, with its natural ingredients and effectiveness, is quickly gaining popularity among those looking to improve their vision.
Kevinnut
September 1, 2024 @ 3:44 pm
https://sweetbonanza.network/# sweet bonanza 100 tl
Darrelnog
September 1, 2024 @ 5:35 pm
guvenilir slot siteleri 2024: slot siteleri guvenilir – bonus veren slot siteleri
Kevinnut
September 1, 2024 @ 9:53 pm
http://slotsiteleri.bid/# 2024 en iyi slot siteleri
Ремонт Apple Watch
September 2, 2024 @ 10:05 pm
ремонт эппл вотч
Ремонт холодильников
September 2, 2024 @ 10:14 pm
Профессиональный сервисный центр по ремонту холодильников и морозильных камер.
Мы предлагаем: ремонт холодильника
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Darrelnog
September 2, 2024 @ 11:07 pm
en guvenilir slot siteleri: deneme bonusu veren siteler – deneme bonusu veren siteler
Tory Yoh
September 3, 2024 @ 3:01 am
Should I submit a writing portfolio to Columbia University if I plan on being a creative writing major?
Ремонт Apple Watch
September 3, 2024 @ 6:45 am
apple watch ремонт
ремонт айпадов в москве
September 3, 2024 @ 2:12 pm
Профессиональный сервисный центр по ремонту планетов в том числе Apple iPad.
Мы предлагаем: ipad ремонт
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
binance h"anvisning
September 3, 2024 @ 2:40 pm
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
ремонт ноутбуков в москве
September 3, 2024 @ 3:47 pm
Профессиональный сервисный центр по ремонту ноутбуков и компьютеров.дронов.
Мы предлагаем:ремонт ноутбуков москва центр
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
ремонт техники в спб
September 3, 2024 @ 4:07 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем:сервисные центры по ремонту техники в спб
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
maria togel login alternatif
September 3, 2024 @ 4:39 pm
I am extremely impressed with your writing skills as well as with
the layout on your blog. Is this a paid theme or did you modify it yourself?
Either way keep up the nice quality writing, it is
rare to see a great blog like this one nowadays.
Darrelnog
September 3, 2024 @ 6:04 pm
canl? slot siteleri: guvenilir slot siteleri – slot siteleri bonus veren
ремонт квадрокоптеров москве
September 3, 2024 @ 9:43 pm
Профессиональный сервисный центр по ремонту радиоуправляемых устройства – квадрокоптеры, дроны, беспилостники в том числе Apple iPad.
Мы предлагаем: ремонт камеры квадрокоптера
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
ремонт бытовой техники в спб
September 3, 2024 @ 9:55 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – ремонт бытовой техники
ремонт бытовой техники в москве
September 3, 2024 @ 10:25 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – сервисный центр в москве
Kevinnut
September 4, 2024 @ 12:45 am
http://slotsiteleri.bid/# guvenilir slot siteleri 2024
ремонт техники профи в екб
September 4, 2024 @ 2:21 am
Если вы искали где отремонтировать сломаную технику, обратите внимание – ремонт бытовой техники
Scottger
September 4, 2024 @ 2:58 am
пин ап вход: pin up – пинап казино
Scottfup
September 4, 2024 @ 4:27 am
вавада: казино вавада – vavada
ремонт ipad
September 4, 2024 @ 5:07 am
Профессиональный сервисный центр по ремонту планетов в том числе Apple iPad.
Мы предлагаем: сервис ipad москва
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
ремонт бытовой техники петербург
September 4, 2024 @ 6:44 am
Если вы искали где отремонтировать сломаную технику, обратите внимание – ремонт бытовой техники в спб
ремонт ipad в москве
September 4, 2024 @ 7:06 am
Профессиональный сервисный центр по ремонту радиоуправляемых устройства – квадрокоптеры, дроны, беспилостники в том числе Apple iPad.
Мы предлагаем: ремонт камеры квадрокоптера
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Scottfup
September 4, 2024 @ 10:29 am
pin up: пинап казино – pin up казино
LeslieFon
September 4, 2024 @ 11:59 am
пин ап pin up пин ап казино вход
WarrenByday
September 4, 2024 @ 12:05 pm
http://1xbet.contact/# 1хбет официальный сайт
ремонт ноутбуков в москве
September 4, 2024 @ 12:15 pm
Профессиональный сервисный центр по ремонту ноутбуков и компьютеров.дронов.
Мы предлагаем:починка ноутбуков
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
ремонт техники в спб
September 4, 2024 @ 12:51 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем:сервисные центры в санкт петербурге
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
ремонт бытовой техники
September 4, 2024 @ 2:08 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – профи тех сервис москва
сервис профи екатеринбург
September 4, 2024 @ 2:23 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – профи екб
Scottfup
September 4, 2024 @ 2:46 pm
1xbet зеркало рабочее на сегодня: 1хбет – 1xbet официальный сайт
Scottfup
September 4, 2024 @ 8:50 pm
1xbet зеркало: 1хбет – 1xbet зеркало рабочее на сегодня
piff bar carts
September 4, 2024 @ 9:59 pm
Helpful info. Fortunate me I found your site by chance, and I am shocked why this accident did not took place earlier!
I bookmarked it.
ремонт техники профи в москве
September 4, 2024 @ 10:54 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – сервис центр в москве
Scottfup
September 5, 2024 @ 1:09 am
1вин официальный сайт: 1win официальный сайт – 1вин
Scottger
September 5, 2024 @ 2:39 am
пинап казино: pin up – пин ап казино вход
WarrenByday
September 5, 2024 @ 3:08 am
http://1xbet.contact/# 1хбет зеркало
ハンクメルツ
September 5, 2024 @ 3:59 am
Join Slope Game to feel the thrill and excitement in every moment.
porn
September 5, 2024 @ 4:22 am
Excellent, what a webpage it is! This weblog provides valuable facts to
us, keep it up.
Scottger
September 5, 2024 @ 5:41 am
пин ап казино: пин ап вход – пин ап зеркало
Scottfup
September 5, 2024 @ 7:12 am
1win официальный сайт: 1win вход – 1win
WarrenByday
September 5, 2024 @ 8:45 am
http://1xbet.contact/# 1xbet зеркало
Scottfup
September 5, 2024 @ 11:34 am
1вин сайт: 1win зеркало – 1win вход
ремонт техники профи в новосибирск
September 5, 2024 @ 5:23 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – ремонт бытовой техники в новосибирск
Ремонт Айфонов
September 5, 2024 @ 5:43 pm
Профессиональный сервисный центр по ремонту Apple iPhone в Москве.
Мы предлагаем: вызвать мастера по ремонту iphone
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Ремонт телефонов
September 5, 2024 @ 5:48 pm
сервисные центры по ремонту мобильных телефонов
ремонт телевизоров в москве
September 5, 2024 @ 6:01 pm
ремонт телевизора на дому в москве
Ремонт ИБП в Москве
September 5, 2024 @ 6:03 pm
Профессиональный сервисный центр по ремонту источников бесперебойного питания.
Мы предлагаем: сервис ибп
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
WarrenByday
September 5, 2024 @ 6:21 pm
http://vavada.auction/# вавада рабочее зеркало
Scottfup
September 5, 2024 @ 9:31 pm
1xbet: 1xbet официальный сайт – 1хбет официальный сайт
WarrenByday
September 5, 2024 @ 11:33 pm
http://vavada.auction/# vavada casino
ремонт бытовой техники новосибирск
September 6, 2024 @ 2:36 am
Если вы искали где отремонтировать сломаную технику, обратите внимание – сервис центр в новосибирске
Scottfup
September 6, 2024 @ 2:58 am
1xbet зеркало рабочее на сегодня: 1xbet официальный сайт – 1xbet скачать
Ремонт Айфонов
September 6, 2024 @ 3:29 am
Профессиональный сервисный центр по ремонту Apple iPhone в Москве.
Мы предлагаем: мастер ремонта apple
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Ремонт ИБП в Москве
September 6, 2024 @ 3:54 am
Профессиональный сервисный центр по ремонту источников бесперебойного питания.
Мы предлагаем: мастер по ремонту ибп
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Scottfup
September 6, 2024 @ 7:08 am
1xbet зеркало рабочее на сегодня: 1xbet скачать – 1хбет
WarrenByday
September 6, 2024 @ 8:57 am
https://1xbet.contact/# 1xbet зеркало рабочее на сегодня
ремонт бытовой техники барнаул
September 6, 2024 @ 11:51 am
Если вы искали где отремонтировать сломаную технику, обратите внимание – сервис центр в барнауле
Scottfup
September 6, 2024 @ 12:43 pm
1хбет официальный сайт: зеркало 1хбет – зеркало 1хбет
сервис профи челябинск
September 6, 2024 @ 1:57 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – профи услуги
WarrenByday
September 6, 2024 @ 2:08 pm
http://pin-up.diy/# пин ап казино
Scottfup
September 6, 2024 @ 4:37 pm
пин ап вход: pin up casino – pin up
ремонт телевизоров с выездом на дом
September 6, 2024 @ 4:38 pm
мастерские по ремонту телевизоров в москве
сервис профи барнаул
September 6, 2024 @ 9:02 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – профи тех сервис барнаул
Jesseciz
September 6, 2024 @ 9:21 pm
The Value of Resonance Mitigation Tools in Mechanical Systems
In manufacturing contexts, machines and turning systems act as the core of production. However, one of the most common problems that might affect the functionality and lifespan is vibrations. Resonance could cause a variety of problems, including reduced perfection as well as efficiency to increased damage, in the end resulting in pricey downtime and repairs. This is when vibration management tools becomes critical.
Why Vibration Management proves Important
Resonance within machinery may bring about several adverse consequences:
Decreased Operational Productivity: Excessive resonance might cause discrepancies and instability, reducing total effectiveness in the systems. Such could lead to reduced manufacturing speed as well as greater energy consumption.
Increased Wear and Tear: Continuous vibration increases total damage to machine components, leading to more regular maintenance along with the chance for unanticipated unexpected issues. Such a situation doesn’t merely heightens production expenses but also shortens the durability of your equipment.
Protection Hazards: Excessive resonance can bring significant safety concerns for the machinery and the equipment as well as the operators. In extreme situations, severe cases, these can cause devastating equipment failure, endangering workers and causing significant devastation to the environment.
Precision and Production Quality Issues: For fields that require high precision, such as manufacturing or aerospace, oscillations may lead to inaccuracies during the production process, producing flawed products along with greater waste.
Affordable Solutions for Vibration Management
Putting money in the vibration management equipment is not merely necessary but a sound investment for any company involved with machinery. Our state-of-the-art vibration control systems are designed to engineered to reduce oscillation in all machinery or spinning equipment, ensuring uninterrupted and efficient operations.
Something that distinguishes our systems above the rest is its cost-effectiveness. We understand the significance of keeping costs low in the modern competitive marketplace, which is why our offerings include high-quality vibration regulation systems at costs that remain budget-friendly.
By choosing our systems, you aren’t simply securing your equipment along with improving its operational effectiveness but also putting resources towards the long-term success in your organization.
Conclusion
Vibration control remains a critical component in preserving the operational performance, safety, as well as lifetime of your equipment. Through our cost-effective oscillation control systems, it is possible to be certain your operations function efficiently, all manufactured items remain top-tier, as well as your employees stay safe. Don’t permit vibrations affect your company—put money in the proper tools today.
сервис профи челябинск
September 6, 2024 @ 10:58 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – тех профи
ремонт бытовой техники в екатеринбурге
September 6, 2024 @ 11:12 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем:ремонт бытовой техники в екб
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Ремонт варочных панелей в Москве
September 6, 2024 @ 11:13 pm
Профессиональный сервисный центр по ремонту варочных панелей и индукционных плит.
Мы предлагаем: ремонт варочных панелей на дому в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
LelandNix
September 7, 2024 @ 4:39 am
jakie prawo jazdy na quada
Jeffreyvef
September 8, 2024 @ 1:10 am
https://pharm24on.com/# people pharmacy zocor
24 store pharmacy reviews
ремонт техники в екб
September 8, 2024 @ 2:46 am
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем:сервисные центры в екатеринбурге
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Ремонт варочных панелей в Москве
September 8, 2024 @ 2:51 am
Профессиональный сервисный центр по ремонту варочных панелей и индукционных плит.
Мы предлагаем: сервис варочных панелей
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Jeffreyvef
September 8, 2024 @ 4:13 am
https://pharm24on.com/# rx pharmacy cialis
viagra pharmacy reviews online
Playmod
September 8, 2024 @ 7:52 am
I really like this article. You can send any article like this. Playmods
DrstoEquak
September 8, 2024 @ 10:28 am
adipex online pharmacy diet pills: Kemadrin – northern pharmacy
ремонт фотоаппаратов в москве
September 8, 2024 @ 10:29 am
отремонтировать цифровой фотоаппарат
Jeffreyvef
September 8, 2024 @ 1:14 pm
https://onlineph24.com/# bactrim uk pharmacy
viagra at tesco pharmacy
DrstoEquak
September 8, 2024 @ 3:39 pm
wellbutrin target pharmacy: pro cialis pharmacy – AebgtydaY
Jeffreyvef
September 8, 2024 @ 3:49 pm
https://drstore24.com/# target finasteride pharmacy
target pharmacy nexium
ремонт фотоаппаратов в москве
September 8, 2024 @ 8:22 pm
ремонт затвора фотоаппарата
C.Lewis
September 8, 2024 @ 9:20 pm
Hey there! I’m Charles, your guide to generating income in your sleep– well, practically. Welcome to the 1K a Day System, where we turn your digital dreams into cold difficult money. Are you prepared to stop scrolling and start earning? Let’s ditch those cent techniques and get ready for some major bank. Join us, and let’s hit those $1K days together!
phieuguige-grab-bat-net
September 8, 2024 @ 10:09 pm
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me. phieuguige-grab-bat-net
Jeffreyvef
September 9, 2024 @ 12:57 am
https://drstore24.com/# priligy online pharmacy
ventolin target pharmacy
DrstoEquak
September 9, 2024 @ 1:38 am
pharmacy price of cialis: online mexican pharmacy – pharmacy one rx
Jeffreyvef
September 9, 2024 @ 3:39 am
https://drstore24.com/# costa rica pharmacy percocet
online international pharmacy
glo cart
September 9, 2024 @ 6:13 am
WOW just what I was looking for. Came here by searching for
glo extracts
DrstoEquak
September 9, 2024 @ 6:51 am
Nitroglycerin: fanda pharmacy hong kong cialis – mypharmacy
Charles
September 9, 2024 @ 7:49 am
Hi, it’s Charles here, concerning you from the land of limitless chance– or as we like to call it, the 1K a Day System. Here, we teach you how to make more than a well-fed squirrel collects nuts for the winter season. If you’re prepared to pile up those digital acorns, hop on board! Let’s make your checking account as plump as those saucy critters by registering today.
Jesseciz
September 9, 2024 @ 9:36 am
vibration analysis
The Value of Vibration Regulation Equipment in Machines
Across production contexts, machines along with rotational equipment constitute the support of production. However, a of the most widespread concerns which might obstruct the functionality and lifespan remains vibrations. Vibration might bring about an array of issues, such as decreased perfection as well as productivity resulting in heightened damage, finally bringing about costly downtime as well as maintenance. This is where resonance control apparatus becomes essential.
Why Oscillation Control proves Necessary
Vibration inside industrial equipment might lead to several detrimental impacts:
Lowered Functional Effectiveness: Excessive vibrations might cause misalignment along with unbalance, lowering the effectiveness of such equipment. This can bring about slower production schedules and elevated electricity usage.
Heightened Erosion: Ongoing vibration accelerates overall erosion to mechanical parts, causing more frequent maintenance and an chance for unexpected breakdowns. Such a scenario not only elevates operational costs but also decreases the durability for the machinery.
Security Hazards: Excessive vibrations can pose substantial security risks to the machinery and the machines and the workers. In, severe cases, such vibrations can cause disastrous equipment failure, threatening operators as well as bringing about extensive damage to the premises.
Exactness along with Manufacturing Quality Issues: Within sectors which require precise production, for example manufacturing and aerospace, vibrations could bring about flaws during production, resulting in defects and more waste.
Reasonably Priced Approaches towards Vibration Regulation
Investing in vibration management systems is not merely a necessity and a wise choice for all businesses that any company involved with equipment. Our modern vibration regulation equipment are designed to intended to reduce oscillation in any equipment or spinning equipment, ensuring seamless and efficient operations.
Something that distinguishes these apparatus above the rest is its its cost-effectiveness. We understand the importance of cost-effectiveness inside the competitive market of today, which is why we provide premium vibration management solutions at prices that won’t break the bank.
By selecting our systems, you’re not only preserving your mechanical systems as well as improving its productivity but also putting investment towards the enduring achievement in your organization.
In Conclusion
Resonance mitigation is an essential element of maintaining the effectiveness, safety, as well as lifetime of your machinery. With these reasonably priced vibration control equipment, you can be certain your operations run smoothly, all goods are of high quality, as well as your workers stay safe. Never let vibrations undermine your machinery—make an investment in the correct apparatus now.
Harryilled
September 9, 2024 @ 9:39 am
plinko real money
Digital casino platforms offer an engaging selection of gaming options, numerous of them now include virtual currency as a method of payment. Among the leading platforms, BC Casino, Panda Casino, Casino Axe, and Kingz Casino are gaining popularity, as Bitstarz Casino is notable through several awards. Cloud Bet Casino is recognized for being a regulated crypto casino, providing user safety and fairness in gameplay, as well as Fairspin together with MB Casino feature an extensive variety of crypto-friendly games.
When it comes to casino dice games, crypto casinos like Bitcoin Dice Casino provide an exciting gambling experience, enabling bettors to stake using Bitcoin and alternative cryptos for example Ether, Litecoin, Dogecoin, Binance Coin, and USDT.
For a majority of online gambling enthusiasts, picking the right casino provider is essential. Thunderkick, Play’n Go Casino, Red Tiger Casino, Quickspin, Pragmatic Play Casino, Playtech, Nolimit City, Net Entertainment, ELK Studios, and Microgaming are known as the leading game providers famous for their unique slot games, high-quality graphics, and easy-to-use interfaces.
Casino streams has grown into an exciting style for bettors to engage with online casinos. Popular streamers like ClassyBeef, Roshtein Casino, Labowsky, DeuceAce, and Xposed stream their gameplay, frequently showing massive jackpots and giving strategies for the best strategies for casino games.
Additionally, casinos such as BC Game Casino, Bitkingz Casino, and Rocketpot also feature Plinko-style games, a fan-favorite game with straightforward mechanics with large possibilities for high payouts.
Comprehending responsible gambling, rebate offers, and anonymous play in virtual casinos is necessary for gamblers trying to improve their gambling journey. Picking a secure wallet, looking for no-registration-required casinos, and getting tips for popular games like Aviator Casino Game helps players to stay informed while playing the excitement of gambling.
Jeffreyvef
September 9, 2024 @ 2:01 pm
https://drstore24.com/# malaysia online pharmacy store
ritalin online pharmacy reviews
DrstoEquak
September 9, 2024 @ 4:17 pm
lisinopril online pharmacy no prescription: people’s pharmacy lisinopril – detrol la online pharmacy
ремонт бытовой техники в челябинске
September 9, 2024 @ 4:23 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – выездной ремонт бытовой техники в челябинске
EasydrFlack
September 9, 2024 @ 4:55 pm
concerta pharmacy prices: finpecia inhouse pharmacy – skelaxin prices pharmacy
EasydrFlack
September 9, 2024 @ 5:00 pm
optimal rx pharmacy: people’s pharmacy prozac – pharmacy online track order
Jeffreyvef
September 9, 2024 @ 5:52 pm
https://pharm24on.com/# lexapro discount pharmacy
online international pharmacy
xnxx
September 9, 2024 @ 6:25 pm
porn
I savour, result in I found just what I was taking a look for.
You have ended my four day long hunt! God Bless you man. Have a great day.
Bye
LelandNix
September 9, 2024 @ 9:38 pm
kat b
DrstoEquak
September 9, 2024 @ 10:01 pm
viagra certified pharmacy: simvastatin uk pharmacy – how much does percocet cost at the pharmacy
LarryWrary
September 10, 2024 @ 12:35 am
Тогда, когда производится замена обвязки, соответственно брус также освобождается от давления и реализуется замена, потому что для проведения замены поднятие не больше десяти сантиметров, которое не выступает критичным в том числе для внутреннего обустройства.
нижний брус из лиственных более долговечней и успешно доказал свою надежность своей надежностью и стойкостью к гниению. Несмотря на это, ее обязательно следует защищать при помощи биоцидного средства, как и прочие стропила.
Наша организация занимается не лишь ремонтом конструкций, дополнительно улучшением полов. Клиенты зачастую оформляют заказ на теплые полы с тепловой изоляцией мы Укомплектовываем клиента материаллом и обеспечиваем выгодные скидки.
888starzPi
September 10, 2024 @ 12:45 am
Для Android доступно скачать 888 starz на андроид бесплатно.
ремонт техники профи в челябинске
September 10, 2024 @ 2:16 am
Если вы искали где отремонтировать сломаную технику, обратите внимание – профи тех сервис челябинск
EasydrFlack
September 10, 2024 @ 4:00 am
meds rx pharmacy: viagra online us pharmacy – online pharmacy sildenafil 100mg
EasydrFlack
September 10, 2024 @ 4:05 am
viagra in malaysia pharmacy: mexican pharmacy abilify – walgreen online pharmacy
SEO Kuntul
September 10, 2024 @ 4:43 am
My spouse and I stumbled over here by a different page and thought I might
check things out. I like what I see so now i am following you.
Look forward to exploring your web page for a second time.
Affiliate Marketing Training
September 10, 2024 @ 6:56 am
Hey there! Just wanted to pop by and inform you how much I admire your blog. Your knowledge in affiliate marketing is genuinely commendable. Making money from home has never been easier thanks to affiliate marketing. It’s all about discovering the perfect goods to promote and developing relationships with your audience. With dedication and persistence, anyone can thrive in this industry. Your blog is a goldmine trove of information for aspiring affiliate marketers. Keep up the great work!
Duanetix
September 10, 2024 @ 7:26 am
Aquel volante de nivelacion con cesta de embrague simboliza un proceso fundamental para garantizar el funcionamiento optimo del propulsor y la impulsion de un camion. El desbalance en esta parte puede producir oscilaciones, sonido, desgaste excesivo de los partes e hasta fallas. Anteriormente, el estabilizacion se realizaba posterior a sacar el volante del propulsion, aunque las tecnologias modernas permiten llevar a cabo este metodo sin intermediarios en el vehiculo, lo que esto disminuye tiempo y dinero.
?Que es el Desajuste?
El falta de equilibrio simboliza una circunstancia en la que la volumen de un elemento giratorio (en este particular supuesto, el rotativo de equilibrado con compartimento de embrague) se distribuye de modo desigual en comparacion a su eje de rotacion de revolucion. Esta situacion causa cargas centrifugas que a su vez causan sacudidas.
Causas fundamentales del Desbalance del Volador de Balanceo con Canasto de Embrague:
Irregularidades de manufactura y union: Hasta minimas diferencias en la estructura de los componentes pueden generar falta de equilibrio.
Uso y problemas: El operacion prolongada, el sobrecalentamiento y los fallas mecanicas podrian alterar la peso y resultar en desequilibrio.
Montaje o servicio deficiente: Una ensamblaje inapropiada de la cesta de embrague o servicios deficientes tambien tienden a generar falta de equilibrio.
DrstoEquak
September 10, 2024 @ 7:32 am
percocet pharmacy cost: rx express pharmacy stockton ca – dutasteride online pharmacy
DrstoEquak
September 10, 2024 @ 1:30 pm
Promethazine: stokes pharmacy – medi rx pharmacy
EasydrFlack
September 10, 2024 @ 3:25 pm
Zebeta: wich store or pharmacy sales hgh – celebrex pharmacy prices
Ремонт фотоаппаратов
September 10, 2024 @ 3:55 pm
Профессиональный сервисный центр по ремонту фото техники от зеркальных до цифровых фотоаппаратов.
Мы предлагаем: ремонт зеркальных фотоаппаратов
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Sonia
September 10, 2024 @ 11:00 pm
inter77
I loved as much as you will receive carried out right here.
The sketch is attractive, your authored material stylish.
nonetheless, you command get got an impatience over that you wish be delivering
the following. unwell unquestionably come more formerly again as exactly the same nearly a lot often inside
case you shield this increase.
DrstoEquak
September 10, 2024 @ 11:32 pm
ed pills online: concerta pharmacy coupons – lipitor pharmacy card
RobertVof
September 11, 2024 @ 1:49 am
Aquel volante de equilibrado con canasto de embrague simboliza un sistema fundamental con el fin de certificar el rendimiento excelente del propulsor y la transferencia de un automotor. El desequilibrio en esta unidad tiende a provocar temblores, sonido, deterioro rapido de los componentes e incluso fallos. Tradicionalmente, el estabilizacion se efectuaba luego de sacar el volante del motor, sin embargo las avances tecnologicos actuales posibilitan llevar a cabo dicho sistema de manera directa en el vehiculo, lo que esto ahorra duracion y costos.
Como se define el Desbalance?
El desequilibrio simboliza una circunstancia en la que la volumen de un componente rotacional (en el actual caso, el girador de nivelacion con compartimento de embrague) se ubica de forma irregular respecto a su punto de rotacion de movimiento circular. Lo cual genera esfuerzos centrifugos que en consecuencia generan vibraciones.
Principales del Desequilibrio del Volador de Estabilizacion con Recipiente de Embrague:
Inexactitudes de construccion y ensamblaje: Incluso insignificantes diferencias en la diseno de los componentes podrian originar desequilibrio.
Deterioro y desperfectos: El funcionamiento prolongado, el temperatura elevada y los danos mecanicos llegan a modificar la peso y provocar en falta de equilibrio.
Ensamblaje o reparacion inapropiada: Una montaje inapropiada de la compartimento de embrague o arreglos inadecuados de igual manera tienden a generar desajuste.
Ремонт фотоаппаратов в Москве
September 11, 2024 @ 3:04 am
Профессиональный сервисный центр по ремонту фото техники от зеркальных до цифровых фотоаппаратов.
Мы предлагаем: срочный ремонт фотоаппаратов
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
DrstoEquak
September 11, 2024 @ 5:08 am
target store pharmacy hours: northwest pharmacy – publix pharmacy online ordering
Affiliate Marketing Einnahmen
September 11, 2024 @ 6:50 am
Hey there! Just popping in to give a shoutout to your awesome blog. Your insights on affiliate marketing are genuinely tremendous. Making money from home has never been more achievable with affiliate marketing. It’s a wonderful way to generate passive income by promoting goods you stand behind. Your blog is a treasure trove of insights for emerging online entrepreneurs. Keep doing what you’re doing!
RobertAmipt
September 11, 2024 @ 11:47 am
propecia uk pharmacy: cialis pharmacy pricing – mexican pharmacy weight loss
ремонт бытовой техники краснодар
September 11, 2024 @ 1:45 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – сервисный центр в краснодаре
Josephglype
September 11, 2024 @ 4:06 pm
online pharmacy acyclovir boots pharmacy kamagra AebgEmoli
Josephglype
September 11, 2024 @ 4:11 pm
cheapest online pharmacy india celexa online pharmacy what does viagra cost at a pharmacy
MauriceJap
September 11, 2024 @ 4:27 pm
reputable indian online pharmacy: best online pharmacy india – Online medicine order
MauriceJap
September 11, 2024 @ 4:33 pm
value rx pharmacy irvine: pharmacy online cheap – Co-Amoxiclav
RobertAmipt
September 11, 2024 @ 5:20 pm
india pharmacy ambien: united pharmacy naltrexone – medco pharmacy cialis
Ремонт планшетов в Москве
September 11, 2024 @ 5:54 pm
Профессиональный сервисный центр по ремонту планшетов в Москве.
Мы предлагаем: ремонт планшета
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Arthurpax
September 11, 2024 @ 6:59 pm
http://indianpharmacy.company/# buy prescription drugs from india
ремонт бытовой техники в москве
September 11, 2024 @ 7:28 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: сервисные центры в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
888starzPi
September 11, 2024 @ 8:05 pm
Скачайте приложение для мобильных ставок 888Starz и начните выигрывать уже сегодня https://forum.wialon.com/profile.php?id=270523
RobertAmipt
September 11, 2024 @ 9:37 pm
top 10 online pharmacy in india: online shopping pharmacy india – online shopping pharmacy india
ремонт бытовой техники в краснодаре
September 11, 2024 @ 11:44 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – ремонт бытовой техники
RobertWab
September 12, 2024 @ 12:06 am
娛樂城排行
娛樂城推薦與優惠詳解
在現今的娛樂世界中,線上娛樂城已成為眾多玩家的首選。無論是喜歡真人遊戲、老虎機還是體育賽事,每個玩家都能在娛樂城中找到自己的樂趣。以下是一些熱門的娛樂城及其優惠活動,幫助您在選擇娛樂平台時做出明智的決定。
各大熱門娛樂城介紹
1. 富遊娛樂城
富遊娛樂城以其豐富的遊戲選擇和慷慨的優惠活動吸引了大量玩家。新會員只需註冊即可免費獲得體驗金 $168,無需儲值即可輕鬆試玩。此外,富遊娛樂城還提供首存禮金 100% 獎勵,最高可領取 $1000。
2. AT99娛樂城
AT99娛樂城以高品質的遊戲體驗和優秀的客戶服務聞名。該平台提供各種老虎機和真人遊戲,並定期推出新遊戲,讓玩家保持新鮮感。
3. BCR娛樂城
BCR娛樂城是一個新興的平台,專注於提供豐富的體育賽事投注選項。無論是足球、籃球還是其他體育賽事,BCR都能為玩家提供即時的投注體驗。
熱門遊戲推薦
WM真人視訊百家樂
WM真人視訊百家樂是許多玩家的首選,該遊戲提供了真實的賭場體驗,並且玩法簡單,容易上手。
戰神賽特老虎機
戰神賽特老虎機以其獨特的主題和豐富的獎勵機制,成為老虎機愛好者的最愛。該遊戲結合了古代戰神的故事背景,讓玩家在遊戲過程中感受到無窮的樂趣。
最新優惠活動
富遊娛樂城註冊送體驗金
富遊娛樂城新會員獨享 $168 體驗金,無需儲值即可享受全場遊戲,讓您無壓力地體驗不同遊戲的魅力。
VIP 日日返水無上限
富遊娛樂城為 VIP 會員提供無上限的返水優惠,最高可達 0.7%。此活動讓玩家在遊戲的同時,還能享受額外的回饋。
結論
選擇合適的娛樂城不僅能為您的遊戲體驗增色不少,還能通過各種優惠活動獲得更多的利益。無論是新會員還是資深玩家,都能在這些推薦的娛樂城中找到適合自己的遊戲和活動。立即註冊並體驗這些優質娛樂平台,享受無限的遊戲樂趣!
Arthurpax
September 12, 2024 @ 12:35 am
https://indianpharmacy.company/# online shopping pharmacy india
сервис центры в новосибирске
September 12, 2024 @ 1:25 am
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем:ремонт бытовой техники в новосибирске
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
RobertAmipt
September 12, 2024 @ 2:53 am
online order medicine: precision rx pharmacy – sam’s club pharmacy
Ремонт планшетов
September 12, 2024 @ 3:50 am
Профессиональный сервисный центр по ремонту планшетов в Москве.
Мы предлагаем: замена стекла на планшете
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
MauriceJap
September 12, 2024 @ 5:01 am
boots pharmacy kamagra: online pharmacy jobs – online pharmacy lortab
MauriceJap
September 12, 2024 @ 5:08 am
misoprostol malaysia pharmacy: medicine online shopping – xl pharmacy sildenafil
Affiliate Marketing Quotes Video
September 12, 2024 @ 6:47 am
Hello there! Just dropping in to praise your remarkable blog. Your knowledge on affiliate marketing are genuinely inspiring. Making money from home has never been more accessible thanks to affiliate marketing. It’s all about leveraging your internet presence and promoting goods or services that resonate with your audience. Your blog is a treasured resource for anyone exploring affiliate marketing. Keep on the fantastic work!
RobertAmipt
September 12, 2024 @ 7:19 am
mexico drug stores pharmacies: mexican online pharmacies prescription drugs – best online pharmacies in mexico
Arthurpax
September 12, 2024 @ 10:34 am
https://mexicopharmacy.cheap/# buying prescription drugs in mexico
Josephglype
September 12, 2024 @ 10:53 am
indian pharmacy online indian pharmacy reputable indian pharmacies
Josephglype
September 12, 2024 @ 10:59 am
oxybutynin online pharmacy differin online pharmacy doxycycline mexican pharmacy
сервис профи казань
September 12, 2024 @ 12:25 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – профи услуги
ремонт техники в новосибирске
September 12, 2024 @ 12:30 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем:ремонт крупногабаритной техники в новосибирске
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
RobertAmipt
September 12, 2024 @ 1:36 pm
buying prescription drugs in mexico online: mexican border pharmacies shipping to usa – mexican rx online
RobertAmipt
September 12, 2024 @ 6:16 pm
Aebgkeymn: online pharmacy adderall – meloxicam pharmacy
MauriceJap
September 12, 2024 @ 7:29 pm
top 10 pharmacies in india: india pharmacy mail order – indian pharmacies safe
MauriceJap
September 12, 2024 @ 7:36 pm
pharmacy bachelor degree online: pharmacy vardenafil – pharmacy on line
Josephglype
September 12, 2024 @ 10:06 pm
п»їbest mexican online pharmacies mexican mail order pharmacies reputable mexican pharmacies online
Josephglype
September 12, 2024 @ 10:12 pm
asacol pharmacy card magellan rx pharmacy help desk domperidone uk pharmacy
сервис профи казань
September 12, 2024 @ 11:16 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – ремонт бытовой техники в казани
RobertAmipt
September 13, 2024 @ 12:49 am
mexican mail order pharmacies: buying prescription drugs in mexico – mexico drug stores pharmacies
Ремонт видеокамер в Москве
September 13, 2024 @ 1:34 am
Профессиональный сервисный центр по ремонту видео техники а именно видеокамер.
Мы предлагаем: ремонт видеокамер в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Arthurpax
September 13, 2024 @ 3:03 am
https://indianpharmacy.company/# online shopping pharmacy india
RobertAmipt
September 13, 2024 @ 5:40 am
princeton university store pharmacy: flomax pharmacy training – acyclovir pharmacy
Arthurpax
September 13, 2024 @ 8:45 am
http://mexicopharmacy.cheap/# mexican drugstore online
Josephglype
September 13, 2024 @ 9:12 am
mexico drug stores pharmacies mexico pharmacies prescription drugs mexico pharmacies prescription drugs
Josephglype
September 13, 2024 @ 9:18 am
Online medicine order buy medicines online in india buy medicines online in india
MauriceJap
September 13, 2024 @ 9:24 am
mexican mail order pharmacies: mexican mail order pharmacies – medication from mexico pharmacy
RobertAmipt
September 13, 2024 @ 11:38 am
best online pharmacy india: Online medicine order – online pharmacy india
ремонт бытовой техники в красноярске
September 13, 2024 @ 11:51 am
Если вы искали где отремонтировать сломаную технику, обратите внимание – ремонт техники в красноярске
Ремонт видеокамер в Москве
September 13, 2024 @ 12:58 pm
Профессиональный сервисный центр по ремонту видео техники а именно видеокамер.
Мы предлагаем: ремонт видеокамер на дому
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
https://Www.Waste-Ndc.pro/community/profile/tressa79906983/
September 13, 2024 @ 3:28 pm
Hi there, this weekend is good for me, because this time i am reading
this enormous inforjative paragraph here aat my house. https://Www.Waste-Ndc.pro/community/profile/tressa79906983/
RobertAmipt
September 13, 2024 @ 3:58 pm
apollo pharmacy store locator: toronto pharmacy online – riteaid pharmacy
Arthurpax
September 13, 2024 @ 6:23 pm
https://pharmbig24.com/# online pharmacy adipex-p
Josephglype
September 13, 2024 @ 8:01 pm
buy prescription drugs from india cheapest online pharmacy india best india pharmacy
Josephglype
September 13, 2024 @ 8:08 pm
india pharmacy mail order best india pharmacy best india pharmacy
ремонт техники в москве
September 13, 2024 @ 9:38 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: сервис центры бытовой техники москва
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
RobertAmipt
September 13, 2024 @ 10:01 pm
pharmacies in mexico that ship to usa: mexico pharmacies prescription drugs – pharmacies in mexico that ship to usa
MauriceJap
September 13, 2024 @ 10:50 pm
buying prescription drugs in mexico online: buying prescription drugs in mexico online – medicine in mexico pharmacies
MauriceJap
September 13, 2024 @ 10:56 pm
reputable mexican pharmacies online: buying prescription drugs in mexico – reputable mexican pharmacies online
сервис профи красноярск
September 13, 2024 @ 11:12 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – ремонт техники в красноярске
сервис профи нижний новгород
September 13, 2024 @ 11:13 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – ремонт техники в нижнем новгороде
Arthurpax
September 14, 2024 @ 12:02 am
https://pharmbig24.com/# dillons pharmacy
RobertAmipt
September 14, 2024 @ 2:29 am
indian pharmacy: cheapest online pharmacy india – top online pharmacy india
ремонт бытовой техники в москве
September 14, 2024 @ 6:49 am
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: сервис центры бытовой техники москва
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Josephglype
September 14, 2024 @ 7:03 am
best india pharmacy pharmacy website india reputable indian online pharmacy
Josephglype
September 14, 2024 @ 7:08 am
buying prescription drugs in mexico best online pharmacies in mexico medicine in mexico pharmacies
RobertWab
September 14, 2024 @ 7:42 am
娛樂城
娛樂城推薦與優惠詳解
在現今的娛樂世界中,線上娛樂城已成為眾多玩家的首選。無論是喜歡真人遊戲、老虎機還是體育賽事,每個玩家都能在娛樂城中找到自己的樂趣。以下是一些熱門的娛樂城及其優惠活動,幫助您在選擇娛樂平台時做出明智的決定。
各大熱門娛樂城介紹
1. 富遊娛樂城
富遊娛樂城以其豐富的遊戲選擇和慷慨的優惠活動吸引了大量玩家。新會員只需註冊即可免費獲得體驗金 $168,無需儲值即可輕鬆試玩。此外,富遊娛樂城還提供首存禮金 100% 獎勵,最高可領取 $1000。
2. AT99娛樂城
AT99娛樂城以高品質的遊戲體驗和優秀的客戶服務聞名。該平台提供各種老虎機和真人遊戲,並定期推出新遊戲,讓玩家保持新鮮感。
3. BCR娛樂城
BCR娛樂城是一個新興的平台,專注於提供豐富的體育賽事投注選項。無論是足球、籃球還是其他體育賽事,BCR都能為玩家提供即時的投注體驗。
熱門遊戲推薦
WM真人視訊百家樂
WM真人視訊百家樂是許多玩家的首選,該遊戲提供了真實的賭場體驗,並且玩法簡單,容易上手。
戰神賽特老虎機
戰神賽特老虎機以其獨特的主題和豐富的獎勵機制,成為老虎機愛好者的最愛。該遊戲結合了古代戰神的故事背景,讓玩家在遊戲過程中感受到無窮的樂趣。
最新優惠活動
富遊娛樂城註冊送體驗金
富遊娛樂城新會員獨享 $168 體驗金,無需儲值即可享受全場遊戲,讓您無壓力地體驗不同遊戲的魅力。
VIP 日日返水無上限
富遊娛樂城為 VIP 會員提供無上限的返水優惠,最高可達 0.7%。此活動讓玩家在遊戲的同時,還能享受額外的回饋。
結論
選擇合適的娛樂城不僅能為您的遊戲體驗增色不少,還能通過各種優惠活動獲得更多的利益。無論是新會員還是資深玩家,都能在這些推薦的娛樂城中找到適合自己的遊戲和活動。立即註冊並體驗這些優質娛樂平台,享受無限的遊戲樂趣!
RobertAmipt
September 14, 2024 @ 8:25 am
buying prescription drugs in mexico: mexican pharmaceuticals online – best online pharmacies in mexico
Arthurpax
September 14, 2024 @ 9:47 am
http://indianpharmacy.company/# reputable indian online pharmacy
ремонт бытовой техники нижний новгород
September 14, 2024 @ 9:50 am
Если вы искали где отремонтировать сломаную технику, обратите внимание – выездной ремонт бытовой техники в нижнем новгороде
ремонт техники профи в новосибирске
September 14, 2024 @ 10:11 am
Если вы искали где отремонтировать сломаную технику, обратите внимание – техпрофи
RobertWab
September 14, 2024 @ 11:26 am
娛樂城推薦
娛樂城推薦與優惠詳解
在現今的娛樂世界中,線上娛樂城已成為眾多玩家的首選。無論是喜歡真人遊戲、老虎機還是體育賽事,每個玩家都能在娛樂城中找到自己的樂趣。以下是一些熱門的娛樂城及其優惠活動,幫助您在選擇娛樂平台時做出明智的決定。
各大熱門娛樂城介紹
1. 富遊娛樂城
富遊娛樂城以其豐富的遊戲選擇和慷慨的優惠活動吸引了大量玩家。新會員只需註冊即可免費獲得體驗金 $168,無需儲值即可輕鬆試玩。此外,富遊娛樂城還提供首存禮金 100% 獎勵,最高可領取 $1000。
2. AT99娛樂城
AT99娛樂城以高品質的遊戲體驗和優秀的客戶服務聞名。該平台提供各種老虎機和真人遊戲,並定期推出新遊戲,讓玩家保持新鮮感。
3. BCR娛樂城
BCR娛樂城是一個新興的平台,專注於提供豐富的體育賽事投注選項。無論是足球、籃球還是其他體育賽事,BCR都能為玩家提供即時的投注體驗。
熱門遊戲推薦
WM真人視訊百家樂
WM真人視訊百家樂是許多玩家的首選,該遊戲提供了真實的賭場體驗,並且玩法簡單,容易上手。
戰神賽特老虎機
戰神賽特老虎機以其獨特的主題和豐富的獎勵機制,成為老虎機愛好者的最愛。該遊戲結合了古代戰神的故事背景,讓玩家在遊戲過程中感受到無窮的樂趣。
最新優惠活動
富遊娛樂城註冊送體驗金
富遊娛樂城新會員獨享 $168 體驗金,無需儲值即可享受全場遊戲,讓您無壓力地體驗不同遊戲的魅力。
VIP 日日返水無上限
富遊娛樂城為 VIP 會員提供無上限的返水優惠,最高可達 0.7%。此活動讓玩家在遊戲的同時,還能享受額外的回饋。
結論
選擇合適的娛樂城不僅能為您的遊戲體驗增色不少,還能通過各種優惠活動獲得更多的利益。無論是新會員還是資深玩家,都能在這些推薦的娛樂城中找到適合自己的遊戲和活動。立即註冊並體驗這些優質娛樂平台,享受無限的遊戲樂趣!
RobertAmipt
September 14, 2024 @ 12:35 pm
п»їbest mexican online pharmacies: medication from mexico pharmacy – mexico drug stores pharmacies
MauriceJap
September 14, 2024 @ 12:50 pm
mail order pharmacy india: cheapest online pharmacy india – online shopping pharmacy india
MauriceJap
September 14, 2024 @ 12:57 pm
does pharmacy sell viagra: rx crossroads pharmacy phone number – india online pharmacy viagra
Arthurpax
September 14, 2024 @ 3:03 pm
https://pharmbig24.com/# online pharmacy venlafaxine
Josephglype
September 14, 2024 @ 5:38 pm
Online medicine home delivery world pharmacy india best india pharmacy
RobertAmipt
September 14, 2024 @ 6:22 pm
mexican mail order pharmacies: mexican border pharmacies shipping to usa – buying prescription drugs in mexico
ремонт бытовой техники новосибирск
September 14, 2024 @ 8:59 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – профи тех сервис новосибирск
RobertAmipt
September 14, 2024 @ 10:38 pm
mexico drug stores pharmacies: mexican rx online – mexican border pharmacies shipping to usa
Ремонт стиральных машин
September 14, 2024 @ 11:51 pm
Профессиональный сервисный центр по ремонту стиральных машин с выездом на дом по Москве.
Мы предлагаем: сервисные центры по ремонту стиральных машин
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
ремонт техники в казани
September 15, 2024 @ 12:01 am
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: сервисные центры по ремонту техники в казани
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Arthurpax
September 15, 2024 @ 12:56 am
https://mexicopharmacy.cheap/# best online pharmacies in mexico
MauriceJap
September 15, 2024 @ 3:08 am
online pharmacy fioricet: vancouver pharmacy viagra – best online pharmacy tadalafil
MauriceJap
September 15, 2024 @ 3:15 am
mexican online pharmacies prescription drugs: mexican border pharmacies shipping to usa – mexico drug stores pharmacies
Josephglype
September 15, 2024 @ 4:16 am
indian pharmacy buy medicines online in india п»їlegitimate online pharmacies india
Josephglype
September 15, 2024 @ 4:22 am
buying prescription drugs in mexico online medicine in mexico pharmacies mexico drug stores pharmacies
RobertAmipt
September 15, 2024 @ 4:27 am
medication from mexico pharmacy: mexican mail order pharmacies – mexico pharmacies prescription drugs
Arthurpax
September 15, 2024 @ 6:32 am
http://mexicopharmacy.cheap/# mexico drug stores pharmacies
woles togel
September 15, 2024 @ 7:16 am
croxy rocks
Wow that was unusual. I just wrote an very long comment but after I clicked submit my comment didn’t appear.
Grrrr… well I’m not writing all that over again. Anyhow, just wanted
to say wonderful blog!
RobertAmipt
September 15, 2024 @ 8:43 am
best india pharmacy: pharmacy website india – india pharmacy
Ремонт стиральных машин в Москве
September 15, 2024 @ 10:56 am
Профессиональный сервисный центр по ремонту стиральных машин с выездом на дом по Москве.
Мы предлагаем: ремонт стиральных машин в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
ремонт техники в казани
September 15, 2024 @ 11:09 am
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: сервисные центры в казани
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
RobertAmipt
September 15, 2024 @ 2:36 pm
indian pharmacy paypal: indian pharmacies safe – indian pharmacies safe
Josephglype
September 15, 2024 @ 2:41 pm
best cialis online pharmacy drug costs ciprofloxacin online pharmacy
Josephglype
September 15, 2024 @ 2:46 pm
mexico drug stores pharmacies mexican online pharmacies prescription drugs buying prescription drugs in mexico
Arthurpax
September 15, 2024 @ 4:23 pm
http://mexicopharmacy.cheap/# reputable mexican pharmacies online
MauriceJap
September 15, 2024 @ 5:24 pm
viagra pharmacy us: cytotec in malaysia pharmacy – buying viagra from pharmacy
MauriceJap
September 15, 2024 @ 5:31 pm
mexican pharmaceuticals online: reputable mexican pharmacies online – buying from online mexican pharmacy
сервис профи пермь
September 15, 2024 @ 6:50 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – тех профи
RobertAmipt
September 15, 2024 @ 6:54 pm
venlafaxine xr online pharmacy: buy viagra tesco pharmacy – alliance rx pharmacy
Arthurpax
September 15, 2024 @ 10:03 pm
https://pharmbig24.online/# ketoconazole pharmacy
RobertAmipt
September 16, 2024 @ 12:58 am
united pharmacy tamoxifen: international pharmacies that ship to the usa – rx pharmacy meaning
Josephglype
September 16, 2024 @ 1:23 am
cheapest pharmacy viagra amoxicillin online pharmacy no prescription erythromycin online pharmacy
RobertAmipt
September 16, 2024 @ 5:18 am
mexican mail order pharmacies: mexican rx online – buying from online mexican pharmacy
ремонт техники профи в перми
September 16, 2024 @ 6:01 am
Если вы искали где отремонтировать сломаную технику, обратите внимание – профи услуги
ремонт бытовой техники в москве
September 16, 2024 @ 7:26 am
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: ремонт крупногабаритной техники в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
MauriceJap
September 16, 2024 @ 7:37 am
mexican mail order pharmacies: buying prescription drugs in mexico online – mexican border pharmacies shipping to usa
MauriceJap
September 16, 2024 @ 7:44 am
Levitra Oral Jelly: buy cialis cheap us pharmacy – synthroid mexico pharmacy
Arthurpax
September 16, 2024 @ 7:45 am
http://mexicopharmacy.cheap/# mexican online pharmacies prescription drugs
Michaeldus
September 16, 2024 @ 9:59 am
Пеларгонии сортовые
Пеларгонии видовые: идеальный вариант для вашего дома и огорода
Если ты подбираете растения, что будут восхищать тебя их красотой и благоуханием, одновременно с тем не требуя сложного обслуживания, элитные герани — лучший выбор. Этих цветы наделены уникальными характеристиками, которые превращают их ведущими в категории декоративных цветов.
Почему сортовые пеларгонии?
Легкость и легкость в уходе
Пеларгонии не требуют особых факторов для развития и быстро приспосабливаются к многим условиям. Они прекрасно чувствуют себя как в жилище, так и на свежем воздухе. Не беспокойтесь о прихотливых цветах — пеларгонии требуется лишь поливать по степени исчезновения влаги грунта и получать удовольствие от их процветанием.
Яркие и разнообразные тона
Определенный сорт пеларгоний содержит свои особенные оттенки и формы. Разновидности, такие как, ТА Монако, впечатляют интенсивными оттенками и выразительными соцветиями. Это цветы, что мгновенно захватывают интерес и вносят живые подчеркивания в любом месте.
Приятный благоухание, создающий атмосферу
Пеларгонии не просто облагораживают дом — они предоставляют его приятным, незаметным благоуханием. Этот естественный аромат помогает придать чувство уюта и покоя, а к тому же работает как природный отпугиватель для мошек.
Долгое цветение
Разновидные пеларгонии продолжают радовать вид своим цветением в течение нескольких месяцев. Люди будут восхищаться их красотой с начала лета и до холодов периода. Такое непрекращающееся цветение — редкое достоинство среди декоративных видов.
Идеальный выбор для любого пространства
Герани универсальны — их желательно выращивать как в контейнерах на подоконниках, так и в на участке. Небольшие кусты, например ЮВ Кардинал, хорошо подходят в украшенных кашпо, а виды, как Магнус, будут декорацией клумбы.
Для чего необходимо отдать предпочтение именно пеларгонии?
Данные цветы — не просто декоративный элемент оформления. Они значительно доминируют среди других цветов из-за своей легкости в уходе, декоративности и продолжительному процветанию. Их живописные тона придают особенную среду, будь то в помещении или на участке. Герани — это лучший сочетание эстетики и функциональности.
Останавливайтесь на пеларгонии — формируйте в своем окружении эстетику без лишних забот!
RobertAmipt
September 16, 2024 @ 11:21 am
best india pharmacy: indian pharmacy – top 10 online pharmacy in india
Josephglype
September 16, 2024 @ 11:35 am
mail order pharmacy india Online medicine order cheapest online pharmacy india
Josephglype
September 16, 2024 @ 11:40 am
adderall online pharmacy online pharmacy ed effexor pharmacy assistance
Arthurpax
September 16, 2024 @ 1:15 pm
https://pharmbig24.com/# domperidone from inhouse pharmacy
ремонт бытовой техники в ростове на дону
September 16, 2024 @ 2:03 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – техпрофи
ремонт техники в москве
September 16, 2024 @ 4:34 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: ремонт бытовой техники в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
MauriceJap
September 16, 2024 @ 9:41 pm
pain meds online pharmacy: simvastatin uk pharmacy – nexium uk pharmacy
MauriceJap
September 16, 2024 @ 9:48 pm
pharmacy rx one coupon codes: viagra muscat pharmacy – dexamethasone pharmacy
RobertAmipt
September 16, 2024 @ 10:32 pm
mexican mail order pharmacies: mexican pharmaceuticals online – mexican border pharmacies shipping to usa
Josephglype
September 16, 2024 @ 10:47 pm
mexico pharmacies prescription drugs mexican drugstore online purple pharmacy mexico price list
Josephglype
September 16, 2024 @ 10:53 pm
mexico drug stores pharmacies mexico drug stores pharmacies mexico drug stores pharmacies
Ремонт игровых консолей
September 16, 2024 @ 11:25 pm
Профессиональный сервисный центр по ремонту игровых консолей Sony Playstation, Xbox, PSP Vita с выездом на дом по Москве.
Мы предлагаем: ремонт консолей
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
ремонт техники профи в ростове на дону
September 17, 2024 @ 12:53 am
Если вы искали где отремонтировать сломаную технику, обратите внимание – техпрофи
Ремонт видеокарт
September 17, 2024 @ 1:30 am
Профессиональный сервисный центр по ремонту компьютерных видеокарт по Москве.
Мы предлагаем: диагностика видеокарты
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
RobertAmipt
September 17, 2024 @ 3:06 am
cialis online american pharmacy: drug store near me – austria pharmacy online
Ремонт игровых приставок в Москве
September 17, 2024 @ 10:19 am
Профессиональный сервисный центр по ремонту игровых консолей Sony Playstation, Xbox, PSP Vita с выездом на дом по Москве.
Мы предлагаем: надежный сервис ремонта игровых консолей
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Ремонт фотовспышек
September 17, 2024 @ 5:36 pm
Профессиональный сервисный центр по ремонту фототехники в Москве.
Мы предлагаем: купить в москве фотовспышку дешево
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Подробнее на сайте сервисного центра remont-vspyshek-realm.ru
Ремонт персональных компьютеров
September 17, 2024 @ 7:35 pm
Профессиональный сервисный центр по ремонту компьютероной техники в Москве.
Мы предлагаем: ремонт компьютеров в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
rupiahtoto
September 17, 2024 @ 9:25 pm
cuan128
Fantastic beat ! I would like to apprentice whilst you amend your website, how
could i subscribe for a weblog site? The account
aided me a applicable deal. I have been a little bit familiar of this your broadcast offered vivid clear idea
Dominic David
September 17, 2024 @ 9:29 pm
I am constantly impressed by the depth and detail in your posts You have a gift for making complex topics easily understandable
Ремонт проекторов в Москве
September 17, 2024 @ 10:24 pm
Профессиональный сервисный центр по ремонту фото техники от зеркальных до цифровых фотоаппаратов.
Мы предлагаем: отремонтировать проекционный экран
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Ремонт видеокарт в Москве
September 18, 2024 @ 12:51 am
Профессиональный сервисный центр по ремонту компьютерных видеокарт по Москве.
Мы предлагаем: ремонт видеокарты
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
pragmatic demo
September 18, 2024 @ 1:35 am
laskartogel
Hello very nice site!! Guy .. Beautiful .. Wonderful ..
I will bookmark your site and take the feeds also? I am glad
to search out so many helpful information here in the publish, we
want develop more strategies on this regard, thank you for sharing.
. . . . .
JamesCoels
September 18, 2024 @ 4:44 am
gates of olympus demo turkce Gates of Olympus Gates of Olympus
JamesCoels
September 18, 2024 @ 4:49 am
gate of olympus oyna gates of olympus demo turkce gates of olympus oyna demo
kdg789
September 18, 2024 @ 7:31 am
emas168
Hello to every one, the contents existing at this web page are really amazing
for people knowledge, well, keep up the nice work fellows.
rajabandot
September 18, 2024 @ 10:00 am
pp ml hd
naturally like your website but you need to check the spelling on quite a few of your posts.
A number of them are rife with spelling issues and I to
find it very bothersome to tell the reality on the other hand I’ll certainly come back again.
JamesCoels
September 18, 2024 @ 2:25 pm
betine guncel giris betine promosyon kodu betine sikayet
JamesCoels
September 18, 2024 @ 2:31 pm
casibom casibom giris casibom
kepritogel
September 18, 2024 @ 2:48 pm
latoto latoto latoto latoto
We’re a group of volunteers and starting a new scheme in our
community. Your website provided us with valuable info to work
on. You have done a formidable job and our whole community will be thankful to you.
Georgetax
September 18, 2024 @ 3:51 pm
http://starzbet.shop/# starzbet giris
Georgetax
September 18, 2024 @ 9:27 pm
https://gatesofolympusoyna.online/# gates of olympus demo
akun demo
September 18, 2024 @ 10:52 pm
mawartoto login mawartoto login mawartoto login mawartoto login
Magnificent items from you, man. I have bear in mind your stuff previous to and you’re just extremely great.
I really like what you have obtained right here, really like what you’re saying and the
way in which through which you assert it. You are making it enjoyable and you
still take care of to keep it sensible. I can not
wait to read far more from you. That is actually a terrific website.
Магазин раковин
September 18, 2024 @ 11:03 pm
Хочу поделиться опытом покупки в одном интернет-магазине сантехники. Решил обновить ванную комнату и искал место, где можно найти широкий выбор раковин и ванн. Этот магазин приятно удивил своим ассортиментом и сервисом. Там есть всё: от классических чугунных ванн до современных акриловых моделей.
Если вам нужна умывальник в ванную , то это точно туда. Цены конкурентные, а качество товаров подтверждено сертификатами. Консультанты помогли с выбором, ответили на все вопросы. Доставка пришла вовремя, и установка прошла без проблем. Остался очень доволен покупкой и сервисом.
Ремонт блоков питания
September 19, 2024 @ 12:24 am
Профессиональный сервисный центр по ремонту компьютерных блоков питания в Москве.
Мы предлагаем: ремонт блока питания цена
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
JamesCoels
September 19, 2024 @ 1:13 am
gates of olympus demo Gates of Olympus gates of olympus turkce
JamesCoels
September 19, 2024 @ 1:19 am
betine guncel giris betine guncel giris betine sikayet
сервис по ремонту телефонов
September 19, 2024 @ 2:28 am
Сломался телефон, думал покупать новый, но решил попробовать отремонтировать. Обратился в этот сервисный центр и не пожалел. Профессионалы своего дела быстро восстановили мой телефон. Рекомендую посетить их сайт: разбил экран починить телефон.
ремонт техники профи в самаре
September 19, 2024 @ 3:17 am
ремонт бытовой техники самара
ремонт кондиционеров
September 19, 2024 @ 3:25 am
<a href=”https://remont-kondicionerov-wik.ru”>кондиционер ремонт</a>
hantuslot
September 19, 2024 @ 5:47 am
I’ve been surfing online more than 4 hours today, yet I never found any interesting article like
yours. It’s pretty worth enough for me. In my view,
if all website owners and bloggers made good content as you did, the internet will be a lot more useful than ever before.
Georgetax
September 19, 2024 @ 11:53 pm
http://starzbet.shop/# starz bet giris
tokekwin
September 20, 2024 @ 1:45 am
kapten69
Amazing! Its genuinely remarkable paragraph, I have got much clear idea on the topic of from this paragraph.
Ремонт персональных компьютеров в Москве
September 20, 2024 @ 3:49 am
Профессиональный сервисный центр по ремонту компьютероной техники в Москве.
Мы предлагаем: качественный ремонт компьютеров
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
JasonHop
September 20, 2024 @ 5:18 am
kometa casino рабочее зеркало
Казино Kometa: Превосходный Выбор для Цифровых Развлечений
В сфере виртуальных казино Kometa Casino получило известность благодаря разнообразию слотов, привлекательным бонусам и первоклассному обслуживанию. Эта сайт привлекает внимание пользователей во всех странах своими исключительными акциями и регулярными акциями. В этой статье мы проанализируем, почему Kometa является выдающейся площадок для азартных игр.
Преимущества Kometa
Одним из ключевых факторов, делающих особенным Kometa Casino, является внимание на потребности клиентов. Система предлагает свыше тысячи слотов, в которых все сможет выбрать игру. Это могут быть как классические автоматы, и еще инновационные игры с новаторскими возможностями. Приятным дополнением является то, что Казино Kometa обеспечивает круглосуточную сопровождение клиентов, гарантируя комфортное и защищенное игровое пространство.
Ключевые особенности Kometa:
Год начала работы: 2024
Сертификация: Curacao
Количество игр: Более 1000
Поддержка: Круглосуточная онлайн-чат и почта
Мобильная версия: Да
Методы оплаты: Skrill
Защита: Шифрование SSL
Начальные бонусы
Ключевой особенностью Казино Kometa считаются привлекательные стартовые предложения для начинающих пользователей. После входа на сайт пользователи получают доступ к эксклюзивным бонусам, чем могут начать игру с минимальными рисками. Эти промо создают благоприятные условия для новичков, создавая условия улучшить свои возможности выиграть с самого старта.
Широкий ассортимент игр
Kometa Casino гарантирует огромное разнообразие игр на все предпочтения. Клиенты могут испытать удовольствие классическими слотами, настольными играми, а также играми с живыми дилерами. Благодаря передовым технологиям визуальных эффектов и звуковому сопровождению, каждый игрок может глубоко войти в развлечения.
Постоянные события и турниры
Для каждого клиента платформа постоянно организует акции и соревнования с ценными призами. Акции запускаются каждый месяц, что делает игровой опыт более захватывающим и захватывающим. Это позволяет игрокам не только наслаждаться игрой, но и получать дополнительные бонусы и награды.
Почему стоит выбрать
Казино Kometa — это превосходное соединение широкого ассортимента, отличной поддержки и защищенной платформы. Система отличается своим вниманием к пользователям и желанием модернизировать развлечения. Без учета опыта, все откроет в Казино Kometa нечто, что позволит его пребывание на сайте захватывающим и приятным.
Вступайте в Kometa и играйте с удовольствием захватывающими ощущениями и увлекательными слотами всегда!
Georgetax
September 20, 2024 @ 5:54 am
http://starzbet.shop/# starzbet guncel giris
Ремонт видео камер наблюдения
September 20, 2024 @ 5:38 pm
Профессиональный сервисный центр по ремонту камер видео наблюдения по Москве.
Мы предлагаем: ремонт камер видеонаблюдения
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
сервис центры в нижнем новгороде
September 20, 2024 @ 7:47 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: ремонт бытовой техники в нижнем новгороде
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Normansox
September 20, 2024 @ 10:47 pm
se puede comprar viagra sin receta: comprar viagra contrareembolso 48 horas – se puede comprar viagra sin receta
Normansox
September 20, 2024 @ 11:33 pm
farmacia online envГo gratis: comprar tadalafilo – farmacia online envГo gratis
Ronalditady
September 21, 2024 @ 12:19 am
farmacia online barcelona comprar cialis online sin receta farmacia online 24 horas
Jamestew
September 21, 2024 @ 5:07 am
sildenafilo sandoz 100 mg precio: Viagra sildenafilo – sildenafilo 100mg farmacia
DennisRurne
September 21, 2024 @ 5:07 am
https://tadalafilo.bid/# farmacia online madrid
Jamestew
September 21, 2024 @ 5:12 am
farmacias direct: farmacia envio gratis – farmacia online espaГ±a envГo internacional
DennisRurne
September 21, 2024 @ 5:12 am
https://tadalafilo.bid/# farmacia online 24 horas
Ronalditady
September 21, 2024 @ 6:37 am
sildenafilo cinfa 100 mg precio farmacia viagra generico comprar viagra online en andorra
FloydBer
September 21, 2024 @ 7:58 am
http://farmaciaeu.com/# farmacias online seguras en espaГ±a
farmacia online 24 horas
Normansox
September 21, 2024 @ 10:24 am
farmacia online madrid: farmacias online seguras – farmacias online baratas
Ronalditady
September 21, 2024 @ 10:52 am
farmacia online envГo gratis comprar tadalafilo farmacia online madrid
Normansox
September 21, 2024 @ 11:24 am
farmacia online envГo gratis: comprar cialis online seguro – farmacia online madrid
FloydBer
September 21, 2024 @ 1:29 pm
http://tadalafilo.bid/# farmacias online seguras
farmacia online madrid
DennisRurne
September 21, 2024 @ 3:19 pm
http://farmaciaeu.com/# farmacias online seguras en espaГ±a
DennisRurne
September 21, 2024 @ 3:25 pm
http://farmaciaeu.com/# farmacia online barata y fiable
Jamestew
September 21, 2024 @ 3:30 pm
farmacias direct: Precio Cialis 20 Mg – farmacia online barata
Ремонт часов
September 21, 2024 @ 6:55 pm
Профессиональный сервисный центр по ремонту кнаручных часов от советских до швейцарских в Москве.
Мы предлагаем: срочный ремонт часов
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Ronalditady
September 21, 2024 @ 7:18 pm
farmacia online envГo gratis tadalafilo farmacia barata
ремонт бытовой техники тюмень
September 21, 2024 @ 8:02 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – техпрофи
ремонт бытовой техники в перми
September 21, 2024 @ 8:57 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: сервис центры бытовой техники пермь
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
JasonHop
September 21, 2024 @ 9:44 pm
Онлайн-казино Kometa: Оптимальный Выбор для Онлайн Развлечений
В мире виртуальных казино Казино Kometa получило известность благодаря множеству игр, щедрым поощрениям и первоклассному поддержке. Эта платформа захватывает интерес игроков по всему миру своими уникальными возможностями и регулярными промо. В представленной описании мы проанализируем, почему Казино Kometa считается высоко оцениваемой игровых площадок.
Преимущества Kometa
Основным аспектом, выделяющих Kometa Casino, является фокус на потребности клиентов. Сайт предоставляет огромное количество развлечений, где все откроет любимое развлечение. Это могут быть как классические слоты, и еще новые игры с инновационными функциями. Бонусом является то, что Kometa Casino обеспечивает постоянную поддержку клиентов, обеспечивая комфортное и защищенное окружение.
Главные параметры Kometa:
Год основания: 2024
Лицензия: Curacao
Количество игр: Огромное количество
Поддержка: 24/7 чат и почта
Поддержка мобильных устройств: Доступно
Способы оплаты: Mastercard
Надежность: SSL-шифрование
Приветственные бонусы
Ключевой особенностью Kometa Casino являются привлекательные акции для новичков для новичков. Сразу после регистрации новички получают доступ к особым акциям, что позволяет стартовать с меньшими затратами. Эти промо предоставляют выгодные шансы для новичков, предоставляя шанс увеличить свои успех с самого старта.
Широкий ассортимент игр
Kometa гарантирует широкий выбор слотов на любой вкус. Пользователи могут испытать удовольствие привычными автоматами, настольными играми, а также играми с живыми дилерами. Благодаря передовым технологиям визуальных эффектов и аудио, любой пользователь может глубоко войти в процесс игры.
Частые промо и активности
Для игроков платформа регулярно предлагает турниры и турниры с выгодными наградами. Турниры запускаются каждый месяц, создавая игровой опыт интересным и насыщенным. Это дает возможность пользователям не только играть игрой, но и получать призы и выигрыши.
Почему Kometa Casino?
Kometa — это идеальное сочетание разнообразных игр, качественного обслуживания и надежной системы. Сайт выделяется своим заботой о клиентах и постоянным стремлением модернизировать развлечения. Без учета опыта, любой найдет в Kometa Casino то, что сделает его путешествие в мире игр захватывающим и удобным.
Присоединяйтесь к Kometa и играйте с удовольствием яркими эмоциями и увлекательными слотами каждый день!
pos4d
September 21, 2024 @ 10:11 pm
shio ular togel 2023
Excellent web site. Plenty of useful information here.
I am sending it to a few buddies ans additionally sharing in delicious.
And certainly, thank you to your effort!
Normansox
September 22, 2024 @ 1:13 am
viagra online rГЎpida: Viagra sildenafilo – se puede comprar sildenafil sin receta
DennisRurne
September 22, 2024 @ 2:11 am
http://sildenafilo.men/# viagra 100 mg precio en farmacias
DennisRurne
September 22, 2024 @ 2:16 am
https://sildenafilo.men/# sildenafilo cinfa sin receta
Jamestew
September 22, 2024 @ 2:17 am
sildenafilo 50 mg comprar online: sildenafilo – viagra para hombre precio farmacias
akun demo slot
September 22, 2024 @ 2:54 am
alexistogel alexistogel alexistogel alexistogel
Howdy! Someone in my Myspace group shared this site with us
so I came to check it out. I’m definitely loving the information. I’m bookmarking and
will be tweeting this to my followers! Outstanding blog and terrific design and style.
DennisRurne
September 22, 2024 @ 11:41 am
http://farmaciaeu.com/# farmacias online baratas
DennisRurne
September 22, 2024 @ 11:45 am
https://tadalafilo.bid/# п»їfarmacia online espaГ±a
Jamestew
September 22, 2024 @ 12:07 pm
farmacia online envГo gratis: gran farmacia online – farmacia online madrid
Jesseciz
September 22, 2024 @ 2:35 pm
балансировка дымососа
ООО «Кинематика»: Профессиональные продукты для производственной балансирования и анализов промышленных устройств
Организация «Кинематика» фокусируется на изготовлении и изготовлении высококачественных систем для сбалансированности и анализа техники. Изделия фирмы используется для техобслуживания и восстановления разнообразных устройств, включая системы вентиляции, мельницы, карданные валы, системы передачи и металлообрабатывающие станки.
Главные продукты фирмы:
1. Модуль Арбаланс — переносной прибор балансировки вибраций
Инструмент предназначен для балансировки вращательных систем оборудования в заводской сборке в собственных узлах. Arbalance предлагает точное выполнение уравновешивания с небольшими затратами. Ключевые особенности устройства:
Автоматизированная система расчета: работает автономно.
Графики вибрации и спектральный анализ: помогают выявлять неисправности.
Двойной канал измерения: осуществляет замеры вибрации одновременно в двух осях, сокращая время процесса.
Цена: 84 тыс. руб.
2. Balcom-1A — компактный вибродиагностический прибор
Модуль Балком-1А подходит для уравновешивания роторных систем в системных подшипниках. Главной особенностью является простота использования и автоматизированный процесс расчета. Система дополнена:
Два канала измерения вибрации.
Анализ спектра вибраций для диагностики состояния техники.
Цена: 73 тыс. руб.
3. Балансировочная система Балком-2 — измерительная система для балансировочных станков
Балком-2 внедряется в станциях балансировки для оценки вибраций. Он гарантирует надежность сбалансированности вращательных механизмов и других агрегатов в производстве.
Цена: от 90 тыс. руб. до 95000 руб. в зависимости от оборудования.
4. Система Балком-4 — для балансировки в нескольких плоскостях валов
Система Балком-4 применяется для балансировки составных карданных валов и внедряется в индустриальных установках. Этот прибор обеспечивает стабильные показатели и точные измерения при многоплоскостной сбалансированности.
Цена: от 100000 руб. до 126000 рублей.
Инновационные решения отслеживания и проверки
ООО «Кинематика» также разрабатывает решения для непрерывной диагностики техники. Эти системы оснащены индуктивные датчики для отслеживания вибраций и других показателей. Например, модуль Реконт помогает отслеживать редукторы по параметрам точности и скорости вращения.
FloydBer
September 22, 2024 @ 3:19 pm
https://tadalafilo.bid/# farmacias online seguras
farmacias online seguras en espaГ±a
Ремонт парогенераторов в Москве
September 22, 2024 @ 4:17 pm
Профессиональный сервисный центр по ремонту парогенераторов в Москве.
Мы предлагаем: парогенератор ремонт
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Normansox
September 22, 2024 @ 5:00 pm
farmacia online espaГ±a envГo internacional: Cialis generico – farmacia online espaГ±a envГo internacional
akun demo
September 22, 2024 @ 6:25 pm
olxtoto olxtoto olxtoto olxtoto
Hey there! I’ve been reading your web site for some time now and
finally got the courage to go ahead and give you a shout
out from Atascocita Tx! Just wanted to tell you keep up the great work!
FloydBer
September 22, 2024 @ 8:52 pm
http://sildenafilo.men/# sildenafilo cinfa 100 mg precio farmacia
п»їfarmacia online espaГ±a
DennisRurne
September 22, 2024 @ 9:01 pm
https://sildenafilo.men/# se puede comprar viagra sin receta
DennisRurne
September 22, 2024 @ 9:06 pm
http://farmaciaeu.com/# farmacia online barata
ремонт бытовой техники волгоград
September 22, 2024 @ 9:06 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – ремонт бытовой техники
protogel
September 22, 2024 @ 10:00 pm
wifitoto
Hey there! I’ve been following your site for a while now and finally got the courage to go ahead and give you a shout out from Austin Tx!
Just wanted to tell you keep up the good work!
bong da lu
September 22, 2024 @ 10:28 pm
Bongdalu cập nhật tin tức bóng đá nóng hổi, thể thao sôi động và giải trí hấp dẫn
rongbachkim
September 23, 2024 @ 3:24 am
Rồng Bạch Kim – Soi cầu lô chính xác miễn phí chính xác số #1 2024
EdisonMaype
September 23, 2024 @ 4:34 am
Farmacia online miglior prezzo: Cialis generico prezzo – comprare farmaci online all’estero
Edwardinfor
September 23, 2024 @ 5:46 am
top farmacia online [url=https://tadalafilit.com/#]Cialis generico recensioni[/url] farmacie online affidabili
CharlesRaF
September 23, 2024 @ 6:57 am
https://farmaciait.men/# Farmacie online sicure
comprare farmaci online con ricetta
Charlessop
September 23, 2024 @ 7:35 am
Farmacie on line spedizione gratuita [url=https://farmaciait.men/#]farmacia online migliore[/url] Farmacie online sicure
ремонт бытовой техники в москве
September 23, 2024 @ 9:02 am
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: ремонт крупногабаритной техники в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Louisgot
September 23, 2024 @ 9:59 am
Farmacie on line spedizione gratuita: Farmacie online sicure – migliori farmacie online 2024
EdisonMaype
September 23, 2024 @ 11:49 am
farmacia online piГ№ conveniente: Farmacie che vendono Cialis senza ricetta – farmacia online
Danielhen
September 23, 2024 @ 12:19 pm
вибродиагностика
Предприятие «Кинематика»: Качественные услуги для производственной балансировки и проверки промышленного оборудования
Компания ООО «Кинематика» работает на создании и изготовлении точных устройств для сбалансированности и анализа техники. Продукция компании применяется для ремонта и восстановления многих устройств, включая системы вентиляции, дробилки, валовые системы, системы передачи и станки для металла.
Главные решения предприятия:
1. Арбаланс устройство — компактный прибор балансировки вибраций
Система разработан для динамической балансировки техники в заводской сборке в родных подшипниках. Арбаланс даёт точное выполнение уравновешивания с уменьшенными затратами. Главные достоинства:
Автоматизированная система расчета: полностью автоматизирована.
Спектральная диагностика: помогают выявлять неполадки.
Измерение по двум плоскостям: осуществляет замеры вибрационные колебания в одно время в двух плоскостях, улучшая результаты.
Цена: 84000 руб.
2. Модуль Балком-1А — компактный виброметр-балансировщик
Балком-1А предназначен для балансировки вращательных механизмов в собственных подшипниках. Ключевым преимуществом является простота использования и автоматизированный процесс расчета. Также устройство оснащено:
Два вибрационных канала.
Спектральный анализ для технической диагностики технического состояния оборудования.
Цена: 73 тыс. руб.
3. Модуль Балком-2 — измерительная система для систем балансировки
Прибор Балком-2 используется в станциях балансировки для виброизмерений. Он предлагает точность сбалансированности вращательных механизмов и другого оборудования в промышленности.
Цена: от 90 тыс. руб. до 95000 рублей в зависимости от сборки.
4. Система Балком-4 — для балансировки многосоставных систем карданных валов
Система Балком-4 создан для уравновешивания валов и внедряется в технических комплексах. Этот модуль даёт надежные результаты и прецизионность при многоплоскостной сбалансированности.
Цена: от 100000 рублей до 126 тыс. руб..
Новейшие технологии диагностики и диагностики
Фирма «Кинематика» также создает устройства для непрерывного мониторинга техники. Эти устройства оснащены бесконтактные датчики для оценки колебаний и других показателей. Например, система Реконт позволяет отслеживать состояние редукторов по точности работы механизмов и оборотам.
Charlessop
September 23, 2024 @ 4:59 pm
acquisto farmaci con ricetta [url=http://farmaciait.men/#]acquisto farmaci con ricetta[/url] comprare farmaci online con ricetta
EdisonMaype
September 23, 2024 @ 6:22 pm
comprare farmaci online all’estero: Cialis generico recensioni – Farmacia online miglior prezzo
ремонт бытовой техники в красноярске
September 23, 2024 @ 7:41 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: ремонт бытовой техники в красноярске
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Louisgot
September 23, 2024 @ 8:48 pm
top farmacia online: Farmacie online sicure – farmacie online sicure
888starzPi
September 23, 2024 @ 10:10 pm
Не забудьте ввести 888 starz bonus code для получения фрибетов.
sluggers
September 24, 2024 @ 12:04 am
You’ve made some good points there. I checked on the net for more info about the issue and found
most individuals will go along with your views on this web site.
EdisonMaype
September 24, 2024 @ 1:38 am
Farmacie on line spedizione gratuita: Cialis generico prezzo – farmacie online sicure
motchill
September 24, 2024 @ 2:22 am
Motchilltv.fyi – Trang web xem phim Online chất lượng Full HD với giao diện thân thiện, trực quan cùng kho phim với hơn 15.000+ bộ phim mới và phim hot hiện nay.
Charlessop
September 24, 2024 @ 3:40 am
migliori farmacie online 2024 [url=https://farmaciait.men/#]Farmacia online piu conveniente[/url] farmaci senza ricetta elenco
Charlessop
September 24, 2024 @ 3:45 am
acquisto farmaci con ricetta [url=http://farmaciait.men/#]п»їFarmacia online migliore[/url] п»їFarmacia online migliore
bongdalu
September 24, 2024 @ 4:24 am
Bongdalu cập nhật tin tức bóng đá nóng hổi, thể thao sôi động và giải trí hấp dẫn.
ремонт бытовой техники в уфе
September 24, 2024 @ 4:45 am
Если вы искали где отремонтировать сломаную технику, обратите внимание – сервис центр в уфе
j200m
September 24, 2024 @ 4:49 am
rajabandot rajabandot rajabandot rajabandot
Thanks for your personal marvelous posting! I genuinely enjoyed reading it, you are a great
author.I will be sure to bookmark your blog and definitely will come back someday.
I want to encourage yourself to continue your great writing, have a
nice holiday weekend!
сервис центры в красноярске
September 24, 2024 @ 6:43 am
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: сервис центры бытовой техники красноярск
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Louisgot
September 24, 2024 @ 7:54 am
farmacia online piГ№ conveniente: Cialis generico 20 mg 8 compresse prezzo – farmaci senza ricetta elenco
Louisgot
September 24, 2024 @ 8:00 am
farmacie online autorizzate elenco: Cialis generico prezzo – Farmacia online miglior prezzo
PatrickMourO
September 24, 2024 @ 9:55 am
viagra originale in 24 ore contrassegno: viagra online siti sicuri – cerco viagra a buon prezzo
tlover tonet
September 24, 2024 @ 12:00 pm
Just wanna state that this is very useful, Thanks for taking your time to write this.
أنابيب الفولاذ المقاوم للصدأ
September 24, 2024 @ 2:18 pm
Ductile Iron Pipes in Iraq As one of the most reliable sources for ductile iron pipes in Iraq, ElitePipe Factory is dedicated to providing products of the highest quality. Our ductile iron pipes are engineered for superior strength and flexibility, making them suitable for demanding applications such as water distribution and sewage systems. With advanced manufacturing techniques and rigorous quality control, ElitePipe Factory ensures that each pipe meets the highest standards of performance and durability. Trust ElitePipe Factory for your ductile iron pipe needs and visit our website at ElitePipe Iraq.
Richardpeado
September 24, 2024 @ 2:52 pm
top farmacia online [url=https://tadalafilit.com/#]Cialis generico 5 mg prezzo[/url] farmacia online piГ№ conveniente
KennethmAl
September 24, 2024 @ 5:15 pm
http://sildenafilit.pro/# cerco viagra a buon prezzo
Farmacia online miglior prezzo
PatrickMourO
September 24, 2024 @ 5:58 pm
п»їFarmacia online migliore: Cialis generico 20 mg 8 compresse prezzo – migliori farmacie online 2024
JasonHop
September 24, 2024 @ 7:08 pm
Казино Kometa: Лучший Вариант для Виртуальных Азартных игр
В мире виртуальных казино Kometa завоевало популярность благодаря множеству развлечений, щедрым бонусам и высококачественному сервису. Эта платформа удерживает интерес клиентов во всех странах своими уникальными возможностями и регулярными промо. В данной статье мы обсудим, почему Казино Kometa является одной из лучших площадок для азартных игр.
Достоинства Kometa Casino
Одним из ключевых факторов, отличающих Kometa Casino, является фокус на интересы игроков. Платформа гарантирует свыше тысячи игр, в которых все сможет выбрать игру. Это могут быть привычные слоты, а также новые варианты с инновационными возможностями. Приятным дополнением является то, что Казино Kometa предлагает круглосуточную сопровождение пользователей, создавая приятное и безопасное окружение.
Основные характеристики Kometa Casino:
Дата запуска: 2024
Сертификация: Curacao
Количество игр: Свыше тысячи
Поддержка: Круглосуточная чат и email
Мобильный доступ: Да
Варианты платежей: Skrill
Защита: Шифрование SSL
Приветственные бонусы
Ключевой особенностью Казино Kometa признаны щедрые стартовые предложения для начинающих пользователей. После входа на сайт новички могут воспользоваться к эксклюзивным промоакциям, что дает возможность начать игру с минимальными рисками. Эти бонусы предоставляют комфортные возможности для новичков, предоставляя шанс повысить свои шансы на победу с самого первого захода.
Огромный выбор игр
Казино Kometa предоставляет широкий выбор слотов на любые интересы. Клиенты могут играть привычными автоматами, играми за столом, а также играми с живыми дилерами. Благодаря высокому качеству графики и музыкальному фону, каждый игрок может полностью погрузиться в игровой процесс.
Постоянные события и турниры
Для каждого клиента система часто предлагает турниры и соревнования с выгодными наградами. Турниры организуются регулярно, что делает игровой опыт более захватывающим и захватывающим. Это дает возможность клиентам не только наслаждаться развлечениями, но и зарабатывать дополнительные бонусы и выигрыши.
Почему стоит выбрать
Kometa Casino — это идеальное сочетание широкого ассортимента, отличной поддержки и защищенной платформы. Платформа славится своим фокусом на клиентах и желанием улучшать игровой опыт. Без учета опыта, любой откроет в Kometa Casino то, что сделает его пребывание на сайте увлекательным и комфортным.
Становитесь частью Казино Kometa и играйте с удовольствием яркими эмоциями и захватывающими играми всегда!
EdgarPiede
September 24, 2024 @ 7:57 pm
farmacie online affidabili: farmacia online migliore – farmacia online senza ricetta
EdgarPiede
September 24, 2024 @ 8:03 pm
farmacie online affidabili: Cialis generico farmacia – Farmacia online piГ№ conveniente
сервис центры в ростове на дону
September 24, 2024 @ 9:20 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем:ремонт крупногабаритной техники в ростове на дону
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
CENDANA77
September 24, 2024 @ 10:32 pm
slot gaming online gacor
MIABET
September 24, 2024 @ 10:33 pm
PLATFORM GAME ONLINE
Richardpeado
September 25, 2024 @ 1:39 am
viagra online consegna rapida [url=http://sildenafilit.pro/#]viagra subito[/url] viagra originale recensioni
Richardpeado
September 25, 2024 @ 1:45 am
viagra ordine telefonico [url=http://sildenafilit.pro/#]viagra senza prescrizione[/url] cialis farmacia senza ricetta
ремонт кондиционеров сервис центры в москве
September 25, 2024 @ 5:17 am
[url=https://remont-kondicionerov-wik.ru]ремонт кондиционеров москва[/url]
EdgarPiede
September 25, 2024 @ 7:28 am
farmaci senza ricetta elenco: Brufen antinfiammatorio – migliori farmacie online 2024
EdgarPiede
September 25, 2024 @ 7:34 am
farmacie online affidabili: Farmacie che vendono Cialis senza ricetta – Farmacie on line spedizione gratuita
negonikla
September 25, 2024 @ 1:28 pm
https://lorrd.ru/test/pgs/podkoghnye_pryschi_i_ih_osobennosti.html
Richardpeado
September 25, 2024 @ 2:30 pm
Farmacia online miglior prezzo [url=http://tadalafilit.com/#]Cialis generico recensioni[/url] top farmacia online
binance
September 25, 2024 @ 5:30 pm
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article. https://accounts.binance.com/en/register?ref=JHQQKNKN
EdgarPiede
September 25, 2024 @ 7:26 pm
Farmacie online sicure: Cialis generico recensioni – Farmacia online miglior prezzo
EdgarPiede
September 25, 2024 @ 7:32 pm
farmacia online: Ibuprofene 600 prezzo senza ricetta – п»їFarmacia online migliore
24kbet
September 26, 2024 @ 1:23 am
저는 모든 목소리가 소중히 여겨지는 긍정적이고 존중하는 환경을 조성하는 데 전념하고 있습니다. 여러분의 생각을 나누고, 의미 있는 토론에 참여하며, 서로를 지원해 주시길 권장합니다. 함께 영감을 주고 고양시키는 공간을 만들어 봅시다!
또한, 관리자가 제 블로그를 공유해 주시면 감사하겠습니다. 감사합니다!
24kbet
Richardpeado
September 26, 2024 @ 3:08 am
acquistare farmaci senza ricetta [url=https://farmaciait.men/#]farmacie online sicure[/url] Farmacia online miglior prezzo
Richardpeado
September 26, 2024 @ 3:15 am
le migliori pillole per l’erezione [url=http://sildenafilit.pro/#]viagra[/url] viagra naturale
EdgarPiede
September 26, 2024 @ 7:24 am
viagra naturale: viagra senza prescrizione – le migliori pillole per l’erezione
KennethMIP
September 26, 2024 @ 9:14 am
http://www.jili.com
JILI SLOT GAMES: Sự Lựa Chọn Hàng Đầu Cho Các Tín Đồ Casino Trực Tuyến
JILI Casino là một nhà phát hành game nổi tiếng với nhiều năm kinh nghiệm trong ngành công nghiệp giải trí trực tuyến. Tại JILI, chúng tôi cam kết mang đến cho người chơi những trải nghiệm độc đáo và đẳng cấp, thông qua việc đổi mới không ngừng và cải thiện chất lượng từng sản phẩm. Những giá trị cốt lõi của chúng tôi không chỉ dừng lại ở việc tạo ra các trò chơi xuất sắc, mà còn tập trung vào việc cung cấp các tính năng vượt trội để đáp ứng nhu cầu của người chơi trên toàn cầu.
Sự Đa Dạng Trong Các Trò Chơi Slot
JILI nổi tiếng với loạt trò chơi slot đa dạng và hấp dẫn. Từ các slot game cổ điển đến những trò chơi với giao diện hiện đại và tính năng độc đáo, JILI Slot luôn đem đến cho người chơi những phút giây giải trí tuyệt vời. Các trò chơi được thiết kế với đồ họa sống động, âm thanh chân thực và những vòng quay thú vị, đảm bảo rằng người chơi sẽ luôn bị cuốn hút.
Ưu Điểm Nổi Bật Của JILI Casino
Đổi mới và sáng tạo: Mỗi trò chơi tại JILI Casino đều mang đến sự mới mẻ với lối chơi hấp dẫn và giao diện bắt mắt.
Chất lượng cao: JILI không ngừng cải tiến để đảm bảo mỗi sản phẩm đều đạt chất lượng tốt nhất, từ trải nghiệm người chơi đến tính năng trò chơi.
Nền tảng đa dạng: JILI Casino cung cấp nhiều loại game khác nhau, từ slot, bắn cá đến các trò chơi truyền thống, phù hợp với mọi sở thích của người chơi.
Chương Trình Khuyến Mại JILI
JILI Casino không chỉ nổi bật với chất lượng game mà còn thu hút người chơi bởi các chương trình khuyến mại hấp dẫn. Người chơi có thể tham gia vào nhiều sự kiện, từ khuyến mãi nạp tiền, hoàn trả đến các chương trình tri ân dành riêng cho thành viên VIP. Những ưu đãi này không chỉ tăng cơ hội chiến thắng mà còn mang lại giá trị cộng thêm cho người chơi.
Nổ Hủ City Và Các Trò Chơi Hấp Dẫn Khác
JILI không chỉ có slot games mà còn cung cấp nhiều thể loại game đa dạng khác như bắn cá, bài và nhiều trò chơi giải trí khác. Nổi bật trong số đó là Nổ Hủ City – nơi người chơi có thể thử vận may và giành được những giải thưởng lớn. Sự kết hợp giữa lối chơi dễ hiểu và các tính năng độc đáo của Nổ Hủ City chắc chắn sẽ mang lại những khoảnh khắc giải trí đầy thú vị.
Tham Gia JILI Casino Ngay Hôm Nay
Với sự đa dạng về trò chơi, các tính năng vượt trội và những chương trình khuyến mại hấp dẫn, JILI Casino là sự lựa chọn không thể bỏ qua cho những ai yêu thích trò chơi trực tuyến. Hãy truy cập trang web chính thức của JILI ngay hôm nay để trải nghiệm thế giới giải trí không giới hạn và giành lấy những phần thưởng hấp dẫn từ các trò chơi của chúng tôi!
PatrickMourO
September 26, 2024 @ 12:59 pm
migliori farmacie online 2024: Farmacie on line spedizione gratuita – Farmacie online sicure
ремонт телевизоров
September 26, 2024 @ 1:34 pm
сервисный центре предлагает ремонт телевизоров на дому в москве недорого – ремонт телевизоров недорого
Профи ремонт техники
September 26, 2024 @ 4:14 pm
Сервисный центр предлагает ремонт духовых шкафов gaggenau качественый ремонт духового шкафа gaggenau
Richardpeado
September 26, 2024 @ 4:40 pm
farmacia online piГ№ conveniente [url=https://brufen.pro/#]Ibuprofene 600 generico prezzo[/url] farmacie online sicure
Richardpeado
September 26, 2024 @ 4:46 pm
farmacia online senza ricetta [url=https://brufen.pro/#]Ibuprofene 600 prezzo senza ricetta[/url] Farmacia online miglior prezzo
EdgarPiede
September 26, 2024 @ 7:32 pm
acquistare farmaci senza ricetta: Cialis generico 5 mg prezzo – comprare farmaci online all’estero
EdgarPiede
September 26, 2024 @ 7:38 pm
acquisto farmaci con ricetta: Tadalafil generico migliore – farmacia online
RichardKap
September 26, 2024 @ 10:10 pm
DB賭場:最佳線上遊戲平台評價介紹
DB賭場,前身為PM娛樂網站,於2023年徹底更名為【DB多寶遊戲】。這場品牌更新的過渡,DB賭場更加專注於帶來綜合性的線上娛樂體驗,為玩家帶來更廣泛的娛樂項目與全新的遊戲服務。無論是百家樂、運動博彩還是其他受歡迎遊戲,DB遊戲平台都能符合玩家的偏好。
多寶遊戲的推出與擴展 在亞洲賭場市場中,DB遊戲平台快速壯大,成為數不清的玩家的首要選擇平台之一。隨著PM品牌的品牌更名,DB多寶遊戲聚焦於提升使用者體驗,並致力於創造一個穩定、迅速且透明的娛樂環境。從娛樂項目到付款選項,DB賭場不斷尋求卓越,為玩家帶來首選的線上賭場體驗。
DB遊戲平台的遊戲項目與特色
百家樂遊戲 DB賭場最為知名的是其多樣化的百家樂選項。平台帶來多個版本的莊家遊戲,包括經典百家樂和無手續費百家樂,適應各種玩家的期待。透過實時荷官的實時互動,玩家可以享受真實的賭場體驗。
體育博彩 作為一個全面平台,DB遊戲平台還帶來各類賽事投注的投注服務。從足球賽事、籃球到網球賽事等流行體育項目,玩家都可以任何時候參與體育博彩,體驗比賽的快感與投注的樂趣。
促銷活動與獎金 DB遊戲平台頻繁推出廣泛的促銷活動,為新舊會員提供各種折扣與紅利。這些優惠不僅提高了遊戲的刺激感,還為玩家推動更多賺取獎勵的機會。
DB娛樂網站的反響與優勢 在2024年的最新娛樂網站排行榜中,DB娛樂網站獲得了極高評價,並且因其多樣的遊戲選擇、便利的提款效率和廣泛的促銷活動而獲得玩家喜愛。
BrufenBit
September 26, 2024 @ 10:56 pm
Farmacia online miglior prezzo: BRUFEN 600 prezzo in farmacia – farmacie online autorizzate elenco
BrufenBit
September 26, 2024 @ 11:06 pm
acquisto farmaci con ricetta: Brufen 600 prezzo – farmacie online sicure
da 88
September 26, 2024 @ 11:23 pm
Can you be da 88 more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
Michaelwem
September 27, 2024 @ 12:13 am
Farmacie online sicure [url=https://farmaciait.men/#]acquisto farmaci con ricetta[/url] farmacie online affidabili
Davidpania
September 27, 2024 @ 1:59 am
http://brufen.pro/# BRUFEN 600 prezzo in farmacia
BRUFEN 600 prezzo in farmacia
Davidpania
September 27, 2024 @ 2:09 am
http://brufen.pro/# Brufen 600 prezzo
Ibuprofene 600 generico prezzo
Davidpania
September 27, 2024 @ 2:19 am
https://brufen.pro/# BRUFEN 600 prezzo in farmacia
Ibuprofene 600 prezzo senza ricetta
Davidpania
September 27, 2024 @ 2:22 am
https://brufen.pro/# BRUFEN 600 prezzo in farmacia
BRUFEN 600 mg 30 compresse prezzo
dmt cartridges
September 27, 2024 @ 4:51 am
Attractive section of content. I just stumbled upon your site
and in accession capital to assert that I get actually
enjoyed account your blog posts. Any way I’ll be subscribing
to your feeds and even I achievement you access consistently quickly.
Richardpeado
September 27, 2024 @ 5:11 am
acquistare farmaci senza ricetta [url=https://farmaciait.men/#]migliori farmacie online 2024[/url] acquistare farmaci senza ricetta
Richardpeado
September 27, 2024 @ 5:17 am
Farmacia online piГ№ conveniente [url=https://farmaciait.men/#]farmacia online migliore[/url] top farmacia online
Ремонт техники
September 27, 2024 @ 5:56 am
Сервисный центр предлагает ремонт холодильника maunfeld адреса адреса ремонта холодильников maunfeld
EdgarPiede
September 27, 2024 @ 7:28 am
farmacie online autorizzate elenco: Farmacie online sicure – farmacia online
ArnoldNog
September 27, 2024 @ 10:08 am
Rybelsus 7mg: Buy semaglutide pills – Semaglutide pharmacy price
Josephcek
September 27, 2024 @ 11:36 am
lasix tablet: cheap lasix – lasix furosemide 40 mg
Sergiorax
September 27, 2024 @ 2:52 pm
http://rybelsus.tech/# semaglutide
AlfredMef
September 27, 2024 @ 3:49 pm
ventolin drug [url=https://ventolininhaler.pro/#]buy Ventolin[/url] ventolin 4mg
ремонт бытовой техники воронеж
September 27, 2024 @ 4:16 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – ремонт бытовой техники
Daviddoomi
September 27, 2024 @ 7:13 pm
rybelsus generic: rybelsus price – rybelsus cost
Daviddoomi
September 27, 2024 @ 7:18 pm
ventolin price us: ventolin tab 4mg – online ventolin
Sergiorax
September 27, 2024 @ 7:45 pm
https://furosemide.men/# lasix generic name
Charlessmela
September 27, 2024 @ 8:48 pm
Купить жилую бытовку 149 900? (в т.ч. НДС)
Аренда бытовки для проживания
8 000? в месяц (в т.ч. НДС)
Внешний размер 5850х2400х2400(в)
Каркас сварной, верхняя и нижняя рамка из стального гнутого швеллера 120х50х3, стойки стальные из уголка 75х5 или гнутые 90х90х20
Кровля металлическая сварная толщиной 1,5 мм
Защитная окраска грунт-эмалью 3в1 металлокаркаса и кровли
Черновой пол стальной оцинкованный
ArnoldNog
September 27, 2024 @ 9:55 pm
how much is generic neurontin: where to buy neurontin – neurontin 100mg capsule price
da88
September 27, 2024 @ 10:51 pm
Can you be https://da88.promo/ more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
CENDANA77
September 27, 2024 @ 11:15 pm
Thankyou for this post | Game Online : CENDANA77
Garuda88
September 27, 2024 @ 11:15 pm
Thankyou for this post | Game Online : Garuda88
AlfredMef
September 28, 2024 @ 1:48 am
can i buy ventolin over the counter australia [url=https://ventolininhaler.pro/#]ventolin for sale[/url] ventolin best price
AlfredMef
September 28, 2024 @ 1:55 am
how much is ventolin in canada [url=http://ventolininhaler.pro/#]buy Ventolin[/url] cheap ventolin inhaler
Josephcek
September 28, 2024 @ 4:38 am
ordering prednisone: price for 15 prednisone – prednisone uk over the counter
RAPI88
September 28, 2024 @ 5:17 am
Thankyou for this post | Game Online : RAPI88
Sergiorax
September 28, 2024 @ 6:04 am
http://prednisolone.pro/# prednisone 5mg coupon
sogou
September 28, 2024 @ 6:55 am
detik288 detik288 detik288
What i do not realize is in truth how you are not really much more well-preferred
than you might be now. You’re so intelligent.
You realize thus considerably when it comes to this subject, made me for my
part consider it from so many various angles.
Its like men and women aren’t involved until it is one thing
to accomplish with Woman gaga! Your personal stuffs nice.
At all times handle it up!
Daviddoomi
September 28, 2024 @ 7:05 am
ventolin uk price: Ventolin inhaler best price – ventolin script
Daviddoomi
September 28, 2024 @ 7:10 am
lasix medication: furosemide 40mg – lasix online
Sergiorax
September 28, 2024 @ 12:31 pm
https://prednisolone.pro/# where can i get prednisone
ремонт macbook в москве
September 28, 2024 @ 12:43 pm
Профессиональный сервисный центр по ремонту компьютеров и ноутбуков в Москве.
Мы предлагаем: профессиональный ремонт macbook
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Josephgox
September 28, 2024 @ 2:11 pm
furosemide 40mg: cheap lasix – furosemide 100mg
Josephgox
September 28, 2024 @ 3:19 pm
neurontin 800 mg tablets best price: prescription medication neurontin – neurontin cap 300mg
Daviddoomi
September 28, 2024 @ 6:46 pm
neurontin 300 mg coupon: neurontin 200 mg – neurontin 200 mg
Daviddoomi
September 28, 2024 @ 6:51 pm
lasix 40mg: furosemide online – buy furosemide online
Capitlip
September 28, 2024 @ 11:32 pm
Аренда яхты на Пхукете https://arendayachtphuket.ru/ Проведите незабываемый день на яхте с друзьями, организовав барбекю на палубе и водные игры.
Capitlip
September 28, 2024 @ 11:35 pm
Аренда яхты Тайланд https://arendayachtphuket.ru/ Наслаждайтесь индивидуальным обслуживанием на борту, включая личного шеф-повара и экипаж, готовых выполнить все ваши запросы.
Sergiorax
September 28, 2024 @ 11:42 pm
https://ventolininhaler.pro/# ventolin medicine
Josephgox
September 29, 2024 @ 1:46 am
purchase prednisone canada: prednisone 20 mg tablets – prednisone 20mg tab price
Josephgox
September 29, 2024 @ 2:59 am
prednisone 475: 2.5 mg prednisone daily – prednisone 2 mg daily
Sergiorax
September 29, 2024 @ 5:50 am
https://gabapentin.site/# neurontin pills for sale
Daviddoomi
September 29, 2024 @ 5:50 am
prescription drug neurontin: neurontin gabapentin – 300 mg neurontin
Daviddoomi
September 29, 2024 @ 5:55 am
prednisone in canada: prednisone acetate – prednisone tablet 100 mg
Josephcek
September 29, 2024 @ 6:10 am
neurontin cap 300mg: buy neurontin 100 mg – neurontin singapore
اسرار البلوت
September 29, 2024 @ 2:54 pm
Good way of explaining, and pleasant piece of writing to obtain facts about my presentation topic, which i am going to deliver
in academy.
Sergiorax
September 29, 2024 @ 5:22 pm
http://prednisolone.pro/# prednisone otc uk
сервис центры в тюмени
September 29, 2024 @ 5:25 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: ремонт крупногабаритной техники в тюмени
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Charlessmela
September 29, 2024 @ 5:52 pm
Купить бытовку под раздевалку 149 900? (в т.ч. НДС)
Аренда бытовок под раздевалку
8 000? в месяц (в т.ч. НДС)
внешний размер 6000х2400х2500(в);
окно ПВХ с поворотно-откидной створкой;
утепленные пол, потолок, стены;
отделка потолка панелями ПВХ;
отделка стен ДВП, линолеум на полу
Daviddoomi
September 29, 2024 @ 6:05 pm
ventolin 2mg tablet: ventolin prices – ventolin 2mg
Daviddoomi
September 29, 2024 @ 6:11 pm
lasix pills: furosemide online – lasix for sale
Josephgox
September 29, 2024 @ 7:55 pm
rybelsus price: Buy compounded semaglutide online – Semaglutide pharmacy price
vicclub
September 29, 2024 @ 8:41 pm
Can you be vicclub more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
vic club
September 29, 2024 @ 8:52 pm
Can you be vic club more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
togelup
September 30, 2024 @ 12:37 am
slot demo slot demo slot demo
Wow, amazing blog layout! How long have you been blogging for?
you made blogging look easy. The overall look of your
site is magnificent, let alone the content!
Jameskem
September 30, 2024 @ 1:18 am
http://indiadrugs.pro/# pharmacy website india
Ремонт кондиционеров в Москве
September 30, 2024 @ 5:28 am
Профессиональный сервисный центр по ремонту кондиционеров в Москве.
Мы предлагаем: срочный ремонт кондиционеров москва
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
44 Winchester ammo
September 30, 2024 @ 5:37 am
Thanks very nice blog!
Ремонт гироскутеров в Москве
September 30, 2024 @ 6:47 am
Профессиональный сервисный центр по ремонту гироскутеров в Москве.
Мы предлагаем: ремонт гироскутеров
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Ремонт моноблоков
September 30, 2024 @ 6:53 am
Профессиональный сервисный центр по ремонту моноблоков в Москве.
Мы предлагаем: ремонт моноблоков москва
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Jameskem
September 30, 2024 @ 7:36 am
https://indiadrugs.pro/# top online pharmacy india
Jameskem
September 30, 2024 @ 12:29 pm
http://mexicanpharma.icu/# medicine in mexico pharmacies
RichardKap
September 30, 2024 @ 7:00 pm
娛樂城推薦
DB娛樂城:頂級線上娛樂網站評價介紹
DB遊戲平台,前身為PM娛樂城,於2023年徹底更名為【DB多寶遊戲】。這場品牌更新的期間,DB娛樂城更加專注於提供綜合性的線上服務體驗,為玩家推動更多的選擇範圍與獨特的遊戲服務。無論是百家樂、體育博彩還是其他熱門娛樂項目,DB遊戲平台都能適應玩家的興趣。
多寶遊戲的推出與發展 在亞洲線上娛樂市場中,DB娛樂城快速崛起,成為大量玩家的首選平台之一。隨著PM集團的品牌重塑,DB多寶遊戲聚焦於提升客戶體驗,並努力建立一個安全、快速且公平的賭場環境。從遊戲內容到結算方式,DB娛樂城不斷尋求卓越,為玩家帶來最佳的線上娛樂服務。
DB娛樂網站的遊戲項目與亮點
百家樂遊戲 DB賭場最為著名的是其多樣化的百家樂玩法。平台推出多個版本的百家樂玩法,包括常見百家樂和無佣金百家樂,符合各種玩家的需求。透過直播荷官的實時互動,玩家可以感受真實的賭場體驗。
體育博彩 作為一個綜合娛樂平台,DB賭場還推出各類賽事投注的投注選項。從足球、籃球遊戲到網球等受歡迎賽事,玩家都可以隨處體驗體育博彩,感受賽事的緊張感與博彩的快感。
促銷活動與獎金 DB遊戲平台經常推出廣泛的促銷活動,為新老玩家推動各種福利與獎勵。這些優惠不僅增加了遊戲的刺激感,還為玩家帶來更多賺取獎金的機會。
DB遊戲平台的回饋與亮點 在2024年的最新遊戲平台排行榜中,DB娛樂城獲得了卓越評價,並且因其廣泛的遊戲選擇、便利的提款效率和豐富的促銷活動而深受玩家喜愛。
Ремонт Айпадов в Москве
September 30, 2024 @ 10:30 pm
Профессиональный сервисный центр по ремонту планшетов в том числе Apple iPad.
Мы предлагаем: починка айпада в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
bong da lu
September 30, 2024 @ 11:01 pm
https://cloudim.copiny.com/question/details/id/891279
Jameskem
September 30, 2024 @ 11:43 pm
https://mexicanpharma.icu/# reputable mexican pharmacies online
bong da lu
October 1, 2024 @ 12:55 am
https://gravatar.com/zestful3698555eca
bong da lu
October 1, 2024 @ 2:34 am
https://pt.picmix.com/profile/Bongdalu0101
Charlessmela
October 1, 2024 @ 3:03 am
купить строительную бытовку
Купить жилую бытовку 149 900? (в т.ч. НДС)
Аренда бытовки для проживания
8 000? в месяц (в т.ч. НДС)
Внешний размер 5850х2400х2400(в)
Каркас сварной, верхняя и нижняя рамка из стального гнутого швеллера 120х50х3, стойки стальные из уголка 75х5 или гнутые 90х90х20
Кровля металлическая сварная толщиной 1,5 мм
Защитная окраска грунт-эмалью 3в1 металлокаркаса и кровли
Черновой пол стальной оцинкованный
bong da lu
October 1, 2024 @ 3:45 am
https://notabug.org/Bongdalu0101
RogerRoR
October 1, 2024 @ 5:16 am
online canadian pharmacy reviews: Canadian Pharmacy – canadian family pharmacy
RogerRoR
October 1, 2024 @ 5:23 am
buy prescription drugs from india: Indian pharmacy online – cheapest online pharmacy india
Jameskem
October 1, 2024 @ 6:02 am
http://mexicanpharma.icu/# medication from mexico pharmacy
RobertSek
October 1, 2024 @ 7:20 am
mexican online pharmacies prescription drugs [url=https://mexicanpharma.icu/#]mexican pharma[/url] medicine in mexico pharmacies
Jameskem
October 1, 2024 @ 10:41 am
http://mexicanpharma.icu/# reputable mexican pharmacies online
RogerRoR
October 1, 2024 @ 5:15 pm
п»їlegitimate online pharmacies india: indian pharmacy – best india pharmacy
RogerRoR
October 1, 2024 @ 5:22 pm
reputable mexican pharmacies online: mexican pharma – mexican drugstore online
Jameskem
October 1, 2024 @ 5:24 pm
http://mexicanpharma.icu/# mexico pharmacies prescription drugs
Heather R. X. Dixon
October 1, 2024 @ 8:52 pm
From the insightful commentary to the captivating writing, every word of this post is top-notch. Kudos to the author for producing such fantastic content.
lucky 88
October 1, 2024 @ 10:39 pm
Can you be lucky 88 more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
Ремонт посудомоечных машин в Москве
October 1, 2024 @ 11:33 pm
Профессиональный сервисный центр по ремонту посудомоечных машин с выездом на дом в Москве.
Мы предлагаем: ремонт посудомойки москва
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
da 88
October 2, 2024 @ 1:44 am
Can you be da 88 more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
da88tube
October 2, 2024 @ 2:03 am
https://smotra.ru/users/da88tube/
da88
October 2, 2024 @ 3:34 am
http://emseyi.com/user/DA88tube
Jameskem
October 2, 2024 @ 4:13 am
https://indiadrugs.pro/# п»їlegitimate online pharmacies india
hoki777
October 2, 2024 @ 8:47 am
mawartoto login mawartoto login mawartoto login
What’s up to every body, it’s my first pay a quick visit of this blog; this webpage consists of amazing and
truly good stuff in support of readers.
RobertSek
October 2, 2024 @ 9:55 am
Online medicine order [url=http://indiadrugs.pro/#]online Indian pharmacy[/url] indian pharmacy paypal
Jameskem
October 2, 2024 @ 3:12 pm
http://mexicanpharma.icu/# reputable mexican pharmacies online
Jameskem
October 2, 2024 @ 8:05 pm
https://mexicanpharma.icu/# mexican rx online
Сервисный центр в Москве
October 3, 2024 @ 1:25 am
Сервисный центр предлагает стоимость ремонта планшета hp адреса ремонта планшетов hp
Jameskem
October 3, 2024 @ 2:49 am
https://canadapharma.shop/# canadian pharmacies comparison
Ремонт МФУ в Москве
October 3, 2024 @ 3:33 am
Профессиональный сервисный центр по ремонту МФУ в Москве.
Мы предлагаем: ремонт мфу москва
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Stephentot
October 3, 2024 @ 6:11 am
https://clssansordonnance.icu/# Pharmacie sans ordonnance
HeathDex
October 3, 2024 @ 6:53 am
pharmacie en ligne livraison europe: pharmacie en ligne – trouver un mГ©dicament en pharmacie
HeathDex
October 3, 2024 @ 6:58 am
pharmacie en ligne: cialis sans ordonnance – trouver un mГ©dicament en pharmacie
favorites carts
October 3, 2024 @ 7:17 am
Great article.
aryan
October 3, 2024 @ 7:57 am
Laptops are essential devices for work, education, and entertainment, offering portability and versatility. They come in various configurations, catering to different needs, from basic models for browsing and document editing to high-performance machines for gaming
alex
October 3, 2024 @ 7:57 am
I really enjoyed this article! Your tips on incorporating mindfulness into everyday life are practical and easy to follow. It’s amazing how small changes can make such a big difference. Thank you for sharing your insights!”k
jhon
October 3, 2024 @ 7:58 am
“I really enjoyed this article! Your tips on incorporating mindfulness into everyday life are practical and easy to follow. It’s amazing how small changes can make such a big difference. Thank you for sharing your insights!”
mike
October 3, 2024 @ 7:58 am
Ultra Instinct is a powerful and advanced state of heightened combat ability in the “Dragon Ball” series. It allows a fighter to react and move without consciously thinking, enabling them to dodge and counterattacks instinctively.
alex
October 3, 2024 @ 7:59 am
“Thank you for your thoughtful and engaging writing! Your ability to convey complex ideas simply makes your work enjoyable and accessible. I always look forward to your insights and perspectives!”
kevin
October 3, 2024 @ 7:59 am
This form is characterized by extreme speed, precision, and power, as well as a calm, focused mindset. Goku is the most notable character to achieve Ultra Instinct, using it to face formidable opponents. Its mastery requires immense physical and mental training, as it pushes the body to operate on pure reflex, transcending traditional fighting limits.
alex
October 3, 2024 @ 8:01 am
“Thank you for your thoughtful and engaging writing! Your ability to convey complex ideas simply makes your work enjoyable and accessible. I always look forward to your insights and perspectives!”
talha
October 3, 2024 @ 8:09 am
Being a good writer is not just about knowing how to string words together; it’s about crafting a narrative that resonates with readers. A good writer possesses the ability to convey emotions, paint vivid images, and invite readers into their world. Through careful attention to detail and a passion for storytelling, they can turn the mundane into the extraordinary. Whether it’s a whimsical tale or a poignant reflection, a skilled writer knows that the magic lies not just in the words, but in the connections they create with their audience.
taha
October 3, 2024 @ 8:10 am
A good writer weaves words into captivating stories, evoking emotions and sparking imagination. They connect with readers, transforming ordinary moments into extraordinary narratives that resonate long after the last page.
Goku
October 3, 2024 @ 8:10 am
A good writer possesses the power to inspire and engage through their words. They blend creativity with clarity, crafting narratives that captivate readers and leave a lasting impact on their hearts.
Charlescoamp
October 3, 2024 @ 8:14 am
pharmacie en ligne france livraison internationale [url=https://clssansordonnance.icu/#]Cialis generique prix[/url] pharmacie en ligne avec ordonnance
Stephentot
October 3, 2024 @ 11:59 am
https://pharmaciepascher.pro/# pharmacie en ligne sans ordonnance
HeathDex
October 3, 2024 @ 3:26 pm
pharmacies en ligne certifiГ©es: pharmacie en ligne – pharmacie en ligne pas cher
Stephentot
October 3, 2024 @ 5:43 pm
http://clssansordonnance.icu/# pharmacie en ligne pas cher
Ремонт принтеров
October 3, 2024 @ 8:38 pm
Профессиональный сервисный центр по ремонту принтеров в Москве.
Мы предлагаем: принтеры ремонт выезд дом
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
HeathDex
October 3, 2024 @ 11:56 pm
pharmacie en ligne sans ordonnance: pharmacie en ligne pas cher – pharmacie en ligne sans ordonnance
Charlescoamp
October 4, 2024 @ 12:54 am
trouver un mГ©dicament en pharmacie [url=http://pharmaciepascher.pro/#]Pharmacies en ligne certifiees[/url] Pharmacie en ligne livraison Europe
Registrácia na binance
October 4, 2024 @ 1:56 am
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
ремонт техники в уфе
October 4, 2024 @ 3:36 am
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем:ремонт бытовой техники в уфе
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
da88
October 4, 2024 @ 5:31 am
https://groups.google.com/g/da88tube/c/lr0EkjveHLk
balap toto
October 4, 2024 @ 6:14 am
birutoto birutoto birutoto
Hello there! Do you know if they make any plugins to help with SEO?
I’m trying to get my blog to rank for some targeted
keywords but I’m not seeing very good results.
If you know of any please share. Thanks!
BrooksMaimb
October 4, 2024 @ 7:18 am
Kantorbola adalah situs gaming online terbaik di indonesia , kunjungi situs RTP kantor bola untuk mendapatkan informasi akurat rtp diatas 95% . Kunjungi juga link alternatif kami di kantorbola77 dan kantorbola99 .
Ремонт плоттеров
October 4, 2024 @ 8:09 am
Профессиональный сервисный центр по ремонту плоттеров в Москве.
Мы предлагаем: ремонт широкоформатных плоттеров
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Stephentot
October 4, 2024 @ 8:17 am
https://pharmaciepascher.pro/# pharmacie en ligne france livraison internationale
Charlescoamp
October 4, 2024 @ 11:23 am
Viagra vente libre pays [url=http://vgrsansordonnance.com/#]Viagra femme ou trouver[/url] SildГ©nafil Teva 100 mg acheter
Ремонт объективов в Москве
October 4, 2024 @ 1:03 pm
Профессиональный сервисный центр по ремонту объективов в Москве.
Мы предлагаем: ремонт объектив фотоаппарат
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
gbo338
October 4, 2024 @ 2:07 pm
pas77 pas77
pas77
whoah this blog is wonderful i like studying your
articles. Keep up the great work! You know, lots of individuals are hunting round for this
info, you can help them greatly.
HeathDex
October 4, 2024 @ 4:32 pm
acheter mГ©dicament en ligne sans ordonnance: Cialis generique prix – Achat mГ©dicament en ligne fiable
Charlescoamp
October 4, 2024 @ 5:23 pm
Pharmacie Internationale en ligne [url=http://clssansordonnance.icu/#]cialis prix[/url] pharmacie en ligne pas cher
HeathDex
October 5, 2024 @ 12:32 am
pharmacie en ligne livraison europe: pharmacie en ligne pas cher – pharmacie en ligne pas cher
Chloe
October 5, 2024 @ 2:59 am
Ich stehe total auf manner die wissen was sie wollen and free online live webcams auf geile gesprache! Wenn du auf junge curvy blondienen mit gro?en Brusten stehst bist du bei mir genau richtig.
Ремонт серверов в Москве
October 5, 2024 @ 4:11 am
Профессиональный сервисный центр по ремонту серверов в Москве.
Мы предлагаем: ремонт серверных материнских плат
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Anonymous
October 5, 2024 @ 6:00 am
Unconditional love is basic goodness and the total acceptance of someone
binance
October 5, 2024 @ 7:43 am
Your article helped me a lot, is there any more related content? Thanks!
HeathDex
October 5, 2024 @ 8:33 am
Sildenafil teva 100 mg sans ordonnance: Sildenafil Viagra – Viagra vente libre pays
HeathDex
October 5, 2024 @ 8:37 am
Viagra femme ou trouver: Meilleur Viagra sans ordonnance 24h – Viagra pas cher inde
Charlescoamp
October 5, 2024 @ 9:33 am
Pharmacie sans ordonnance [url=http://pharmaciepascher.pro/#]Pharmacies en ligne certifiees[/url] trouver un mГ©dicament en pharmacie
Stephentot
October 5, 2024 @ 11:40 am
http://vgrsansordonnance.com/# Viagra homme prix en pharmacie sans ordonnance
HeathDex
October 5, 2024 @ 5:49 pm
vente de mГ©dicament en ligne: pharmacie en ligne sans ordonnance – Pharmacie Internationale en ligne
HeathDex
October 5, 2024 @ 5:54 pm
Viagra homme prix en pharmacie sans ordonnance: viagra sans ordonnance – Viagra 100mg prix
Charlescoamp
October 5, 2024 @ 8:31 pm
pharmacie en ligne france livraison belgique [url=http://pharmaciepascher.pro/#]Achat mГ©dicament en ligne fiable[/url] Pharmacie en ligne livraison Europe
BarryLig
October 6, 2024 @ 1:19 am
https://rybelsus.shop/# rybelsus coupon
JeremyJex
October 6, 2024 @ 1:22 am
ozempic generic [url=https://ozempic.art/#]buy ozempic pills online[/url] buy cheap ozempic
Franktouth
October 6, 2024 @ 6:00 am
https://ozempic.art/# ozempic coupon
JeremyJex
October 6, 2024 @ 6:54 am
ozempic generic [url=https://ozempic.art/#]ozempic[/url] ozempic online
RamonHoato
October 6, 2024 @ 9:54 am
buy ozempic: buy cheap ozempic – ozempic
da88
October 6, 2024 @ 10:59 am
https://git.project-hobbit.eu/da88tube
BarryLig
October 6, 2024 @ 12:40 pm
http://rybelsus.shop/# buy semaglutide online
raja787
October 6, 2024 @ 2:12 pm
partai togel partai togel partai togel
Hello to every one, the contents existing at this website are truly awesome for people knowledge, well, keep up the nice work fellows.
bola88
October 6, 2024 @ 6:44 pm
slot demo slot demo slot demo
Hi there to all, the contents present at this site are
really amazing for people knowledge, well, keep up the nice work fellows.
Nasir Mckinney
October 6, 2024 @ 7:44 pm
I couldn’t stop scrolling and reading, your content is truly one-of-a-kind. Thank you for all the time and effort you put into creating such amazing content.
Ремонт сетевых хранилищ
October 6, 2024 @ 9:46 pm
Профессиональный сервисный центр по ремонту сетевых хранилищ в Москве.
Мы предлагаем: цены на ремонт сетевых хранилищ
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Ремонт сигвеев в Москве
October 6, 2024 @ 10:12 pm
Профессиональный сервисный центр по ремонту сигвеев в Москве.
Мы предлагаем: сигвей ремонт
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
BarryLig
October 7, 2024 @ 12:03 am
https://rybelsus.shop/# buy semaglutide online
JeremyJex
October 7, 2024 @ 12:15 am
rybelsus cost [url=http://rybelsus.shop/#]buy semaglutide online[/url] semaglutide cost
Haroldjaf
October 7, 2024 @ 4:11 am
buy ozempic pills online: buy cheap ozempic – ozempic
negonikla
October 7, 2024 @ 5:08 am
https://defa.com.ru//images/art/pochemu_vam_sleduet_chitaty_statyi_s_sovetami_v_internete.html
Haroldjaf
October 7, 2024 @ 1:57 pm
cheapest rybelsus pills: semaglutide tablets – semaglutide online
Ремонт автомагнитол в Москве
October 7, 2024 @ 4:51 pm
Профессиональный сервисный центр по ремонту автомагнитол в Москве.
Мы предлагаем: ремонт автомагнитол в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
pallets of clothing for sale
October 7, 2024 @ 7:12 pm
Hello, i think that i saw you visited my web site thus i came to “return the favor”.I am
trying to find things to improve my site!I suppose its ok to use a few of your ideas!!
PinUpboacy
October 8, 2024 @ 12:50 am
пин ап: pin up kz – пинап кз
PinUpboacy
October 8, 2024 @ 1:28 am
пин ап казино онлайн https://dixi.kz/ пин ап казино
Ремонт планшетов в Москве
October 8, 2024 @ 7:10 am
Профессиональный сервисный центр по ремонту планшетов в Москве.
Мы предлагаем: ремонт экрана планшета
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
BarryLig
October 8, 2024 @ 9:28 am
http://rybelsus.shop/# buy rybelsus online
сейф купить
October 8, 2024 @ 10:59 am
В магазине сейфов предлагают где купить сейфы в москве сейф купить в москве
Leroytwime
October 8, 2024 @ 6:41 pm
Профессиональный сервисный центр ремонт мобильных устройств ремонт телефонов недорого
cockfight login
October 8, 2024 @ 11:43 pm
أسعى إلى احترام جميع الأصوات وخلق بيئة إيجابية حيث يمكن للجميع التعبير عن آرائهم والمشاركة في حوار هادف ود بعضهم البعض. لنبنِ معًا مساحة نتبادل فيها التشجيع والدعم!
كما أود أن أشكر الجميع على مشاركة مدونتي مع المزيد من الأشخاص. شكرًا لكم!”
24kbet
Darylkew
October 9, 2024 @ 12:18 am
Nếu bạn là một người đam mê cá cược và yêu thích sự hấp dẫn của những con số, thì sảnh lô đề RGbet chính là điểm đến lý tưởng cho bạn. Tôi đã có cơ hội trải nghiệm tại đây và muốn chia sẻ một số ấn tượng cũng như kinh nghiệm của mình.
RGbet nổi bật với mức tỷ lệ trả thưởng cực kỳ hấp dẫn. Trong quá trình chơi, tôi nhận thấy rằng nhà cái này không chỉ cung cấp nhiều loại hình cược mà còn có những mức cược linh hoạt, phù hợp với mọi nhu cầu của người chơi. Bạn có thể bắt đầu với số tiền nhỏ và dần dần tăng lên khi đã nắm bắt được các quy luật và cách thức hoạt động của trò chơi. Sự đa dạng trong các tùy chọn cược giúp cho trải nghiệm chơi không bị nhàm chán, từ cược lô, đề đến các loại cược khác.
Giao diện của RGbet rất thân thiện, dễ dàng điều hướng. Ngay cả khi bạn là người mới, bạn vẫn có thể nhanh chóng tìm thấy những thông tin cần thiết và bắt đầu đặt cược chỉ trong vài phút. Tôi đặc biệt ấn tượng với hệ thống đổi thưởng siêu tốc của nhà cái. Sau khi trúng thưởng, quá trình rút tiền diễn ra rất nhanh chóng và minh bạch. Tôi đã nhận được tiền thưởng trong tài khoản của mình chỉ sau vài phút, điều này thực sự tạo cảm giác yên tâm và tin tưởng cho người chơi.
Một điểm đáng chú ý khác là dịch vụ chăm sóc khách hàng của RGbet. Đội ngũ hỗ trợ luôn sẵn sàng 24/7, giúp tôi giải quyết mọi thắc mắc nhanh chóng và hiệu quả. Chỉ cần một tin nhắn, tôi đã có thể nhận được câu trả lời rõ ràng và hữu ích.
Ngoài ra, để tăng cơ hội trúng thưởng, tôi cũng muốn chia sẻ một vài mẹo nhỏ. Đầu tiên, hãy luôn theo dõi các thống kê và xu hướng của những con số để đưa ra quyết định cược hợp lý. Thứ hai, hãy đặt ra ngân sách cho mỗi lần chơi để tránh việc chi tiêu quá đà. Cuối cùng, đừng quên tham gia các chương trình khuyến mãi và ưu đãi mà RGbet thường xuyên tổ chức; đây là cơ hội tuyệt vời để gia tăng cơ hội thắng lớn.
Tóm lại, RGbet thực sự là một nhà cái lô đề uy tín với nhiều lợi thế vượt trội. Nếu bạn đang tìm kiếm một sân chơi lô đề an toàn, hấp dẫn và đổi thưởng nhanh chóng, đừng ngần ngại tham gia ngay hôm nay! Chắc chắn rằng bạn sẽ có những trải nghiệm thú vị và có cơ hội trúng thưởng lớn tại đây.
Forrestjed
October 9, 2024 @ 12:35 am
pin up guncel giris: pin up aviator – pin up aviator
Robertspony
October 9, 2024 @ 12:38 am
пинап казино [url=http://pinupru.site/#]пинап казино[/url] пин ап вход
Сейфы второго класса взломостойкости
October 9, 2024 @ 3:27 am
В магазине сейфов предлагают купить сейф 2 взломостойкие сейфы 2 класса
Forrestjed
October 9, 2024 @ 3:51 am
пинап казино: пин ап казино – пин ап
Santoneine
October 9, 2024 @ 4:17 am
https://pinupkz.tech/# пин ап кз
ремонт бытовой техники в волгограде
October 9, 2024 @ 5:14 am
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: сервис центры бытовой техники волгоград
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Williscen
October 9, 2024 @ 9:11 am
rgbet
RGBET: Trang Chủ Cá Cược Uy Tín và Đa Dạng Tại Việt Nam
RGBET đã khẳng định vị thế của mình là một trong những nhà cái hàng đầu tại châu Á và Việt Nam, với đa dạng các dịch vụ cá cược và trò chơi hấp dẫn. Nhà cái này nổi tiếng với việc cung cấp môi trường cá cược an toàn, uy tín và luôn mang lại trải nghiệm tuyệt vời cho người chơi.
Các Loại Hình Cá Cược Tại RGBET
RGBET cung cấp các loại hình cá cược phong phú bao gồm:
Bắn Cá: Một trò chơi đầy kịch tính và thú vị, phù hợp với những người chơi muốn thử thách khả năng săn bắn và nhận về những phần thưởng giá trị.
Thể Thao: Nơi người chơi có thể tham gia đặt cược vào các trận đấu thể thao đa dạng, từ bóng đá, bóng rổ, đến các sự kiện thể thao quốc tế với tỷ lệ cược hấp dẫn.
Game Bài: Đây là một sân chơi dành cho những ai yêu thích các trò chơi bài như tiến lên miền Nam, phỏm, xì dách, và mậu binh. Các trò chơi được phát triển kỹ lưỡng, đem lại cảm giác chân thực và thú vị.
Nổ Hũ: Trò chơi slot với cơ hội trúng Jackpot cực lớn, luôn thu hút sự chú ý của người chơi. Nổ Hũ RGBET là sân chơi hoàn hảo cho những ai muốn thử vận may và giành lấy những phần thưởng khủng.
Lô Đề: Với tỷ lệ trả thưởng cao và nhiều tùy chọn cược, RGBET là nơi lý tưởng cho những người đam mê lô đề.
Tính Năng Nổi Bật Của RGBET
Một trong những điểm nổi bật của RGBET là giao diện thân thiện, dễ sử dụng và hệ thống bảo mật tiên tiến giúp bảo vệ thông tin người chơi. Ngoài ra, nhà cái này còn hỗ trợ các hình thức nạp rút tiền tiện lợi thông qua nhiều kênh khác nhau như OVO, Gopay, Dana, và các chuỗi cửa hàng như Indomaret và Alfamart.
Khuyến Mãi và Dịch Vụ Chăm Sóc Khách Hàng
RGBET không chỉ nổi bật với các trò chơi đa dạng, mà còn cung cấp nhiều chương trình khuyến mãi hấp dẫn như bonus chào mừng, cashback, và các phần thưởng hàng ngày. Đặc biệt, dịch vụ chăm sóc khách hàng của RGBET hoạt động 24/7 với đội ngũ chuyên nghiệp, sẵn sàng hỗ trợ người chơi qua nhiều kênh liên lạc.
Kết Luận
Với những ưu điểm nổi bật về dịch vụ, bảo mật, và trải nghiệm người dùng, RGBET xứng đáng là nhà cái hàng đầu mà người chơi nên lựa chọn khi tham gia vào thế giới cá cược trực tuyến.
Gabrieldak
October 9, 2024 @ 10:43 am
пинап кз https://pinupru.site/# пин ап казино
пинап казино
Forrestjed
October 9, 2024 @ 10:43 am
pin up casino guncel giris: pin up casino guncel giris – pin up casino giris
Randallblita
October 9, 2024 @ 11:17 am
Trong bối cảnh ngành công nghiệp cá cược trực tuyến ngày càng phát triển, việc lựa chọn một nhà cái uy tín trở nên vô cùng quan trọng đối với những người đam mê cá cược.Nhà cái RGBET nổi lên như một sự lựa chọn hàng đầu đáng để bạn quan tâm, hứa hẹn mang đến cho bạn một trải nghiệm cá cược an toàn, công bằng và thú vị. Từ các trò chơi cá cược đa dạng, dịch vụ chăm sóc khách hàng tận tình đến tỷ lệ cược cạnh tranh, Rgbet sở hữu nhiều ưu điểm vượt trội khiến bạn không thể bỏ qua.Hãy cùng khám phá những lý do tại sao bạn cần quan tâm đến nhà cái Rgbet và tại sao đây nên là lựa chọn hàng đầu của bạn trong thế giới cá cược trực tuyến.
Robertspony
October 9, 2024 @ 12:56 pm
пин ап [url=https://pinupkz.tech/#]pin up kz[/url] пин ап казахстан
Luckyjetraketa
October 9, 2024 @ 4:34 pm
Lucky Jet регистрация и бонусы https://raketa-igra.fun/
Gabrieldak
October 9, 2024 @ 5:33 pm
пин ап кз http://pinupkz.tech/# пин ап казино
пин ап казино
Forrestjed
October 9, 2024 @ 6:08 pm
pin up casino guncel giris: pin up casino giris – pin-up casino
seo продвижение медицинских сайтов
October 9, 2024 @ 6:50 pm
Тут делают продвижение seo продвижение медицинских сайтов seo-продвижение медицинских сайтов
разработка сайт медицинского центра
October 9, 2024 @ 7:39 pm
Тут делают продвижение разработка сайтов медицинских центров разработка мед сайтов
Robertspony
October 9, 2024 @ 8:02 pm
pin up aviator [url=http://pinupturkey.pro/#]pin up aviator[/url] pin up aviator
Santoneine
October 10, 2024 @ 12:25 am
http://pinupaz.bid/# pinup az
Gabrieldak
October 10, 2024 @ 1:06 am
pin up https://pinupturkey.pro/# pin up casino guncel giris
пинап казино
Forrestjed
October 10, 2024 @ 2:31 am
pin-up bonanza: pin-up casino – pin up casino giris
Gabrieldak
October 10, 2024 @ 8:40 am
пин ап 634 http://pinupturkey.pro/# pin up casino giris
пинап кз
Santoneine
October 10, 2024 @ 10:33 am
http://pinupkz.tech/# пинап кз
взломостойкие сейфы
October 10, 2024 @ 11:23 am
В магазине сейфов предлагают сейфы взломостойкие класса взломостойкий сейф для дома
Robertspony
October 10, 2024 @ 11:24 am
пин ап казино [url=http://pinupkz.tech/#]пин ап казахстан[/url] пин ап казахстан
russiastarz
October 10, 2024 @ 11:26 am
Все доступные акции и бонусы можно найти на https://888starz-russia.online
Ремонт электросамокатов
October 10, 2024 @ 2:33 pm
Профессиональный сервисный центр по ремонту электросамокатов в Москве.
Мы предлагаем: ремонт электросамоката
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Gabrieldak
October 10, 2024 @ 3:49 pm
пинап кз https://pinupturkey.pro/# pin up guncel giris
pin up
Forrestjed
October 10, 2024 @ 6:26 pm
пин ап: пин ап казахстан – пин ап казино онлайн
сео продвижение медицинских сайтов
October 10, 2024 @ 7:47 pm
Тут делают продвижение seo продвижение медицинских сайтов seo-продвижение медицинских сайтов
創建binance帳戶
October 10, 2024 @ 7:57 pm
Thanks for sharing. I read many of your blog posts, cool, your blog is very good.
создание медицинских сайтов под ключ
October 10, 2024 @ 8:26 pm
Тут делают продвижение разработка сайта клиники разработка сайта медицинской клиники
Santoneine
October 10, 2024 @ 8:36 pm
https://pinupkz.tech/# пинап кз
Gabrieldak
October 10, 2024 @ 11:33 pm
пинап кз http://pinupturkey.pro/# pin up casino giris
pin up казино
Greggslape
October 11, 2024 @ 1:11 am
nhà cái
Nếu bạn là một người đam mê cá cược và yêu thích sự hấp dẫn của những con số, thì sảnh lô đề RGbet chính là điểm đến lý tưởng cho bạn. Tôi đã có cơ hội trải nghiệm tại đây và muốn chia sẻ một số ấn tượng cũng như kinh nghiệm của mình.
RGbet nổi bật với mức tỷ lệ trả thưởng cực kỳ hấp dẫn. Trong quá trình chơi, tôi nhận thấy rằng nhà cái này không chỉ cung cấp nhiều loại hình cược mà còn có những mức cược linh hoạt, phù hợp với mọi nhu cầu của người chơi. Bạn có thể bắt đầu với số tiền nhỏ và dần dần tăng lên khi đã nắm bắt được các quy luật và cách thức hoạt động của trò chơi. Sự đa dạng trong các tùy chọn cược giúp cho trải nghiệm chơi không bị nhàm chán, từ cược lô, đề đến các loại cược khác.
Giao diện của RGbet rất thân thiện, dễ dàng điều hướng. Ngay cả khi bạn là người mới, bạn vẫn có thể nhanh chóng tìm thấy những thông tin cần thiết và bắt đầu đặt cược chỉ trong vài phút. Tôi đặc biệt ấn tượng với hệ thống đổi thưởng siêu tốc của nhà cái. Sau khi trúng thưởng, quá trình rút tiền diễn ra rất nhanh chóng và minh bạch. Tôi đã nhận được tiền thưởng trong tài khoản của mình chỉ sau vài phút, điều này thực sự tạo cảm giác yên tâm và tin tưởng cho người chơi.
Một điểm đáng chú ý khác là dịch vụ chăm sóc khách hàng của RGbet. Đội ngũ hỗ trợ luôn sẵn sàng 24/7, giúp tôi giải quyết mọi thắc mắc nhanh chóng và hiệu quả. Chỉ cần một tin nhắn, tôi đã có thể nhận được câu trả lời rõ ràng và hữu ích.
Ngoài ra, để tăng cơ hội trúng thưởng, tôi cũng muốn chia sẻ một vài mẹo nhỏ. Đầu tiên, hãy luôn theo dõi các thống kê và xu hướng của những con số để đưa ra quyết định cược hợp lý. Thứ hai, hãy đặt ra ngân sách cho mỗi lần chơi để tránh việc chi tiêu quá đà. Cuối cùng, đừng quên tham gia các chương trình khuyến mãi và ưu đãi mà RGbet thường xuyên tổ chức; đây là cơ hội tuyệt vời để gia tăng cơ hội thắng lớn.
Tóm lại, RGbet thực sự là một nhà cái lô đề uy tín với nhiều lợi thế vượt trội. Nếu bạn đang tìm kiếm một sân chơi lô đề an toàn, hấp dẫn và đổi thưởng nhanh chóng, đừng ngần ngại tham gia ngay hôm nay! Chắc chắn rằng bạn sẽ có những trải nghiệm thú vị và có cơ hội trúng thưởng lớn tại đây.
Forrestjed
October 11, 2024 @ 2:05 am
пин ап казино вход: pin up – пин ап казино
bolacasino88
October 11, 2024 @ 3:22 am
Thank you for any other wonderful article. Where else may anybody get that type of info in such an ideal method of writing?
I’ve a presentation subsequent week, and I am at the look for such information.
ремонт техники в воронеже
October 11, 2024 @ 4:24 am
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: ремонт крупногабаритной техники в воронеже
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Ремонт телефонов
October 11, 2024 @ 4:34 am
Профессиональный сервисный центр где отремонтировать телефон ближайший сервис по ремонту телефонов
сео продвижение медицинских сайтов
October 11, 2024 @ 2:23 pm
Тут делают продвижение продвижение клиники seo продвижение медицинских сайтов
создание медицинских сайтов под ключ
October 11, 2024 @ 2:28 pm
Тут делают продвижение создать сайт медицинского центра разработка сайта для медицинского центра
создание медицинского сайта под ключ
October 11, 2024 @ 3:00 pm
Тут делают продвижение создание сайта для клиники seo для медицинского центра
SamuelAlaws
October 11, 2024 @ 4:35 pm
https://semaglutide.win/# semaglutide
gruzpoputno
October 11, 2024 @ 6:32 pm
Ищете газель для перевозки из Новосибирска в Бийск? Мы предложим вам подходящий транспорт https://vk.com/gruz_poputno
Randallblita
October 11, 2024 @ 8:56 pm
rgbet
Trong bối cảnh ngành công nghiệp cá cược trực tuyến ngày càng phát triển, việc lựa chọn một nhà cái uy tín trở nên vô cùng quan trọng đối với những người đam mê cá cược.Nhà cái RGBET nổi lên như một sự lựa chọn hàng đầu đáng để bạn quan tâm, hứa hẹn mang đến cho bạn một trải nghiệm cá cược an toàn, công bằng và thú vị. Từ các trò chơi cá cược đa dạng, dịch vụ chăm sóc khách hàng tận tình đến tỷ lệ cược cạnh tranh, Rgbet sở hữu nhiều ưu điểm vượt trội khiến bạn không thể bỏ qua.Hãy cùng khám phá những lý do tại sao bạn cần quan tâm đến nhà cái Rgbet và tại sao đây nên là lựa chọn hàng đầu của bạn trong thế giới cá cược trực tuyến.
Ремонт Apple iMac в Москве
October 11, 2024 @ 10:58 pm
Профессиональный сервисный центр по ремонту моноблоков iMac в Москве.
Мы предлагаем: сервисный центр imac
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
MichaelHoash
October 12, 2024 @ 12:28 am
generic zithromax medicine [url=http://zithromax.company/#]order zithromax[/url] where can i buy zithromax medicine
MatthewAdact
October 12, 2024 @ 1:20 am
where to buy zithromax in canada: generic zithromax – zithromax azithromycin
SamuelAlaws
October 12, 2024 @ 3:55 am
https://stromectol.agency/# ivermectin 50 mg
Stevenprath
October 12, 2024 @ 4:18 am
http://amoxil.llc/# amoxicillin capsules 250mg
generic zithromax 500mg india
seo packages in india
October 12, 2024 @ 6:40 am
We’re a group of volunteers and starting a
new scheme in our community. Your web site offered us
with valuable information to work on. You’ve done an impressive job and our entire community will be grateful
to you.
Take a look at my homepage :: seo packages in india
Greggslape
October 12, 2024 @ 6:45 am
rgbet
Nếu bạn là một người đam mê cá cược và yêu thích sự hấp dẫn của những con số, thì sảnh lô đề RGbet chính là điểm đến lý tưởng cho bạn. Tôi đã có cơ hội trải nghiệm tại đây và muốn chia sẻ một số ấn tượng cũng như kinh nghiệm của mình.
RGbet nổi bật với mức tỷ lệ trả thưởng cực kỳ hấp dẫn. Trong quá trình chơi, tôi nhận thấy rằng nhà cái này không chỉ cung cấp nhiều loại hình cược mà còn có những mức cược linh hoạt, phù hợp với mọi nhu cầu của người chơi. Bạn có thể bắt đầu với số tiền nhỏ và dần dần tăng lên khi đã nắm bắt được các quy luật và cách thức hoạt động của trò chơi. Sự đa dạng trong các tùy chọn cược giúp cho trải nghiệm chơi không bị nhàm chán, từ cược lô, đề đến các loại cược khác.
Giao diện của RGbet rất thân thiện, dễ dàng điều hướng. Ngay cả khi bạn là người mới, bạn vẫn có thể nhanh chóng tìm thấy những thông tin cần thiết và bắt đầu đặt cược chỉ trong vài phút. Tôi đặc biệt ấn tượng với hệ thống đổi thưởng siêu tốc của nhà cái. Sau khi trúng thưởng, quá trình rút tiền diễn ra rất nhanh chóng và minh bạch. Tôi đã nhận được tiền thưởng trong tài khoản của mình chỉ sau vài phút, điều này thực sự tạo cảm giác yên tâm và tin tưởng cho người chơi.
Một điểm đáng chú ý khác là dịch vụ chăm sóc khách hàng của RGbet. Đội ngũ hỗ trợ luôn sẵn sàng 24/7, giúp tôi giải quyết mọi thắc mắc nhanh chóng và hiệu quả. Chỉ cần một tin nhắn, tôi đã có thể nhận được câu trả lời rõ ràng và hữu ích.
Ngoài ra, để tăng cơ hội trúng thưởng, tôi cũng muốn chia sẻ một vài mẹo nhỏ. Đầu tiên, hãy luôn theo dõi các thống kê và xu hướng của những con số để đưa ra quyết định cược hợp lý. Thứ hai, hãy đặt ra ngân sách cho mỗi lần chơi để tránh việc chi tiêu quá đà. Cuối cùng, đừng quên tham gia các chương trình khuyến mãi và ưu đãi mà RGbet thường xuyên tổ chức; đây là cơ hội tuyệt vời để gia tăng cơ hội thắng lớn.
Tóm lại, RGbet thực sự là một nhà cái lô đề uy tín với nhiều lợi thế vượt trội. Nếu bạn đang tìm kiếm một sân chơi lô đề an toàn, hấp dẫn và đổi thưởng nhanh chóng, đừng ngần ngại tham gia ngay hôm nay! Chắc chắn rằng bạn sẽ có những trải nghiệm thú vị và có cơ hội trúng thưởng lớn tại đây.
MichaelHoash
October 12, 2024 @ 7:35 am
purchase neurontin online [url=https://gabapentin.auction/#]order gabapentin[/url] neurontin 900 mg
imbagacor
October 12, 2024 @ 7:48 am
tajirslot tajirslot
tajirslot
Appreciating the dedication you put into your blog and in depth information you provide.
It’s awesome to come across a blog every once in a
while that isn’t the same outdated rehashed
material. Great read! I’ve saved your site and I’m
including your RSS feeds to my Google account.
MatthewAdact
October 12, 2024 @ 9:03 am
semaglutide: Buy compounded semaglutide online – Rybelsus 14 mg price
Stevenprath
October 12, 2024 @ 12:30 pm
https://gabapentin.auction/# buy brand neurontin
zithromax 500 without prescription
MichaelHoash
October 12, 2024 @ 2:56 pm
minocycline 100mg for acne [url=http://stromectol.agency/#]buy stromectol online[/url] generic ivermectin
SamuelAlaws
October 12, 2024 @ 2:57 pm
http://gabapentin.auction/# buy neurontin canadian pharmacy
Anthonygauri
October 12, 2024 @ 4:14 pm
nhà cái
RGBET: Trang Chủ Cá Cược Uy Tín và Đa Dạng Tại Việt Nam
RGBET đã khẳng định vị thế của mình là một trong những nhà cái hàng đầu tại châu Á và Việt Nam, với đa dạng các dịch vụ cá cược và trò chơi hấp dẫn. Nhà cái này nổi tiếng với việc cung cấp môi trường cá cược an toàn, uy tín và luôn mang lại trải nghiệm tuyệt vời cho người chơi.
Các Loại Hình Cá Cược Tại RGBET
RGBET cung cấp các loại hình cá cược phong phú bao gồm:
Bắn Cá: Một trò chơi đầy kịch tính và thú vị, phù hợp với những người chơi muốn thử thách khả năng săn bắn và nhận về những phần thưởng giá trị.
Thể Thao: Nơi người chơi có thể tham gia đặt cược vào các trận đấu thể thao đa dạng, từ bóng đá, bóng rổ, đến các sự kiện thể thao quốc tế với tỷ lệ cược hấp dẫn.
Game Bài: Đây là một sân chơi dành cho những ai yêu thích các trò chơi bài như tiến lên miền Nam, phỏm, xì dách, và mậu binh. Các trò chơi được phát triển kỹ lưỡng, đem lại cảm giác chân thực và thú vị.
Nổ Hũ: Trò chơi slot với cơ hội trúng Jackpot cực lớn, luôn thu hút sự chú ý của người chơi. Nổ Hũ RGBET là sân chơi hoàn hảo cho những ai muốn thử vận may và giành lấy những phần thưởng khủng.
Lô Đề: Với tỷ lệ trả thưởng cao và nhiều tùy chọn cược, RGBET là nơi lý tưởng cho những người đam mê lô đề.
Tính Năng Nổi Bật Của RGBET
Một trong những điểm nổi bật của RGBET là giao diện thân thiện, dễ sử dụng và hệ thống bảo mật tiên tiến giúp bảo vệ thông tin người chơi. Ngoài ra, nhà cái này còn hỗ trợ các hình thức nạp rút tiền tiện lợi thông qua nhiều kênh khác nhau như OVO, Gopay, Dana, và các chuỗi cửa hàng như Indomaret và Alfamart.
Khuyến Mãi và Dịch Vụ Chăm Sóc Khách Hàng
RGBET không chỉ nổi bật với các trò chơi đa dạng, mà còn cung cấp nhiều chương trình khuyến mãi hấp dẫn như bonus chào mừng, cashback, và các phần thưởng hàng ngày. Đặc biệt, dịch vụ chăm sóc khách hàng của RGBET hoạt động 24/7 với đội ngũ chuyên nghiệp, sẵn sàng hỗ trợ người chơi qua nhiều kênh liên lạc.
Kết Luận
Với những ưu điểm nổi bật về dịch vụ, bảo mật, và trải nghiệm người dùng, RGBET xứng đáng là nhà cái hàng đầu mà người chơi nên lựa chọn khi tham gia vào thế giới cá cược trực tuyến.
MatthewAdact
October 12, 2024 @ 4:29 pm
rybelsus: semaglutide – rybelsus cost
binance signup
October 12, 2024 @ 6:33 pm
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me. https://www.binance.com/cs/join?ref=S5H7X3LP
Stevenprath
October 12, 2024 @ 7:13 pm
https://amoxil.llc/# buy amoxicillin online mexico
zithromax prescription
MichaelHoash
October 12, 2024 @ 9:29 pm
amoxicillin 875 125 mg tab [url=https://amoxil.llc/#]amoxicillin cheapest price[/url] where can i get amoxicillin
MatthewAdact
October 12, 2024 @ 11:30 pm
Semaglutide pharmacy price: Rybelsus 14 mg – Semaglutide pharmacy price
SamuelAlaws
October 13, 2024 @ 12:45 am
http://amoxil.llc/# how much is amoxicillin
Stevenprath
October 13, 2024 @ 2:14 am
https://gabapentin.auction/# how much is neurontin
where can i get zithromax
MichaelHoash
October 13, 2024 @ 4:10 am
neurontin 330 mg [url=http://gabapentin.auction/#]buy gabapentin[/url] neurontin 600 mg coupon
Stevenprath
October 13, 2024 @ 8:56 am
http://amoxil.llc/# amoxicillin generic brand
zithromax canadian pharmacy
instant virtual visa gift card
October 13, 2024 @ 9:19 am
Thanks to my father who stated to me about this webpage, this blog is really awesome.
SamuelAlaws
October 13, 2024 @ 10:29 am
http://semaglutide.win/# rybelsus price
MatthewAdact
October 13, 2024 @ 1:36 pm
Rybelsus 14 mg: Rybelsus 14 mg – semaglutide
xslots
October 13, 2024 @ 3:13 pm
Подпишитесь на @android_1xslots и скачайте APK для 1xSlots с промокодом LEGAL1X для получения бонусов https://t.me/android_1xslots
Stevenprath
October 13, 2024 @ 4:01 pm
http://zithromax.company/# zithromax azithromycin
can you buy zithromax online
Azucena Feilds
October 13, 2024 @ 5:18 pm
Very good post. I am going through many of these issues as well..
MichaelHoash
October 13, 2024 @ 5:39 pm
minocycline 50 mg acne [url=https://stromectol.agency/#]buy stromectol online[/url] minocycline manufacturer
PeterSam
October 13, 2024 @ 8:39 pm
st666 app
ST666 – Lựa Chọn Hàng Đầu Trong Thế Giới Cá Cược Trực Tuyến
Trong thời đại mà ngành công nghiệp cá cược trực tuyến đang bùng nổ, việc chọn một nhà cái uy tín là yếu tố quan trọng để đảm bảo trải nghiệm chơi an toàn và đáng tin cậy. ST666 nhanh chóng khẳng định vị thế của mình trên thị trường với những dịch vụ chất lượng và hệ thống bảo mật cao cấp, mang lại trải nghiệm cá cược công bằng, thú vị và an toàn.
Hệ Thống Trò Chơi Đa Dạng
ST666 cung cấp một danh mục trò chơi cá cược đa dạng, đáp ứng mọi nhu cầu và sở thích của người chơi. Từ thể thao, bắn cá, game bài, cho đến xổ số và live casino, tất cả đều được tối ưu hóa để mang lại cảm giác hứng thú cho người tham gia. Đặc biệt, với Live Casino, người chơi sẽ được trải nghiệm cá cược với các dealer thật thông qua hình thức phát trực tiếp, tạo cảm giác như đang tham gia tại các sòng bài thực tế.
Tỷ Lệ Cược Cạnh Tranh
Một trong những ưu điểm nổi bật của ST666 chính là tỷ lệ cược cực kỳ cạnh tranh, giúp người chơi tối ưu hóa lợi nhuận của mình. Các trận đấu thể thao được cập nhật liên tục với tỷ lệ cược hấp dẫn, tạo cơ hội chiến thắng lớn cho những ai yêu thích cá cược thể thao.
Bảo Mật An Toàn Tuyệt Đối
Yếu tố bảo mật luôn là một trong những tiêu chí hàng đầu khi chọn nhà cái cá cược. ST666 sử dụng công nghệ mã hóa hiện đại để bảo vệ thông tin cá nhân và các giao dịch tài chính của người chơi, đảm bảo mọi dữ liệu luôn được bảo mật tuyệt đối. Người chơi hoàn toàn có thể yên tâm khi tham gia cá cược tại đây.
Chăm Sóc Khách Hàng Chuyên Nghiệp
Dịch vụ chăm sóc khách hàng của ST666 luôn sẵn sàng hỗ trợ 24/7 qua nhiều kênh khác nhau như Live Chat, WhatsApp, Facebook, đảm bảo giải đáp mọi thắc mắc và xử lý các vấn đề nhanh chóng. Đội ngũ nhân viên tại đây được đào tạo chuyên nghiệp, tận tâm và chu đáo, luôn đặt lợi ích của khách hàng lên hàng đầu.
Ưu Đãi Hấp Dẫn
ST666 không chỉ nổi bật với các sản phẩm cá cược mà còn thu hút người chơi bởi các chương trình khuyến mãi hấp dẫn. Người chơi có thể nhận được nhiều ưu đãi như 280k miễn phí khi đăng nhập hàng ngày, cùng các chương trình thưởng nạp và hoàn tiền liên tục, giúp tối đa hóa cơ hội chiến thắng.
Lý Do ST666 Là Lựa Chọn Hàng Đầu
Sự kết hợp hoàn hảo giữa giao diện hiện đại, hệ thống trò chơi đa dạng, dịch vụ khách hàng tận tâm và tỷ lệ cược cạnh tranh đã giúp ST666 trở thành một trong những nhà cái cá cược trực tuyến uy tín hàng đầu tại Việt Nam. Đối với những ai đang tìm kiếm một nhà cái an toàn và chuyên nghiệp, ST666 chắc chắn sẽ là lựa chọn không thể bỏ qua.
Kết Luận
Với những ưu điểm vượt trội, ST666 xứng đáng là địa chỉ cá cược uy tín và an toàn cho tất cả người chơi. Đăng ký ngay hôm nay để trải nghiệm hệ thống cá cược đẳng cấp và nhận những ưu đãi hấp dẫn từ ST666!
MatthewAdact
October 13, 2024 @ 9:15 pm
ivermectin 2mg: stromectol price – buy minocycline 50 mg for humans
Stevenprath
October 14, 2024 @ 12:23 am
http://semaglutide.win/# Rybelsus 7mg
generic zithromax over the counter
MichaelHoash
October 14, 2024 @ 1:10 am
neurontin 214 [url=http://gabapentin.auction/#]cheapest gabapentin[/url] buy neurontin online
Harris Mcmaken
October 14, 2024 @ 1:56 am
Can I just say what a relief to uncover an individual who truly knows what they are talking about over the internet. You certainly know how to bring an issue to light and make it important. A lot more people really need to look at this and understand this side of the story. I was surprised you’re not more popular because you definitely possess the gift.
카지노솔루션
October 14, 2024 @ 3:48 am
Great blog! I found it while browsing Yahoo News. I just want to give you a thumbs up for the great information you have got here on this post. i will come back to you website for more information.
MatthewAdact
October 14, 2024 @ 4:42 am
ivermectin 200 mcg: stromectol generic – stromectol 3 mg
ko66
October 14, 2024 @ 6:01 am
https://www.question-ksa.com/user/ko66vip#google_vignette
Stevenprath
October 14, 2024 @ 7:07 am
http://amoxil.llc/# amoxicillin online canada
buy cheap generic zithromax
MichaelHoash
October 14, 2024 @ 7:50 am
amoxicillin 775 mg [url=http://amoxil.llc/#]amoxil best price[/url] amoxicillin discount coupon
сейф третьего класас взломостойкости
October 14, 2024 @ 8:21 am
магазин сейфов предлагает сейф 3 класс в москве сейф 3 класса взломостойкости
MatthewAdact
October 14, 2024 @ 12:17 pm
neurontin pfizer: buy gabapentin – buy neurontin online no prescription
Stevenprath
October 14, 2024 @ 2:25 pm
https://zithromax.company/# zithromax 500mg price
zithromax z-pak
MichaelHoash
October 14, 2024 @ 3:06 pm
zithromax for sale online [url=https://zithromax.company/#]buy zithromax z-pak online[/url] buy generic zithromax no prescription
SamuelAlaws
October 14, 2024 @ 7:05 pm
http://semaglutide.win/# Rybelsus 7mg
MatthewAdact
October 14, 2024 @ 8:03 pm
zithromax purchase online: zithromax best price – where can i buy zithromax in canada
dxomovoe
October 14, 2024 @ 9:38 pm
Drift Boss 我敬佩过去,但永远崇拜未来
MichaelHoash
October 14, 2024 @ 10:39 pm
rybelsus [url=https://semaglutide.win/#]rybelsus price[/url] Buy semaglutide pills
DavidAsync
October 15, 2024 @ 2:00 am
back roller massager
Over 7 years in development, the Rollga Method™ combines trigger points, acupressure points, fascial lines, meridians, and techniques like ART (Active Release Technique), MAT (Muscle Activation Technique), RPR (Reflexive Performance Reset) and others to optimize one’s health thru a new system of self care.
Download the Rollga App to learn about & tap into all that Rollga has to offer!
Ремонт телефонов
October 15, 2024 @ 4:42 am
Профессиональный сервисный центр ремонт телефонов недорого ремонт телефонов москва
Ремонт телефонов
October 15, 2024 @ 5:50 am
Профессиональный сервисный центр по ремонту сотовых телефонов в Москве.
Мы предлагаем: сервис телефонов
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Сервисный центр в Москве
October 15, 2024 @ 9:21 am
Сервисный центр предлагает срочный ремонт тепловизора seek thermal качественный ремонт тепловизоров seek thermal
casino7k
October 15, 2024 @ 9:43 am
Присоединяйтесь к @casino_7kk, чтобы скачать APK для 7k Casino и активировать промокод ANDROID777 https://t.me/casino_7kk
GregoryZooke
October 15, 2024 @ 11:44 am
buy online drugs http://drugs24.pro/# ed drug comparison
GregoryZooke
October 15, 2024 @ 7:39 pm
drugs that cause ed https://mexicanpharm24.pro/# mexico pharmacies prescription drugs
카지노사이트임대
October 15, 2024 @ 10:16 pm
If you are like me and want to get the best content then visit this website daily because of the features it offers
Goodbye content!
카지노사이트
October 15, 2024 @ 10:36 pm
Aw, this was an exceptionally good post. Taking a few minutes and actual effort to create a good article… but what can I say… I hesitate a whole lot and don’t seem to get nearly anything done.
f8bet
October 16, 2024 @ 2:52 am
I am regular reader, how are you everybody? This post posted at this web site is truly pleasant.
сервис центры в челябинске
October 16, 2024 @ 5:11 am
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: сервисные центры в челябинске
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
сервис центры в барнауле
October 16, 2024 @ 2:19 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: сервисные центры по ремонту техники в барнауле
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
onexlots
October 16, 2024 @ 4:28 pm
Получите приветственные бонусы с промокодом LEGAL1X для 1xslots https://1xslots-russia.top/promocode/
DavidAsync
October 16, 2024 @ 6:02 pm
vibrating foam roller
Over 7 years in development, the Rollga Method™ combines trigger points, acupressure points, fascial lines, meridians, and techniques like ART (Active Release Technique), MAT (Muscle Activation Technique), RPR (Reflexive Performance Reset) and others to optimize one’s health thru a new system of self care.
Download the Rollga App to learn about & tap into all that Rollga has to offer!
Индексация ссылок в Google
October 16, 2024 @ 7:33 pm
Начните массовую индексацию ссылок в Google прямо cейчас!
Быстрая индексация ссылок имеет ключевое значение для успеха вашего онлайн-бизнеса. Чем быстрее поисковые системы обнаружат и проиндексируют ваши ссылки, тем быстрее вы сможете привлечь новую аудиторию и повысить позиции вашего сайта в результатах поиска.
Не теряйте времени! Начните пользоваться нашим сервисом для ускоренной индексации внешних ссылок в Google и Yandex. Зарегистрируйтесь сегодня и получите первые результаты уже завтра. Ваш успех в ваших руках!
favorites carts
October 16, 2024 @ 8:25 pm
Hey terrific blog! Does running a blog similar to this
take a great deal of work? I’ve no knowledge of programming however I was hoping to
start my own blog soon. Anyways, if you have any ideas
or techniques for new blog owners please share.
I know this is off subject but I just needed to ask. Thanks a lot!
slot receh
October 16, 2024 @ 8:25 pm
Way cool! Some extremely valid points! I appreciate you writing this post and also the rest of the site is also very good.
Ремонт телефонов
October 17, 2024 @ 1:39 am
Профессиональный сервисный центр по ремонту сотовых телефонов в Москве.
Мы предлагаем: ремонты телефонов
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
GregoryZooke
October 17, 2024 @ 5:50 am
erectile dysfunction treatments http://mexicanpharm24.pro/# mexican border pharmacies shipping to usa
BoatThomime
October 17, 2024 @ 7:11 am
Приватные круизы по островам https://arendayahtathailand.store/ Познакомьтесь немного здешной флорой и фауной, сняв погранкатер для экскурсии числом задушевным районам.
binance Register
October 17, 2024 @ 7:32 am
Your point of view caught my eye and was very interesting. Thanks. I have a question for you.
BoatThomime
October 17, 2024 @ 10:48 am
Романтический вечер на яхте https://arendayahtathailand.store/ Создайте романтический вечер, сняв сторожевик для круиза сверху весте с шампанским и еще закусками.
BoatThomime
October 17, 2024 @ 12:08 pm
Исследуйте красивые острова https://arendakaterphuket.store/ Запланируйте частный день на катере, кае шпендики сумеют резать сверху здесь также зиждить песочные замки сверху пляже.
poşet imalatı
October 17, 2024 @ 12:31 pm
Greetings! Very useful advice in this particular article! It’s the little changes that produce the most important changes. Thanks a lot for sharing!
RobertJaL
October 17, 2024 @ 1:26 pm
Kometa Casino: Идеальный выбор для ценителей игр на удачу
Если вы являетесь увлекаетесь игровыми автоматами и ищете площадку, что дает доступ к широкий ассортимент игровых автоматов и лайв-игр, а плюс выгодные предложения, Kometa Casino — это та площадка, где вы найдете яркие эмоции. Предлагаем узнаем, что превращает Kometa Casino выдающимся и по каким причинам посетители остановились на этом сайте для досуга.
### Ключевые черты Kometa Casino
Казино Kometa — это международная казино, которая была запущена в 2024 году и сейчас уже получила внимание клиентов по всему миру. Вот некоторые моменты, что сравнивают с другими этот сайт:
Функция Сведения
Год Основания 2024
Охват Международная
Объем игр Больше тысячи
Сертификация Кюрасао
Мобильный доступ Присутствует
Методы платежей Visa, Mastercard, Skrill
Техподдержка 24/7 Чат и Email
Бонусы и Акции Щедрые бонусы
Система безопасности Шифрование SSL
### Что привлекает в Kometa Casino?
#### Бонусная система
Одним из самых привлекательных особенностей Казино Kometa является система бонусов. Чем активнее играете, тем лучше призы и бонусы. Программа включает многоуровневую систему:
– **Уровень 1 — Земля**: Кэшбек 3% от потраченных средств за неделю.
– **Уровень 2 — Луна**: Возврат 5% на ставки от 5 000 до 10 000 RUB.
– **Уровень 3 — Венера**: Возврат 7% при игре от 10 001 до 50 000 RUB.
– **Марс (уровень 4)**: 8% кэшбек при сумме ставок от 50 001 до 150 000 рублей.
– **Юпитер (уровень 5)**: Кэшбек 10% при ставках свыше 150 000 ?.
– **Сатурн (уровень 6)**: 11% бонуса.
– **Уровень 7 — Уран**: Максимальный возврат до 12%.
#### Акции и возврат средств
Для сохранения азарт на высоте, Kometa Casino предоставляет еженедельные бонусы, кэшбек и фриспины для новичков. Постоянные подарки способствуют сохранять интерес на в процессе игры.
#### Большое количество развлечений
Свыше тысячи игр, включая автоматы, настольные игры и живое казино, создают Казино Kometa сайтом, где вы найдете подходящее развлечение. Вы можете наслаждаться как классическими слотами, а также новинками от известных разработчиков. Прямые дилеры создают атмосфере ощущение реального казино, воссоздавая дух казино.
BoatThomime
October 17, 2024 @ 1:27 pm
Плавание с друзьями https://arentakaterpattaya.store/ Образуйте эмоционально-приподнятый телевечер, сняв сторожевик для круиза на закате с шампанским и еще закусками.
GregoryZooke
October 17, 2024 @ 2:09 pm
viagra without a prescription http://mexicanpharm24.pro/# mexico drug stores pharmacies
starzbet
October 17, 2024 @ 4:46 pm
https://ketabawo.asia/bez-rubriki/46925.html
Allenpig
October 17, 2024 @ 5:23 pm
играть в аппарат онлайн бесплатно без регистрации
Ремонт телефонов
October 17, 2024 @ 11:06 pm
Профессиональный сервисный центр ремонт телефонов поблизости от меня ремонт мобильных телефонов в москве
BoatThomime
October 17, 2024 @ 11:58 pm
Романтический вечер на яхте аренда катера Паттайя Запланируйте частный шахсей-вахсей сверху катере, кае дети смогут играть сверху воде также сооружать песчаные замки сверху пляже.
Ремонт ноутбуков в Москве
October 18, 2024 @ 12:12 am
Профессиональный сервисный центр по ремонту сотовых телефонов в Москве.
Мы предлагаем: ремонт ноутбука после залития цена
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
GregoryZooke
October 18, 2024 @ 12:56 am
medications for http://drugs24.pro/# best non prescription ed pills
James Moore
October 18, 2024 @ 1:11 am
My friend recommended this website to me. If you are looking for the best content like me, then visit this website every day. Because it provides recommended content. Thank you.
BoatThomime
October 18, 2024 @ 1:12 am
Арендуйте яхту сегодня аренда катера Паттайя Исследуйте подводный мир, сняв катерок с возможностью сноркелинга на наиболее красивых рифах Таиланда.
ramaslot
October 18, 2024 @ 2:33 am
sambal toto sambal toto sambal
toto
I really like it when individuals get together and share views.
Great blog, continue the good work!
DavidAcild
October 18, 2024 @ 2:57 am
spri roller
Over 7 years in development, the Rollga Method™ combines trigger points, acupressure points, fascial lines, meridians, and techniques like ART (Active Release Technique), MAT (Muscle Activation Technique), RPR (Reflexive Performance Reset) and others to optimize one’s health thru a new system of self care.
Download the Rollga App to learn about & tap into all that Rollga has to offer!
Agustingraph
October 18, 2024 @ 5:02 am
http://indianpharmdelivery.com/# best india pharmacy
top online pharmacy india
Garrettjep
October 18, 2024 @ 11:49 am
https://toleshop.ru/
apkboy
October 18, 2024 @ 11:41 pm
https://www.skincityindia.com/2024/10/07/kak-sdelat-umnyj-televizor-poleznym-10-luchshih/
DavidKep
October 19, 2024 @ 12:00 am
이용 안내 및 주요 정보
배송대행 이용방법
배송대행은 해외에서 구매한 상품을 중간지점(배대지)에 보내고, 이를 통해 한국으로 배송받는 서비스입니다. 먼저, 회원가입을 진행하고, 해당 배대지 주소를 이용해 상품을 주문한 후, 배송대행 신청서를 작성하여 배송 정보를 입력합니다. 모든 과정은 웹사이트를 통해 관리되며, 필요한 경우 고객센터를 통해 지원을 받을 수 있습니다.
구매대행 이용방법
구매대행은 해외 쇼핑몰에서 직접 구매가 어려운 경우, 대행 업체가 대신 구매해주는 서비스입니다. 고객이 원하는 상품 정보를 제공하면, 구매대행 신청서를 작성하고 대행료와 함께 결제하면 업체가 구매를 완료해줍니다. 이후 상품이 배대지로 도착하면 배송대행 절차를 통해 상품을 수령할 수 있습니다.
배송비용 안내
배송비용은 상품의 무게, 크기, 배송 지역에 따라 다르며, 계산기는 웹사이트에서 제공됩니다. 부피무게가 큰 제품이나 특수 제품은 추가 비용이 발생할 수 있습니다. 항공과 해운에 따른 요금 차이도 고려해야 합니다.
부가서비스
추가 포장 서비스, 검역 서비스, 폐기 서비스 등이 제공되며, 필요한 경우 신청서를 작성하여 서비스 이용이 가능합니다. 파손 위험이 있는 제품의 경우 포장 보완 서비스를 신청하는 것이 좋습니다.
관/부가세 안내
수입된 상품에 대한 관세와 부가세는 상품의 종류와 가격에 따라 부과됩니다. 이를 미리 확인하고, 추가 비용을 예상하여 계산하는 것이 중요합니다.
수입금지 품목
가스제품(히터), 폭발물, 위험물 등은 수입이 금지된 품목에 속합니다. 항공 및 해상 운송이 불가하니, 반드시 해당 품목을 확인 후 주문해야 합니다.
폐기/검역 안내
검역이 필요한 상품이나 폐기가 필요한 경우, 사전에 관련 부가서비스를 신청해야 합니다. 해당 사항에 대해 미리 안내받고 처리할 수 있도록 주의해야 합니다.
교환/반품 안내
교환 및 반품 절차는 상품을 배송받은 후 7일 이내에 신청이 가능합니다. 단, 일부 상품은 교환 및 반품이 제한될 수 있으니 사전에 정책을 확인하는 것이 좋습니다.
재고관리 시스템
재고관리는 배대지에서 보관 중인 상품의 상태를 실시간으로 확인할 수 있는 시스템입니다. 재고 신청을 통해 상품의 상태를 확인하고, 필요한 경우 배송 또는 폐기 요청을 할 수 있습니다.
노데이타 처리방법
노데이타 상태의 상품은 배송 추적이 어려운 경우 발생합니다. 이런 경우 고객센터를 통해 문의하고 문제를 해결해야 합니다.
소비세환급 및 Q&A
일본에서 상품을 구매할 때 적용된 소비세를 환급받을 수 있는 서비스입니다. 해당 신청서는 구체적인 절차에 따라 작성하고 제출해야 합니다. Q&A를 통해 자주 묻는 질문을 확인하고, 추가 문의 사항은 고객센터에 연락해 해결할 수 있습니다.
고객지원
고객센터는 1:1 문의, 카카오톡 상담 등을 통해 서비스 이용 중 발생하는 문제나 문의사항을 해결할 수 있도록 지원합니다.
서비스 관련 공지사항
파손이 쉬운 제품의 경우, 추가 포장 보완을 반드시 요청해야 합니다.
가스제품(히터)은 통관이 불가하므로 구매 전 확인 바랍니다.
항공 운송 비용이 대폭 인하되었습니다.
CharlesBum
October 19, 2024 @ 12:10 am
Paxlovid buy online: shop – paxlovid pharmacy
Ремонт духовых шкафов в Москве
October 19, 2024 @ 12:15 am
Профессиональный сервисный центр по ремонту духовых шкафов в Москве.
Мы предлагаем: ремонт духовок в москве на дому
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
DavidKep
October 19, 2024 @ 1:09 am
이용 안내 및 주요 정보
배송대행 이용방법
배송대행은 해외에서 구매한 상품을 중간지점(배대지)에 보내고, 이를 통해 한국으로 배송받는 서비스입니다. 먼저, 회원가입을 진행하고, 해당 배대지 주소를 이용해 상품을 주문한 후, 배송대행 신청서를 작성하여 배송 정보를 입력합니다. 모든 과정은 웹사이트를 통해 관리되며, 필요한 경우 고객센터를 통해 지원을 받을 수 있습니다.
구매대행 이용방법
구매대행은 해외 쇼핑몰에서 직접 구매가 어려운 경우, 대행 업체가 대신 구매해주는 서비스입니다. 고객이 원하는 상품 정보를 제공하면, 구매대행 신청서를 작성하고 대행료와 함께 결제하면 업체가 구매를 완료해줍니다. 이후 상품이 배대지로 도착하면 배송대행 절차를 통해 상품을 수령할 수 있습니다.
배송비용 안내
배송비용은 상품의 무게, 크기, 배송 지역에 따라 다르며, 계산기는 웹사이트에서 제공됩니다. 부피무게가 큰 제품이나 특수 제품은 추가 비용이 발생할 수 있습니다. 항공과 해운에 따른 요금 차이도 고려해야 합니다.
부가서비스
추가 포장 서비스, 검역 서비스, 폐기 서비스 등이 제공되며, 필요한 경우 신청서를 작성하여 서비스 이용이 가능합니다. 파손 위험이 있는 제품의 경우 포장 보완 서비스를 신청하는 것이 좋습니다.
관/부가세 안내
수입된 상품에 대한 관세와 부가세는 상품의 종류와 가격에 따라 부과됩니다. 이를 미리 확인하고, 추가 비용을 예상하여 계산하는 것이 중요합니다.
수입금지 품목
가스제품(히터), 폭발물, 위험물 등은 수입이 금지된 품목에 속합니다. 항공 및 해상 운송이 불가하니, 반드시 해당 품목을 확인 후 주문해야 합니다.
폐기/검역 안내
검역이 필요한 상품이나 폐기가 필요한 경우, 사전에 관련 부가서비스를 신청해야 합니다. 해당 사항에 대해 미리 안내받고 처리할 수 있도록 주의해야 합니다.
교환/반품 안내
교환 및 반품 절차는 상품을 배송받은 후 7일 이내에 신청이 가능합니다. 단, 일부 상품은 교환 및 반품이 제한될 수 있으니 사전에 정책을 확인하는 것이 좋습니다.
재고관리 시스템
재고관리는 배대지에서 보관 중인 상품의 상태를 실시간으로 확인할 수 있는 시스템입니다. 재고 신청을 통해 상품의 상태를 확인하고, 필요한 경우 배송 또는 폐기 요청을 할 수 있습니다.
노데이타 처리방법
노데이타 상태의 상품은 배송 추적이 어려운 경우 발생합니다. 이런 경우 고객센터를 통해 문의하고 문제를 해결해야 합니다.
소비세환급 및 Q&A
일본에서 상품을 구매할 때 적용된 소비세를 환급받을 수 있는 서비스입니다. 해당 신청서는 구체적인 절차에 따라 작성하고 제출해야 합니다. Q&A를 통해 자주 묻는 질문을 확인하고, 추가 문의 사항은 고객센터에 연락해 해결할 수 있습니다.
고객지원
고객센터는 1:1 문의, 카카오톡 상담 등을 통해 서비스 이용 중 발생하는 문제나 문의사항을 해결할 수 있도록 지원합니다.
서비스 관련 공지사항
파손이 쉬운 제품의 경우, 추가 포장 보완을 반드시 요청해야 합니다.
가스제품(히터)은 통관이 불가하므로 구매 전 확인 바랍니다.
항공 운송 비용이 대폭 인하되었습니다.
Samuel Lewis
October 19, 2024 @ 2:16 am
Thanks for sharing the information. I really appreciate your efforts.I am looking forward to your next article. Thank you again.
DavidKep
October 19, 2024 @ 2:22 am
일본배대지
이용 안내 및 주요 정보
배송대행 이용방법
배송대행은 해외에서 구매한 상품을 중간지점(배대지)에 보내고, 이를 통해 한국으로 배송받는 서비스입니다. 먼저, 회원가입을 진행하고, 해당 배대지 주소를 이용해 상품을 주문한 후, 배송대행 신청서를 작성하여 배송 정보를 입력합니다. 모든 과정은 웹사이트를 통해 관리되며, 필요한 경우 고객센터를 통해 지원을 받을 수 있습니다.
구매대행 이용방법
구매대행은 해외 쇼핑몰에서 직접 구매가 어려운 경우, 대행 업체가 대신 구매해주는 서비스입니다. 고객이 원하는 상품 정보를 제공하면, 구매대행 신청서를 작성하고 대행료와 함께 결제하면 업체가 구매를 완료해줍니다. 이후 상품이 배대지로 도착하면 배송대행 절차를 통해 상품을 수령할 수 있습니다.
배송비용 안내
배송비용은 상품의 무게, 크기, 배송 지역에 따라 다르며, 계산기는 웹사이트에서 제공됩니다. 부피무게가 큰 제품이나 특수 제품은 추가 비용이 발생할 수 있습니다. 항공과 해운에 따른 요금 차이도 고려해야 합니다.
부가서비스
추가 포장 서비스, 검역 서비스, 폐기 서비스 등이 제공되며, 필요한 경우 신청서를 작성하여 서비스 이용이 가능합니다. 파손 위험이 있는 제품의 경우 포장 보완 서비스를 신청하는 것이 좋습니다.
관/부가세 안내
수입된 상품에 대한 관세와 부가세는 상품의 종류와 가격에 따라 부과됩니다. 이를 미리 확인하고, 추가 비용을 예상하여 계산하는 것이 중요합니다.
수입금지 품목
가스제품(히터), 폭발물, 위험물 등은 수입이 금지된 품목에 속합니다. 항공 및 해상 운송이 불가하니, 반드시 해당 품목을 확인 후 주문해야 합니다.
폐기/검역 안내
검역이 필요한 상품이나 폐기가 필요한 경우, 사전에 관련 부가서비스를 신청해야 합니다. 해당 사항에 대해 미리 안내받고 처리할 수 있도록 주의해야 합니다.
교환/반품 안내
교환 및 반품 절차는 상품을 배송받은 후 7일 이내에 신청이 가능합니다. 단, 일부 상품은 교환 및 반품이 제한될 수 있으니 사전에 정책을 확인하는 것이 좋습니다.
재고관리 시스템
재고관리는 배대지에서 보관 중인 상품의 상태를 실시간으로 확인할 수 있는 시스템입니다. 재고 신청을 통해 상품의 상태를 확인하고, 필요한 경우 배송 또는 폐기 요청을 할 수 있습니다.
노데이타 처리방법
노데이타 상태의 상품은 배송 추적이 어려운 경우 발생합니다. 이런 경우 고객센터를 통해 문의하고 문제를 해결해야 합니다.
소비세환급 및 Q&A
일본에서 상품을 구매할 때 적용된 소비세를 환급받을 수 있는 서비스입니다. 해당 신청서는 구체적인 절차에 따라 작성하고 제출해야 합니다. Q&A를 통해 자주 묻는 질문을 확인하고, 추가 문의 사항은 고객센터에 연락해 해결할 수 있습니다.
고객지원
고객센터는 1:1 문의, 카카오톡 상담 등을 통해 서비스 이용 중 발생하는 문제나 문의사항을 해결할 수 있도록 지원합니다.
서비스 관련 공지사항
파손이 쉬운 제품의 경우, 추가 포장 보완을 반드시 요청해야 합니다.
가스제품(히터)은 통관이 불가하므로 구매 전 확인 바랍니다.
항공 운송 비용이 대폭 인하되었습니다.
Travisrop
October 19, 2024 @ 5:18 am
buy clopidogrel online: plavix price – Cost of Plavix without insurance
Walterfub
October 19, 2024 @ 7:49 am
Kometa Casino: Идеальный вариант для фанатов игр на удачу
Когда вы любите ставками и рассматриваете площадку, что дает доступ к огромный набор слотов и лайв-игр, а плюс большие бонусы, Kometa Casino — это та площадка, где вы найдете незабываемые впечатления. Предлагаем рассмотрим, какие факторы выделяет это казино таким особенным и почему клиенты остановились на этом сайте для своих развлечений.
### Главные особенности Kometa Casino
Kometa Casino — это всемирная платформа, что была основана в 2024-м и сейчас уже получила признание игроков по международно. Вот основные факты, которые выделяют данную платформу:
Черта Сведения
Год создания 2024-й
Охват Глобальная
Количество Игр Свыше 1000
Регистрация Кюрасао
Мобильная Версия Доступна
Способы Оплаты Visa, Mastercard, Skrill
Техподдержка 24/7 Чат и Email
Приветственные бонусы Щедрые бонусы
Безопасность SSL защита
### Почему выбирают Казино Kometa?
#### Программа лояльности
Одним из интересных функций Kometa Casino является поощрительная программа. Чем больше вы играете, тем выше ваши бонусы. Программа включает семи уровней:
– **Уровень 1 — Земля**: Кэшбек 3% 3% от потраченных средств за 7 дней.
– **Луна (уровень 2)**: Возврат 5% на ставки от 5 000 до 10 000 RUB.
– **Венера (уровень 3)**: Кэшбек 7% при ставках от 10 001 до 50 000 ?.
– **Уровень 4 — Марс**: 8% бонуса при ставках от 50 001 до 150 000 RUB.
– **Уровень 5 — Юпитер**: Возврат 10% при общей ставке свыше 150 000 ?.
– **Уровень 6 — Сатурн**: 11% бонуса.
– **Уровень 7 — Уран**: Максимальный кэшбек до 12%.
#### Постоянные бонусы
Для сохранения игровой активности, Kometa Casino предлагает еженедельные бонусы, кэшбек и спины для новых пользователей. Частые бонусы способствуют удерживать внимание на каждой стадии игры.
#### Огромный каталог игр
Огромное количество развлечений, включая игровые машины, настольные игры и игры с живыми дилерами, превращают Kometa Casino сайтом, где вы найдете игру по душе. Игроки могут насладиться стандартными автоматами, а также новинками от ведущих провайдеров. Дилеры в реальном времени придают атмосфере еще больше реализма, воссоздавая дух казино.
CharlesBum
October 19, 2024 @ 9:56 am
buy plavix: check clopidogrel pro – cheap plavix antiplatelet drug
Ремонт техники с выездом на дом в Москве
October 19, 2024 @ 5:49 pm
Сервисный центр предлагает качественый ремонт кофемашины proficook починить кофемашины proficook
RichardBrusy
October 19, 2024 @ 9:51 pm
ivermectin 3mg tab [url=https://stromectol1st.shop/#]stromectol 1st[/url] buy minocycline 100 mg
Arthursmuck
October 20, 2024 @ 2:03 am
https://stromectol1st.shop/# minocycline pac
п»їlegitimate online pharmacies india
Travisrop
October 20, 2024 @ 2:20 am
п»їpaxlovid: buy here – paxlovid cost without insurance
kdg789
October 20, 2024 @ 2:21 am
Incredible! This blog looks exactly like my old one!
It’s on a completely different topic but it has pretty much the same page layout and
design. Wonderful choice of colors!
CharlesBum
October 20, 2024 @ 6:26 am
paxlovid price: shop – paxlovid price
индексация в гугл
October 20, 2024 @ 7:15 am
Полезный сервис быстрого загона ссылок сайта в индексация поисковой системы – индексация в гугл
happybet
October 20, 2024 @ 7:39 am
At this time it sounds like BlogEngine is the best blogging
platform out there right now. (from what I’ve read) Is that what you’re using on your blog?
RichardBrusy
October 20, 2024 @ 7:52 am
buy rybelsus [url=https://rybelsus.icu/#]cheaper[/url] more
Travisrop
October 20, 2024 @ 11:09 am
buy plavix: generic pills – Cost of Plavix without insurance
슬롯솔루션임대
October 20, 2024 @ 12:45 pm
Hi, I’m new here. I found this board and it was really helpful and helped me a lot. I want to give something back and help others. Like you helped me.
Kala Kreke
October 20, 2024 @ 1:49 pm
Delightful read. The passion is visible, or at least, very well faked.
CharlesBum
October 20, 2024 @ 3:54 pm
ivermectin cost in usa: buy online – stromectol tablets 3 mg
RichardBrusy
October 20, 2024 @ 4:58 pm
good price [url=https://rybelsus.icu/#]cheaper[/url] semaglutide
Charlessof
October 20, 2024 @ 7:00 pm
Garansi Kekalahan
NAGAEMPIRE: Platform Sports Game dan E-Games Terbaik di Tahun 2024
Selamat datang di Naga Empire, platform hiburan online yang menghadirkan pengalaman gaming terdepan di tahun 2024! Kami bangga menawarkan sports game, permainan kartu, dan berbagai fitur unggulan yang dirancang untuk memberikan Anda kesenangan dan keuntungan maksimal.
Keunggulan Pendaftaran dengan E-Wallet dan QRIS
Kami memprioritaskan kemudahan dan kecepatan dalam pengalaman bermain Anda:
Pendaftaran dengan E-Wallet: Daftarkan akun Anda dengan mudah menggunakan e-wallet favorit. Proses pendaftaran sangat cepat, memungkinkan Anda langsung memulai petualangan gaming tanpa hambatan.
QRIS Auto Proses dalam 1 Detik: Transaksi Anda diproses instan hanya dalam 1 detik dengan teknologi QRIS, memastikan pembayaran dan deposit berjalan lancar tanpa gangguan.
Sports Game dan Permainan Kartu Terbaik di Tahun 2024
Naga Empire menawarkan berbagai pilihan game menarik:
Sports Game Terlengkap: Dari taruhan olahraga hingga fantasy sports, kami menyediakan sensasi taruhan olahraga dengan kualitas terbaik.
Kartu Terbaik di 2024: Nikmati permainan kartu klasik hingga variasi modern dengan grafis yang menakjubkan, memberikan pengalaman bermain yang tak terlupakan.
Permainan Terlengkap dan Toto Terlengkap
Kami memiliki koleksi permainan yang sangat beragam:
Permainan Terlengkap: Temukan berbagai pilihan permainan seperti slot mesin, kasino, hingga permainan berbasis keterampilan, semua tersedia di Naga Empire.
Toto Terlengkap: Layanan Toto Online kami menawarkan pilihan taruhan yang lengkap dengan odds yang kompetitif, memberikan pengalaman taruhan yang optimal.
Bonus Melimpah dan Turnover Terendah
Bonus Melimpah: Dapatkan bonus mulai dari bonus selamat datang, bonus setoran, hingga promosi eksklusif. Kami selalu memberikan nilai lebih pada setiap taruhan Anda.
Turnover Terendah: Dengan turnover rendah, Anda dapat meraih kemenangan lebih mudah dan meningkatkan keuntungan dari setiap permainan.
Naga Empire adalah tempat yang tepat bagi Anda yang mencari pengalaman gaming terbaik di tahun 2024. Bergabunglah sekarang dan rasakan sensasi kemenangan di platform yang paling komprehensif!
freesocialslots
October 20, 2024 @ 7:28 pm
Discover the excitement of free social slots at CorgiSlots and unlock daily prizes
julia cruise
October 20, 2024 @ 10:21 pm
I found your perspective on it very engaging. For those looking for more details, this page might be helpful: モンクレール 偽物.
CharlesBum
October 21, 2024 @ 3:48 am
rybelsus.icu: semaglutide – semaglutide
RichardBrusy
October 21, 2024 @ 4:23 am
п»їplavix generic [url=http://clopidogrel.pro/#]plavix price[/url] Clopidogrel 75 MG price
Walterfub
October 21, 2024 @ 4:32 am
Казино Kometa: Идеальный выбор для ценителей азартных игр
Когда вы являетесь любите ставками и выбираете площадку, что предлагает большой набор слотов и игр с живыми дилерами, а также выгодные предложения, Kometa Casino — это тот сайт, в котором вы найдете яркие эмоции. Давайте узнаем, что выделяет Kometa Casino уникальным и по каким причинам посетители отдают предпочтение его для игры.
### Главные особенности Казино Kometa
Казино Kometa — это международная игровая платформа, которая была основана в 2024 году и уже получила интерес игроков по глобально. Вот некоторые черты, которые отличают Kometa Casino:
Функция Детали
Год создания 2024
Глобальная доступность Всемирная
Объем игр Больше тысячи
Регистрация Curacao
Мобильная Версия Присутствует
Варианты оплаты Visa, Mastercard, Skrill
Техподдержка 24/7 Чат и Email
Бонусы и Акции Щедрые бонусы
Система безопасности SSL защита
### Что привлекает в Kometa Casino?
#### Бонусная система
Одним из ключевых фишек Казино Kometa становится система бонусов. Чем активнее играете, тем лучше призы и бонусы. Система состоит из 7 этапов:
– **Уровень 1 — Земля**: Возврат 3% от ставок за неделю.
– **Уровень 2 — Луна**: Возврат 5% при ставках от 5 000 до 10 000 рублей.
– **Уровень 3 — Венера**: 7% возврата при игре от 10 001 до 50 000 ?.
– **Марс (уровень 4)**: 8% бонуса при сумме ставок от 50 001 до 150 000 ?.
– **Уровень 5 — Юпитер**: Возврат 10% при ставках свыше 150 000 рублей.
– **Уровень 6 — Сатурн**: Возврат 11%.
– **Уровень 7 — Уран**: Максимальный возврат до 12%.
#### Акции и возврат средств
Для сохранения высокого уровня азарта, Казино Kometa предлагает бонусы каждую неделю, возврат средств и спины для всех новых игроков. Регулярные вознаграждения способствуют сохранять интерес на протяжении всей игры.
#### Большое количество развлечений
Свыше тысячи игр, включая слоты, настольные развлечения и живое казино, делают Kometa Casino местом, где каждый найдет подходящее развлечение. Вы можете наслаждаться как классическими слотами, так и новейшими играми от ведущих провайдеров. Прямые дилеры придают игровому процессу еще больше реализма, создавая атмосферу настоящего казино.
terminalbet
October 21, 2024 @ 7:49 am
Thanks for finally talking about > Understanding spit < Loved it!
카지노솔루션
October 21, 2024 @ 12:12 pm
Someone essentially helped me create an important post that I can say. This is the first time I visited your web page and I am amazed at the research you have made to create this particular post. Amazing great job.
RichardBrusy
October 21, 2024 @ 12:18 pm
paxlovid cost without insurance [url=http://paxlovid1st.shop/#]paxlovid 1st[/url] paxlovid cost without insurance
CharlesBum
October 21, 2024 @ 12:22 pm
paxlovid pharmacy: buy here – Paxlovid over the counter
Ремонт ноутбуков
October 21, 2024 @ 6:34 pm
Профессиональный сервисный центр по ремонту сотовых телефонов в Москве.
Мы предлагаем: сервис по ремонту ноутбуков
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Jamespoulp
October 21, 2024 @ 9:13 pm
สล็อต888
สล็อต888 เป็นหนึ่งในแพลตฟอร์มเกมสล็อตออนไลน์ที่ได้รับความนิยมสูงสุดในปัจจุบัน โดยมีความโดดเด่นด้วยการให้บริการเกมสล็อตที่หลากหลายและมีคุณภาพ รวมถึงฟีเจอร์ที่ช่วยให้ผู้เล่นสามารถเพลิดเพลินกับการเล่นได้อย่างเต็มที่ ในบทความนี้ เราจะมาพูดถึงฟีเจอร์และจุดเด่นของสล็อต888 ที่ทำให้เว็บไซต์นี้ได้รับความนิยมเป็นอย่างมาก
ฟีเจอร์เด่นของ PG สล็อต888
ระบบฝากถอนเงินอัตโนมัติที่รวดเร็ว สล็อต888 ให้บริการระบบฝากถอนเงินแบบอัตโนมัติที่สามารถทำรายการได้ทันที ไม่ต้องรอนาน ไม่ว่าจะเป็นการฝากหรือถอนก็สามารถทำได้ภายในไม่กี่วินาที รองรับการใช้งานผ่านทรูวอลเล็ทและช่องทางอื่น ๆ โดยไม่มีขั้นต่ำในการฝากถอน
รองรับทุกอุปกรณ์ ทุกแพลตฟอร์ม ไม่ว่าคุณจะเล่นจากอุปกรณ์ใดก็ตาม สล็อต888 รองรับทั้งคอมพิวเตอร์ แท็บเล็ต และสมาร์ทโฟน ไม่ว่าจะเป็นระบบ iOS หรือ Android คุณสามารถเข้าถึงเกมสล็อตได้ทุกที่ทุกเวลาเพียงแค่มีอินเทอร์เน็ต
โปรโมชั่นและโบนัสมากมาย สำหรับผู้เล่นใหม่และลูกค้าประจำ สล็อต888 มีโปรโมชั่นต้อนรับ รวมถึงโบนัสพิเศษ เช่น ฟรีสปินและโบนัสเครดิตเพิ่ม ทำให้การเล่นเกมสล็อตกับเราเป็นเรื่องสนุกและมีโอกาสทำกำไรมากยิ่งขึ้น
ความปลอดภัยสูงสุด เรื่องความปลอดภัยเป็นสิ่งที่สล็อต888 ให้ความสำคัญเป็นอย่างยิ่ง เราใช้เทคโนโลยีการเข้ารหัสข้อมูลขั้นสูงเพื่อปกป้องข้อมูลส่วนบุคคลของลูกค้า ระบบฝากถอนเงินยังมีมาตรการรักษาความปลอดภัยที่เข้มงวด ทำให้ลูกค้ามั่นใจในการใช้บริการกับเรา
ทดลองเล่นสล็อตฟรี
สล็อต888 ยังมีบริการให้ผู้เล่นสามารถทดลองเล่นสล็อตได้ฟรี ซึ่งเป็นโอกาสที่ดีในการทดลองเล่นเกมต่าง ๆ ที่มีอยู่บนเว็บไซต์ เช่น Phoenix Rises, Dream Of Macau, Ways Of Qilin, Caishens Wins และเกมยอดนิยมอื่น ๆ ที่มีกราฟิกสวยงามและรูปแบบการเล่นที่น่าสนใจ
ไม่ว่าจะเป็นเกมแนวผจญภัย เช่น Rise Of Apollo, Dragon Hatch หรือเกมที่มีธีมแห่งความมั่งคั่งอย่าง Crypto Gold, Fortune Tiger, Lucky Piggy ทุกเกมได้รับการออกแบบมาเพื่อสร้างประสบการณ์การเล่นที่น่าจดจำและเต็มไปด้วยความสนุกสนาน
บทสรุป
สล็อต888 เป็นแพลตฟอร์มที่ครบเครื่องเรื่องเกมสล็อตออนไลน์ ด้วยฟีเจอร์ที่ทันสมัย โปรโมชั่นที่น่าสนใจ และระบบรักษาความปลอดภัยที่เข้มงวด ทำให้คุณมั่นใจได้ว่าการเล่นกับสล็อต888 จะเป็นประสบการณ์ที่ปลอดภัยและเต็มไปด้วยความสนุก
RichardBrusy
October 21, 2024 @ 10:11 pm
semaglutide [url=http://rybelsus.icu/#]rybelsus cost[/url] rybelsus price
CharlesBum
October 21, 2024 @ 10:48 pm
buy minocycline 50mg online: minocycline 100mg for acne – minocycline efectos secundarios
pileli sineklik
October 22, 2024 @ 2:03 am
pileli sineklik | Pileli sinekliklerin yer kaplamayan tasarımı hakkında verdiğiniz bilgiler çok işime yarayacak. Küçük evler için ideal bir çözüm olduğunu düşünüyorum.
RichardBrusy
October 22, 2024 @ 7:03 am
antiplatelet drug [url=https://clopidogrel.pro/#]best price on generic[/url] plavix best price
CharlesBum
October 22, 2024 @ 8:11 am
Paxlovid over the counter: buy here – paxlovid buy
Travisrop
October 22, 2024 @ 8:29 am
antiplatelet drug: plavix price – antiplatelet drug
Antonyslara
October 22, 2024 @ 10:56 am
สล็อต888 เป็นหนึ่งในแพลตฟอร์มเกมสล็อตออนไลน์ที่ได้รับความนิยมสูงสุดในปัจจุบัน โดยมีความโดดเด่นด้วยการให้บริการเกมสล็อตที่หลากหลายและมีคุณภาพ รวมถึงฟีเจอร์ที่ช่วยให้ผู้เล่นสามารถเพลิดเพลินกับการเล่นได้อย่างเต็มที่ ในบทความนี้ เราจะมาพูดถึงฟีเจอร์และจุดเด่นของสล็อต888 ที่ทำให้เว็บไซต์นี้ได้รับความนิยมเป็นอย่างมาก
ฟีเจอร์เด่นของ PG สล็อต888
ระบบฝากถอนเงินอัตโนมัติที่รวดเร็ว สล็อต888 ให้บริการระบบฝากถอนเงินแบบอัตโนมัติที่สามารถทำรายการได้ทันที ไม่ต้องรอนาน ไม่ว่าจะเป็นการฝากหรือถอนก็สามารถทำได้ภายในไม่กี่วินาที รองรับการใช้งานผ่านทรูวอลเล็ทและช่องทางอื่น ๆ โดยไม่มีขั้นต่ำในการฝากถอน
รองรับทุกอุปกรณ์ ทุกแพลตฟอร์ม ไม่ว่าคุณจะเล่นจากอุปกรณ์ใดก็ตาม สล็อต888 รองรับทั้งคอมพิวเตอร์ แท็บเล็ต และสมาร์ทโฟน ไม่ว่าจะเป็นระบบ iOS หรือ Android คุณสามารถเข้าถึงเกมสล็อตได้ทุกที่ทุกเวลาเพียงแค่มีอินเทอร์เน็ต
โปรโมชั่นและโบนัสมากมาย สำหรับผู้เล่นใหม่และลูกค้าประจำ สล็อต888 มีโปรโมชั่นต้อนรับ รวมถึงโบนัสพิเศษ เช่น ฟรีสปินและโบนัสเครดิตเพิ่ม ทำให้การเล่นเกมสล็อตกับเราเป็นเรื่องสนุกและมีโอกาสทำกำไรมากยิ่งขึ้น
ความปลอดภัยสูงสุด เรื่องความปลอดภัยเป็นสิ่งที่สล็อต888 ให้ความสำคัญเป็นอย่างยิ่ง เราใช้เทคโนโลยีการเข้ารหัสข้อมูลขั้นสูงเพื่อปกป้องข้อมูลส่วนบุคคลของลูกค้า ระบบฝากถอนเงินยังมีมาตรการรักษาความปลอดภัยที่เข้มงวด ทำให้ลูกค้ามั่นใจในการใช้บริการกับเรา
ทดลองเล่นสล็อตฟรี
สล็อต888 ยังมีบริการให้ผู้เล่นสามารถทดลองเล่นสล็อตได้ฟรี ซึ่งเป็นโอกาสที่ดีในการทดลองเล่นเกมต่าง ๆ ที่มีอยู่บนเว็บไซต์ เช่น Phoenix Rises, Dream Of Macau, Ways Of Qilin, Caishens Wins และเกมยอดนิยมอื่น ๆ ที่มีกราฟิกสวยงามและรูปแบบการเล่นที่น่าสนใจ
ไม่ว่าจะเป็นเกมแนวผจญภัย เช่น Rise Of Apollo, Dragon Hatch หรือเกมที่มีธีมแห่งความมั่งคั่งอย่าง Crypto Gold, Fortune Tiger, Lucky Piggy ทุกเกมได้รับการออกแบบมาเพื่อสร้างประสบการณ์การเล่นที่น่าจดจำและเต็มไปด้วยความสนุกสนาน
บทสรุป
สล็อต888 เป็นแพลตฟอร์มที่ครบเครื่องเรื่องเกมสล็อตออนไลน์ ด้วยฟีเจอร์ที่ทันสมัย โปรโมชั่นที่น่าสนใจ และระบบรักษาความปลอดภัยที่เข้มงวด ทำให้คุณมั่นใจได้ว่าการเล่นกับสล็อต888 จะเป็นประสบการณ์ที่ปลอดภัยและเต็มไปด้วยความสนุก
CharlesBum
October 22, 2024 @ 5:05 pm
buy clopidogrel bisulfate: clopidogrel – buy clopidogrel bisulfate
Ремонт Apple iMac в Москве
October 22, 2024 @ 6:24 pm
Профессиональный сервисный центр по ремонту моноблоков iMac в Москве.
Мы предлагаем: срочный ремонт аймака
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Arthursmuck
October 22, 2024 @ 7:35 pm
http://stromectol1st.shop/# ivermectin 0.5% brand name
mail order pharmacy india
Michaelowepe
October 22, 2024 @ 7:47 pm
Bạn đang tìm kiếm những trò chơi hot nhất và thú vị nhất tại sòng bạc trực tuyến? RGBET tự hào giới thiệu đến bạn nhiều trò chơi cá cược đặc sắc, bao gồm Baccarat trực tiếp, máy xèng, cá cược thể thao, xổ số và bắn cá, mang đến cho bạn cảm giác hồi hộp đỉnh cao của sòng bạc! Dù bạn yêu thích các trò chơi bài kinh điển hay những máy xèng đầy kịch tính, RGBET đều có thể đáp ứng mọi nhu cầu giải trí của bạn.
RGBET Trò Chơi của Chúng Tôi
Bạn đang tìm kiếm những trò chơi hot nhất và thú vị nhất tại sòng bạc trực tuyến? RGBET tự hào giới thiệu đến bạn nhiều trò chơi cá cược đặc sắc, bao gồm Baccarat trực tiếp, máy xèng, cá cược thể thao, xổ số và bắn cá, mang đến cảm giác hồi hộp đỉnh cao của sòng bạc! Dù bạn yêu thích các trò chơi bài kinh điển hay những máy xèng đầy kịch tính, RGBET đều có thể đáp ứng mọi nhu cầu giải trí của bạn.
RGBET Trò Chơi Đa Dạng
Thể thao: Cá cược thể thao đa dạng với nhiều môn từ bóng đá, tennis đến thể thao điện tử.
Live Casino: Trải nghiệm Baccarat, Roulette, và các trò chơi sòng bài trực tiếp với người chia bài thật.
Nổ hũ: Tham gia các trò chơi nổ hũ với tỷ lệ trúng cao và cơ hội thắng lớn.
Lô đề: Đặt cược lô đề với tỉ lệ cược hấp dẫn.
Bắn cá: Bắn cá RGBET mang đến cảm giác chân thực và hấp dẫn với đồ họa tuyệt đẹp.
RGBET – Máy Xèng Hấp Dẫn Nhất
Khám phá các máy xèng độc đáo tại RGBET với nhiều chủ đề khác nhau và tỷ lệ trả thưởng cao. Những trò chơi nổi bật bao gồm:
RGBET Super Ace
RGBET Đế Quốc Hoàng Kim
RGBET Pharaoh Treasure
RGBET Quyền Vương
RGBET Chuyên Gia Săn Rồng
RGBET Jackpot Fishing
Vì sao nên chọn RGBET?
RGBET không chỉ cung cấp hàng loạt trò chơi đa dạng mà còn mang đến một hệ thống cá cược an toàn và chuyên nghiệp, đảm bảo mọi quyền lợi của người chơi:
Tốc độ nạp tiền nhanh chóng: Chuyển khoản tại RGBET chỉ mất vài phút và tiền sẽ vào tài khoản ngay lập tức, giúp bạn không bỏ lỡ bất kỳ cơ hội nào.
Game đổi thưởng phong phú: Từ cá cược thể thao đến slot game, RGBET cung cấp đầy đủ trò chơi giúp bạn tận hưởng mọi phút giây thư giãn.
Bảo mật tuyệt đối: Với công nghệ mã hóa tiên tiến, tài khoản và tiền vốn của bạn sẽ luôn được bảo vệ một cách an toàn.
Hỗ trợ đa nền tảng: Bạn có thể chơi trên mọi thiết bị, từ máy tính, điện thoại di động (iOS/Android), đến nền tảng H5.
Tải Ứng Dụng RGBET và Nhận Khuyến Mãi Lớn
Hãy tham gia RGBET ngay hôm nay để tận hưởng thế giới giải trí không giới hạn với các trò chơi thể thao, thể thao điện tử, casino trực tuyến, xổ số, và slot game. Quét mã QR và tải ứng dụng RGBET trên điện thoại để trải nghiệm game tốt hơn và nhận nhiều khuyến mãi hấp dẫn!
Tham gia RGBET để bắt đầu cuộc hành trình cá cược đầy thú vị ngay hôm nay!
Samuel Rodriguez
October 22, 2024 @ 11:10 pm
I am so glad I found this website. I just wanted to say thank you for reading this fantastic book!!
I definitely enjoyed every little bit of it and you
have marked the book to see new content on your website.
ThomasErrop
October 22, 2024 @ 11:24 pm
Полезная информация на сайте. Все что вы хоте знать об интернете полезный сервис
RichardBrusy
October 22, 2024 @ 11:45 pm
rybelsus.icu [url=http://rybelsus.icu/#]Buy semaglutide[/url] more
CharlesBum
October 23, 2024 @ 2:05 am
paxlovid generic: shop – Paxlovid over the counter
Robertjeata
October 23, 2024 @ 7:34 am
Check AML
Tether TRC20 Payment Validation and AML (AML) Practices
As cryptocurrencies like USDT TRC20 rise in usage for rapid and low-cost transfers, the need for safety and compliance with Anti-Money Laundering regulations increases. Here’s how to verify Tether TRC20 transfers and confirm they’re not connected to illicit operations.
What is USDT TRC20?
USDT TRC20 is a stablecoin on the TRX network, priced in correspondence with the USD. Known for its minimal costs and quickness, it is commonly utilized for global payments. Verifying transfers is important to block links to illicit transfers or other unlawful operations.
Verifying TRON-based USDT Transfers
TRX Explorer — This blockchain explorer allows users to monitor and validate Tether TRON-based transactions using a wallet address or transfer code.
Monitoring — Skilled participants can observe suspicious trends such as high-volume or quick transactions to identify unusual actions.
AML and Dirty Cryptocurrency
Financial Crime Prevention (AML) regulations assist block unlawful financial activity in cryptocurrency. Tools like Chain Analysis and Elliptic allow companies and trading platforms to detect and block criminal crypto, which means money connected to illegal activities.
Solutions for Adherence
TRONSCAN — To verify TRON-based USDT transaction details.
Chainalysis and Elliptic — Employed by trading platforms to guarantee AML conformance and monitor illegal actions.
Final Thoughts
Ensuring secure and legal USDT TRC20 payments is essential. Platforms like TRX Explorer and Anti-Money Laundering systems assist guard users from engaging with illicit funds, supporting a secure and regulated crypto environment.
Winford Bruechert
October 23, 2024 @ 8:09 am
Ils proposent une variété de produits et de ressources qui peuvent vraiment aider à explorer cette thématique en toute sécurité. Ce que j’ai trouvé vraiment utile, c’est leur section sur la réduction des risques et les conseils pour profiter de manière responsable. Ça pourrait être un bon complément à cet article !
bolacasino88
October 23, 2024 @ 9:45 am
Howdy I am so happy I found your webpage, I really
found you by accident, while I was researching on Askjeeve for something else,
Anyhow I am here now and would just like to say thanks a lot for a
tremendous post and a all round interesting blog (I also love the
theme/design), I don’t have time to browse it all at the minute but I have saved it and also
included your RSS feeds, so when I have time I will be back to read much more, Please
do keep up the awesome work.
binance Sign Up
October 23, 2024 @ 10:15 am
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
albaslot
October 23, 2024 @ 11:42 am
Hi there, I log on to your new stuff regularly. Your story-telling style is awesome, keep doing what you’re doing!
Elana Knisley
October 23, 2024 @ 3:17 pm
Encore merci pour ce post et continuez votre excellent travail. ??
ремонт техники в перми
October 23, 2024 @ 6:49 pm
Профессиональный сервисный центр по ремонту бытовой техники с выездом на дом.
Мы предлагаем: сервис центры бытовой техники пермь
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Amina Cassell
October 23, 2024 @ 10:38 pm
Ils proposent une variété de produits et de ressources qui peuvent vraiment aider à explorer cette thématique en toute sécurité. Ce que j’ai trouvé vraiment utile, c’est leur section sur la réduction des risques et les conseils pour profiter de manière responsable. Ça pourrait être un bon complément à cet article !
Jamespoulp
October 23, 2024 @ 10:44 pm
เกมสล็อตออนไลน์
สล็อต888 เป็นหนึ่งในแพลตฟอร์มเกมสล็อตออนไลน์ที่ได้รับความนิยมสูงสุดในปัจจุบัน โดยมีความโดดเด่นด้วยการให้บริการเกมสล็อตที่หลากหลายและมีคุณภาพ รวมถึงฟีเจอร์ที่ช่วยให้ผู้เล่นสามารถเพลิดเพลินกับการเล่นได้อย่างเต็มที่ ในบทความนี้ เราจะมาพูดถึงฟีเจอร์และจุดเด่นของสล็อต888 ที่ทำให้เว็บไซต์นี้ได้รับความนิยมเป็นอย่างมาก
ฟีเจอร์เด่นของ PG สล็อต888
ระบบฝากถอนเงินอัตโนมัติที่รวดเร็ว สล็อต888 ให้บริการระบบฝากถอนเงินแบบอัตโนมัติที่สามารถทำรายการได้ทันที ไม่ต้องรอนาน ไม่ว่าจะเป็นการฝากหรือถอนก็สามารถทำได้ภายในไม่กี่วินาที รองรับการใช้งานผ่านทรูวอลเล็ทและช่องทางอื่น ๆ โดยไม่มีขั้นต่ำในการฝากถอน
รองรับทุกอุปกรณ์ ทุกแพลตฟอร์ม ไม่ว่าคุณจะเล่นจากอุปกรณ์ใดก็ตาม สล็อต888 รองรับทั้งคอมพิวเตอร์ แท็บเล็ต และสมาร์ทโฟน ไม่ว่าจะเป็นระบบ iOS หรือ Android คุณสามารถเข้าถึงเกมสล็อตได้ทุกที่ทุกเวลาเพียงแค่มีอินเทอร์เน็ต
โปรโมชั่นและโบนัสมากมาย สำหรับผู้เล่นใหม่และลูกค้าประจำ สล็อต888 มีโปรโมชั่นต้อนรับ รวมถึงโบนัสพิเศษ เช่น ฟรีสปินและโบนัสเครดิตเพิ่ม ทำให้การเล่นเกมสล็อตกับเราเป็นเรื่องสนุกและมีโอกาสทำกำไรมากยิ่งขึ้น
ความปลอดภัยสูงสุด เรื่องความปลอดภัยเป็นสิ่งที่สล็อต888 ให้ความสำคัญเป็นอย่างยิ่ง เราใช้เทคโนโลยีการเข้ารหัสข้อมูลขั้นสูงเพื่อปกป้องข้อมูลส่วนบุคคลของลูกค้า ระบบฝากถอนเงินยังมีมาตรการรักษาความปลอดภัยที่เข้มงวด ทำให้ลูกค้ามั่นใจในการใช้บริการกับเรา
ทดลองเล่นสล็อตฟรี
สล็อต888 ยังมีบริการให้ผู้เล่นสามารถทดลองเล่นสล็อตได้ฟรี ซึ่งเป็นโอกาสที่ดีในการทดลองเล่นเกมต่าง ๆ ที่มีอยู่บนเว็บไซต์ เช่น Phoenix Rises, Dream Of Macau, Ways Of Qilin, Caishens Wins และเกมยอดนิยมอื่น ๆ ที่มีกราฟิกสวยงามและรูปแบบการเล่นที่น่าสนใจ
ไม่ว่าจะเป็นเกมแนวผจญภัย เช่น Rise Of Apollo, Dragon Hatch หรือเกมที่มีธีมแห่งความมั่งคั่งอย่าง Crypto Gold, Fortune Tiger, Lucky Piggy ทุกเกมได้รับการออกแบบมาเพื่อสร้างประสบการณ์การเล่นที่น่าจดจำและเต็มไปด้วยความสนุกสนาน
บทสรุป
สล็อต888 เป็นแพลตฟอร์มที่ครบเครื่องเรื่องเกมสล็อตออนไลน์ ด้วยฟีเจอร์ที่ทันสมัย โปรโมชั่นที่น่าสนใจ และระบบรักษาความปลอดภัยที่เข้มงวด ทำให้คุณมั่นใจได้ว่าการเล่นกับสล็อต888 จะเป็นประสบการณ์ที่ปลอดภัยและเต็มไปด้วยความสนุก
EdwardJoype
October 24, 2024 @ 1:33 am
pin up: pin up 306 – pinup az
Petronila Veltman
October 24, 2024 @ 2:00 am
Merci pour cet article super intéressant sur [thème de l’article] ! Je voulais juste ajouter un point qui pourrait intéresser certains d’entre vous. Si vous êtes curieux ou cherchez des informations supplémentaires sur les produits liés à l’amélioration de l’expérience de discuter et de futur rencontre et du bien-être personnel, j’ai récemment découvert un site très complet, [Chemsexworld.com]( Chemsexworld.com .?
카지노솔루션
October 24, 2024 @ 2:10 am
The sketch is polished, and the subject you wrote is polished.
Nevertheless, you need to build tension about what you want to convey.
Next. If you protect this increase, you will definitely come back sooner.
The same almost often.
ErnestZeW
October 24, 2024 @ 6:19 am
pin up casino: pin-up – pin up azerbaycan
DavidfainS
October 24, 2024 @ 7:57 am
http://1winbrasil.win/# pinup az
пин ап казино
xxxtubebest.com/xpGfW7Gf677Vk
October 24, 2024 @ 8:41 am
My fzmily every tine say that I am killing myy time hwre at web,but I know
I amm getfing experience daily bby readong such fastidious posts.
NormanDyeft
October 24, 2024 @ 10:43 am
เกมบาคาร่า
เล่นบาคาร่าแบบรวดเร็วทันใจกับสปีดบาคาร่า
ถ้าคุณเป็นแฟนตัวยงของเกมไพ่บาคาร่า คุณอาจจะเคยชินกับการรอคอยในแต่ละรอบการเดิมพัน และรอจนดีลเลอร์แจกไพ่ในแต่ละตา แต่คุณรู้หรือไม่ว่า ตอนนี้คุณไม่ต้องรออีกต่อไปแล้ว เพราะ SA Gaming ได้พัฒนาเกมบาคาร่าโหมดใหม่ขึ้นมา เพื่อให้ประสบการณ์การเล่นของคุณน่าตื่นเต้นยิ่งขึ้น!
ที่ SA Gaming คุณสามารถเลือกเล่นไพ่บาคาร่าในโหมดที่เรียกว่า สปีดบาคาร่า (Speed Baccarat) โหมดนี้มีคุณสมบัติพิเศษและข้อดีที่น่าสนใจมากมาย:
ระยะเวลาการเดิมพันสั้นลง — คุณไม่จำเป็นต้องรอนานอีกต่อไป ในโหมดสปีดบาคาร่า คุณจะมีเวลาเพียง 12 วินาทีในการวางเดิมพัน ทำให้เกมแต่ละรอบจบได้รวดเร็ว โดยเกมในแต่ละรอบจะใช้เวลาเพียง 20 วินาทีเท่านั้น
ผลตอบแทนต่อผู้เล่นสูง (RTP) — เกมสปีดบาคาร่าให้ผลตอบแทนต่อผู้เล่นสูงถึง 4% ซึ่งเป็นมาตรฐานความเป็นธรรมที่ผู้เล่นสามารถไว้วางใจได้
การเล่นเกมที่รวดเร็วและน่าตื่นเต้น — ระยะเวลาที่สั้นลงทำให้เกมแต่ละรอบดำเนินไปอย่างรวดเร็ว ทันใจ เพิ่มความสนุกและความตื่นเต้นในการเล่น ทำให้ประสบการณ์การเล่นของคุณยิ่งสนุกมากขึ้น
กลไกและรูปแบบการเล่นยังคงเหมือนเดิม — แม้ว่าระยะเวลาจะสั้นลง แต่กลไกและกฎของการเล่น ยังคงเหมือนกับบาคาร่าสดปกติทุกประการ เพียงแค่ปรับเวลาให้เล่นได้รวดเร็วและสะดวกขึ้นเท่านั้น
นอกจากสปีดบาคาร่าแล้ว ที่ SA Gaming ยังมีโหมด No Commission Baccarat หรือบาคาร่าแบบไม่เสียค่าคอมมิชชั่น ซึ่งจะช่วยให้คุณสามารถเพลิดเพลินไปกับการเล่นได้โดยไม่ต้องกังวลเรื่องค่าคอมมิชชั่นเพิ่มเติม
เล่นบาคาร่ากับ SA Gaming คุณจะได้รับประสบการณ์การเล่นที่สนุก ทันสมัย และตรงใจมากที่สุด!
ErnestZeW
October 24, 2024 @ 11:33 am
пин ап: пинап кз – пин ап
hornyxxx.win/hornyvidph58e3d0635c68812
October 24, 2024 @ 2:23 pm
Thhis is veery interesting, You’re a veery skoilled blogger.
I hae joiined your feeed aand loook forwad to seeking mofe of ylur fantastic post.
Also, I hav shared your website in my sochial networks!
hornyxxx.win/hornyvid776365813
October 24, 2024 @ 3:35 pm
Thiis design is spectacular! Youu definitely know
hhow tto kep a readerr amused. Between your wit and your
videos, I was almost mkved tto start myy ownn blog (well, almost…HaHa!) Wonderful job.
I really enjoyed wha you had too say, and mre than that, howw yoou presented it.
Tooo cool!
pupis
October 24, 2024 @ 4:22 pm
Скачайте легальные БК и делайте ставки на спорт с мобильного устройства
DavidfainS
October 24, 2024 @ 8:08 pm
http://1winrussia.online/# 1xbet официальный сайт
пинап
ремонт телефонов рядом
October 24, 2024 @ 8:38 pm
Недавно разбил экран своего телефона и обратился в этот сервисный центр. Ребята быстро и качественно починили устройство, теперь работает как новый. Очень рекомендую обратиться к ним за помощью. Вот ссылка на их сайт: вызвать мастера по ремонту айфонов.
Michaelowepe
October 24, 2024 @ 8:43 pm
Bạn đang tìm kiếm những trò chơi hot nhất và thú vị nhất tại sòng bạc trực tuyến? RGBET tự hào giới thiệu đến bạn nhiều trò chơi cá cược đặc sắc, bao gồm Baccarat trực tiếp, máy xèng, cá cược thể thao, xổ số và bắn cá, mang đến cho bạn cảm giác hồi hộp đỉnh cao của sòng bạc! Dù bạn yêu thích các trò chơi bài kinh điển hay những máy xèng đầy kịch tính, RGBET đều có thể đáp ứng mọi nhu cầu giải trí của bạn.
RGBET Trò Chơi của Chúng Tôi
Bạn đang tìm kiếm những trò chơi hot nhất và thú vị nhất tại sòng bạc trực tuyến? RGBET tự hào giới thiệu đến bạn nhiều trò chơi cá cược đặc sắc, bao gồm Baccarat trực tiếp, máy xèng, cá cược thể thao, xổ số và bắn cá, mang đến cảm giác hồi hộp đỉnh cao của sòng bạc! Dù bạn yêu thích các trò chơi bài kinh điển hay những máy xèng đầy kịch tính, RGBET đều có thể đáp ứng mọi nhu cầu giải trí của bạn.
RGBET Trò Chơi Đa Dạng
Thể thao: Cá cược thể thao đa dạng với nhiều môn từ bóng đá, tennis đến thể thao điện tử.
Live Casino: Trải nghiệm Baccarat, Roulette, và các trò chơi sòng bài trực tiếp với người chia bài thật.
Nổ hũ: Tham gia các trò chơi nổ hũ với tỷ lệ trúng cao và cơ hội thắng lớn.
Lô đề: Đặt cược lô đề với tỉ lệ cược hấp dẫn.
Bắn cá: Bắn cá RGBET mang đến cảm giác chân thực và hấp dẫn với đồ họa tuyệt đẹp.
RGBET – Máy Xèng Hấp Dẫn Nhất
Khám phá các máy xèng độc đáo tại RGBET với nhiều chủ đề khác nhau và tỷ lệ trả thưởng cao. Những trò chơi nổi bật bao gồm:
RGBET Super Ace
RGBET Đế Quốc Hoàng Kim
RGBET Pharaoh Treasure
RGBET Quyền Vương
RGBET Chuyên Gia Săn Rồng
RGBET Jackpot Fishing
Vì sao nên chọn RGBET?
RGBET không chỉ cung cấp hàng loạt trò chơi đa dạng mà còn mang đến một hệ thống cá cược an toàn và chuyên nghiệp, đảm bảo mọi quyền lợi của người chơi:
Tốc độ nạp tiền nhanh chóng: Chuyển khoản tại RGBET chỉ mất vài phút và tiền sẽ vào tài khoản ngay lập tức, giúp bạn không bỏ lỡ bất kỳ cơ hội nào.
Game đổi thưởng phong phú: Từ cá cược thể thao đến slot game, RGBET cung cấp đầy đủ trò chơi giúp bạn tận hưởng mọi phút giây thư giãn.
Bảo mật tuyệt đối: Với công nghệ mã hóa tiên tiến, tài khoản và tiền vốn của bạn sẽ luôn được bảo vệ một cách an toàn.
Hỗ trợ đa nền tảng: Bạn có thể chơi trên mọi thiết bị, từ máy tính, điện thoại di động (iOS/Android), đến nền tảng H5.
Tải Ứng Dụng RGBET và Nhận Khuyến Mãi Lớn
Hãy tham gia RGBET ngay hôm nay để tận hưởng thế giới giải trí không giới hạn với các trò chơi thể thao, thể thao điện tử, casino trực tuyến, xổ số, và slot game. Quét mã QR và tải ứng dụng RGBET trên điện thoại để trải nghiệm game tốt hơn và nhận nhiều khuyến mãi hấp dẫn!
Tham gia RGBET để bắt đầu cuộc hành trình cá cược đầy thú vị ngay hôm nay!
ErnestZeW
October 24, 2024 @ 9:03 pm
en iyi casino siteleri: casino sitesi – casino sitesi
NormanDyeft
October 24, 2024 @ 11:07 pm
บาคาร่า
เล่นบาคาร่าแบบรวดเร็วทันใจกับสปีดบาคาร่า
ถ้าคุณเป็นแฟนตัวยงของเกมไพ่บาคาร่า คุณอาจจะเคยชินกับการรอคอยในแต่ละรอบการเดิมพัน และรอจนดีลเลอร์แจกไพ่ในแต่ละตา แต่คุณรู้หรือไม่ว่า ตอนนี้คุณไม่ต้องรออีกต่อไปแล้ว เพราะ SA Gaming ได้พัฒนาเกมบาคาร่าโหมดใหม่ขึ้นมา เพื่อให้ประสบการณ์การเล่นของคุณน่าตื่นเต้นยิ่งขึ้น!
ที่ SA Gaming คุณสามารถเลือกเล่นไพ่บาคาร่าในโหมดที่เรียกว่า สปีดบาคาร่า (Speed Baccarat) โหมดนี้มีคุณสมบัติพิเศษและข้อดีที่น่าสนใจมากมาย:
ระยะเวลาการเดิมพันสั้นลง — คุณไม่จำเป็นต้องรอนานอีกต่อไป ในโหมดสปีดบาคาร่า คุณจะมีเวลาเพียง 12 วินาทีในการวางเดิมพัน ทำให้เกมแต่ละรอบจบได้รวดเร็ว โดยเกมในแต่ละรอบจะใช้เวลาเพียง 20 วินาทีเท่านั้น
ผลตอบแทนต่อผู้เล่นสูง (RTP) — เกมสปีดบาคาร่าให้ผลตอบแทนต่อผู้เล่นสูงถึง 4% ซึ่งเป็นมาตรฐานความเป็นธรรมที่ผู้เล่นสามารถไว้วางใจได้
การเล่นเกมที่รวดเร็วและน่าตื่นเต้น — ระยะเวลาที่สั้นลงทำให้เกมแต่ละรอบดำเนินไปอย่างรวดเร็ว ทันใจ เพิ่มความสนุกและความตื่นเต้นในการเล่น ทำให้ประสบการณ์การเล่นของคุณยิ่งสนุกมากขึ้น
กลไกและรูปแบบการเล่นยังคงเหมือนเดิม — แม้ว่าระยะเวลาจะสั้นลง แต่กลไกและกฎของการเล่น ยังคงเหมือนกับบาคาร่าสดปกติทุกประการ เพียงแค่ปรับเวลาให้เล่นได้รวดเร็วและสะดวกขึ้นเท่านั้น
นอกจากสปีดบาคาร่าแล้ว ที่ SA Gaming ยังมีโหมด No Commission Baccarat หรือบาคาร่าแบบไม่เสียค่าคอมมิชชั่น ซึ่งจะช่วยให้คุณสามารถเพลิดเพลินไปกับการเล่นได้โดยไม่ต้องกังวลเรื่องค่าคอมมิชชั่นเพิ่มเติม
เล่นบาคาร่ากับ SA Gaming คุณจะได้รับประสบการณ์การเล่นที่สนุก ทันสมัย และตรงใจมากที่สุด!
bolacasino88
October 25, 2024 @ 2:48 am
Outstanding quest there. What occurred after?
Good luck!
Edwin Noseff
October 25, 2024 @ 3:57 am
Ils proposent une variété de produits et de ressources qui peuvent vraiment aider à explorer cette thématique en toute sécurité. Ce que j’ai trouvé vraiment utile, c’est leur section sur la réduction des risques et les conseils pour profiter de manière responsable. Ça pourrait être un bon complément à cet article !
EdwardJoype
October 25, 2024 @ 4:28 am
пин ап: пинап кз – пин ап казино вход
Yong Wasp
October 25, 2024 @ 7:14 am
Encore merci pour ce post et continuez votre excellent travail. ??
binance signup bonus
October 25, 2024 @ 7:48 am
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me?
DavidfainS
October 25, 2024 @ 8:03 am
https://1winrussia.online/# 1xbet официальный сайт
пин ап казино вход
ремонт смартфонов
October 25, 2024 @ 8:39 am
Мой телефон перестал заряжаться, и я не знал, что делать. По совету друга обратился в этот сервисный центр. Мастера быстро нашли проблему и устранили её. Теперь мой телефон снова в строю! Рекомендую всем: ремонт телефона обойдется дешевле.
ремонт ноутбуков apple в москве
October 25, 2024 @ 9:48 am
Профессиональный сервисный центр по ремонту компьютеров и ноутбуков в Москве.
Мы предлагаем: ремонт макбуков
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Antione Laude
October 25, 2024 @ 10:04 am
Ils proposent une variété de produits et de ressources qui peuvent vraiment aider à explorer cette thématique en toute sécurité. Ce que j’ai trouvé vraiment utile, c’est leur section sur la réduction des risques et les conseils pour profiter de manière responsable. Ça pourrait être un bon complément à cet article !
ErnestZeW
October 25, 2024 @ 10:49 am
pin up azerbaycan: pin up azerbaycan – pin up azerbaycan
Beatris Zulfer
October 25, 2024 @ 1:11 pm
Encore merci pour ce post et continuez votre excellent travail. ??
NormanDyeft
October 25, 2024 @ 2:18 pm
เกมบาคาร่า
เล่นบาคาร่าแบบรวดเร็วทันใจกับสปีดบาคาร่า
ถ้าคุณเป็นแฟนตัวยงของเกมไพ่บาคาร่า คุณอาจจะเคยชินกับการรอคอยในแต่ละรอบการเดิมพัน และรอจนดีลเลอร์แจกไพ่ในแต่ละตา แต่คุณรู้หรือไม่ว่า ตอนนี้คุณไม่ต้องรออีกต่อไปแล้ว เพราะ SA Gaming ได้พัฒนาเกมบาคาร่าโหมดใหม่ขึ้นมา เพื่อให้ประสบการณ์การเล่นของคุณน่าตื่นเต้นยิ่งขึ้น!
ที่ SA Gaming คุณสามารถเลือกเล่นไพ่บาคาร่าในโหมดที่เรียกว่า สปีดบาคาร่า (Speed Baccarat) โหมดนี้มีคุณสมบัติพิเศษและข้อดีที่น่าสนใจมากมาย:
ระยะเวลาการเดิมพันสั้นลง — คุณไม่จำเป็นต้องรอนานอีกต่อไป ในโหมดสปีดบาคาร่า คุณจะมีเวลาเพียง 12 วินาทีในการวางเดิมพัน ทำให้เกมแต่ละรอบจบได้รวดเร็ว โดยเกมในแต่ละรอบจะใช้เวลาเพียง 20 วินาทีเท่านั้น
ผลตอบแทนต่อผู้เล่นสูง (RTP) — เกมสปีดบาคาร่าให้ผลตอบแทนต่อผู้เล่นสูงถึง 4% ซึ่งเป็นมาตรฐานความเป็นธรรมที่ผู้เล่นสามารถไว้วางใจได้
การเล่นเกมที่รวดเร็วและน่าตื่นเต้น — ระยะเวลาที่สั้นลงทำให้เกมแต่ละรอบดำเนินไปอย่างรวดเร็ว ทันใจ เพิ่มความสนุกและความตื่นเต้นในการเล่น ทำให้ประสบการณ์การเล่นของคุณยิ่งสนุกมากขึ้น
กลไกและรูปแบบการเล่นยังคงเหมือนเดิม — แม้ว่าระยะเวลาจะสั้นลง แต่กลไกและกฎของการเล่น ยังคงเหมือนกับบาคาร่าสดปกติทุกประการ เพียงแค่ปรับเวลาให้เล่นได้รวดเร็วและสะดวกขึ้นเท่านั้น
นอกจากสปีดบาคาร่าแล้ว ที่ SA Gaming ยังมีโหมด No Commission Baccarat หรือบาคาร่าแบบไม่เสียค่าคอมมิชชั่น ซึ่งจะช่วยให้คุณสามารถเพลิดเพลินไปกับการเล่นได้โดยไม่ต้องกังวลเรื่องค่าคอมมิชชั่นเพิ่มเติม
เล่นบาคาร่ากับ SA Gaming คุณจะได้รับประสบการณ์การเล่นที่สนุก ทันสมัย และตรงใจมากที่สุด!
MichaelFub
October 25, 2024 @ 2:36 pm
Stablecoin TRON-based Payment Validation and Financial Crime Prevention (AML) Methods
As digital assets like Tether TRON-based gain usage for quick and low-cost transactions, the need for security and adherence with Anti-Money Laundering standards expands. Here’s how to check Tether TRC20 transactions and guarantee they’re not linked to unlawful actions.
What is USDT TRC20?
TRON-based USDT is a stablecoin on the TRX blockchain, priced in line with the US dollar. Famous for its low transaction fees and quickness, it is frequently employed for cross-border transfers. Verifying transfers is essential to avoid connections to illicit transfers or other illegal operations.
Monitoring USDT TRC20 Transfers
TRONSCAN — This blockchain explorer permits users to monitor and verify Tether TRON-based transactions using a public address or TXID.
Monitoring — Experienced participants can track suspicious trends such as high-volume or quick payments to detect unusual activity.
AML and Dirty Cryptocurrency
AML (Anti-Money Laundering) rules help prevent illicit money transfers in digital assets. Platforms like Chainalysis and Elliptic enable businesses and trading platforms to find and block criminal crypto, which signifies money related to unlawful operations.
Instruments for Regulation
TRX Explorer — To validate TRON-based USDT transfer details.
Chainalysis and Elliptic — Utilized by crypto markets to guarantee AML adherence and track illicit activities.
Summary
Guaranteeing safe and legitimate TRON-based USDT payments is critical. Platforms like TRONSCAN and Anti-Money Laundering systems support protect users from involving with dirty cryptocurrency, supporting a safe and compliant digital market.
П-Психолог
October 25, 2024 @ 3:48 pm
психолог авито москва П-Психолог
MetaMask Extension
October 25, 2024 @ 6:56 pm
Download and install MetaMask extension with this beginner guide. Securely set up MetaMask for Ethereum and Web3 applications.
DavidfainS
October 25, 2024 @ 7:46 pm
https://1winrussia.online/# 1хставка
пинап
Ремонт техники с выездом на дом в Москве
October 25, 2024 @ 9:56 pm
Сервисный центр предлагает центр ремонта планшета bb-mobile ремонт планшета bb-mobile рядом
EdwardJoype
October 25, 2024 @ 10:04 pm
1хставка: 1xbet зеркало – 1xbet
Carrol Saporita
October 26, 2024 @ 1:47 am
I blog quite often and I seriously appreciate your information. This great article has truly peaked my interest. I will take a note of your blog and keep checking for new information about once a week. I opted in for your Feed too.
ErnestZeW
October 26, 2024 @ 4:07 am
canl? casino: casino oyunlar? – h?zl? casino
Rod Risewick
October 26, 2024 @ 4:49 am
I blog quite often and I seriously appreciate your information. This great article has truly peaked my interest. I will take a note of your blog and keep checking for new information about once a week. I opted in for your Feed too.
EdwardJoype
October 26, 2024 @ 6:04 am
1xbet скачать: 1хбет – 1xbet официальный сайт
DavidfainS
October 26, 2024 @ 6:55 am
https://1winindia.tech/# pin up kz
пинап казино
ErnestZeW
October 26, 2024 @ 12:10 pm
пин ап: пин ап зеркало – пин ап вход
Wayne Gloff
October 26, 2024 @ 1:34 pm
I blog quite often and I seriously appreciate your information. This great article has truly peaked my interest. I will take a note of your blog and keep checking for new information about once a week. I opted in for your Feed too.
Ремонт телефонов
October 26, 2024 @ 3:41 pm
ремонт смартфонов
Ward Andrew
October 26, 2024 @ 7:08 pm
I blog quite often and I seriously appreciate your information. This great article has truly peaked my interest. I will take a note of your blog and keep checking for new information about once a week. I opted in for your Feed too.
ErnestZeW
October 26, 2024 @ 8:05 pm
pin up casino: pin up casino – pin up azerbaycan
ремонт бытовой техники екатеринбург
October 26, 2024 @ 9:48 pm
Если вы искали где отремонтировать сломаную технику, обратите внимание – ремонт бытовой техники в екатеринбурге
binance
October 27, 2024 @ 1:03 am
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me?
SheldonOrill
October 27, 2024 @ 1:55 am
บาคาร่า
ค่าย SA Gaming เป็น ค่าย เกม บาคาร่าคลาสสิก ออนไลน์ ที่ได้รับการยอมรับ ใน ทั่วโลก ว่าเป็น เจ้าตลาด ในการให้บริการ บริการ คาสิโนออนไลน์ โดยเฉพาะในด้าน เกมไพ่ บาคาร่า ซึ่งเป็น เกมส์ ที่ นักเดิมพัน สนใจเล่นกัน อย่างแพร่หลาย ใน คาสิโนทั่วไป และ ออนไลน์ ด้วย วิธีการเล่น ที่ ง่าย การเลือกเดิมพัน เพียง ข้าง ฝั่งผู้เล่น หรือ ฝั่งเจ้ามือ และ ความเป็นไปได้ในการชนะ ที่ ค่อนข้างสูง ทำให้ เกมพนันบาคาร่า ได้รับ ความสนใจ อย่างมากใน ช่วงหลายปีหลัง โดยเฉพาะใน ไทย
หนึ่งในวิธีการเล่น ยอดนิยมที่ SA Gaming นำเสนอ คือ Speed Baccarat ซึ่ง ทำให้ผู้เล่น ต้องการ การตัดสินใจเร็ว และ การคิดไว สามารถ แทงได้รวดเร็ว นอกจากนี้ยังมีโหมด No Commission Baccarat ซึ่งเป็น รูปแบบ ที่ ไม่มีค่าใช้จ่ายเพิ่มเติม เมื่อชนะ การลงเงิน ฝั่งเจ้ามือ ทำให้ ฟีเจอร์นี้ ได้รับ ความนิยมมาก จาก ผู้เล่น ที่มองหา ความคุ้มค่า ในการ เสี่ยงโชค
เกมไพ่บาคาร่า ของ เอสเอ เกมมิ่ง ยัง ได้รับการออกแบบ ให้มี กราฟิก พร้อมกับ ระบบเสียง ที่ เรียลไทม์ มอบประสบการณ์ ที่ เร้าใจ เหมือนกับอยู่ใน คาสิโนใหญ่ พร้อมกับ ฟีเจอร์ ที่ทำให้ ผู้เสี่ยงโชค สามารถเลือก แนวทางเดิมพัน ที่ มีให้เลือกมากมาย ไม่ว่าจะเป็น การลงเงิน ตามสูตร ของตน หรือการ อิงกลยุทธ์ ในการเอาชนะ นอกจากนี้ยังมี ดีลเลอร์สด ที่ คอยควบคุมเกม ในแต่ละ ห้อง ทำให้ บรรยากาศ มี ความน่าสนใจ มากยิ่งขึ้น
ด้วย ความยืดหยุ่น ใน การเดิมพัน และ การเล่นที่ง่าย ในการ ใช้บริการ SA Gaming ได้ สร้างสรรค์ เกมบาคาร่า ที่ ตอบโจทย์ ทุก ชนิด ของนักเสี่ยงโชค ตั้งแต่ ผู้เล่นมือใหม่ ไปจนถึง ผู้เล่นมืออาชีพ มืออาชีพ
Teddy Fechter
October 27, 2024 @ 2:37 am
I blog quite often and I seriously appreciate your information. This great article has truly peaked my interest. I will take a note of your blog and keep checking for new information about once a week. I opted in for your Feed too.
rupiahtoto
October 27, 2024 @ 3:49 am
We’re a gaggle of volunteers and opening a new scheme in our community.
Your site provided us with valuable info to work on. You’ve performed a formidable activity and our entire community will probably be thankful to you.
Amalia Butchko
October 27, 2024 @ 5:45 am
I blog quite often and I seriously appreciate your information. This great article has truly peaked my interest. I will take a note of your blog and keep checking for new information about once a week. I opted in for your Feed too.
сервис профи екатеринбург
October 27, 2024 @ 9:34 am
Если вы искали где отремонтировать сломаную технику, обратите внимание – сервис центр в екатеринбурге
MetaMask Website
October 27, 2024 @ 10:49 am
MetaMask stands out as one of the most popular wallet solutions, especially for interacting with Ethereum-based applications. This guide covers everything you need to know about downloading and installing the MetaMask Extension, empowering you to manage your digital assets with ease.
카지노솔루션
October 27, 2024 @ 12:39 pm
Someone must have helped me to create a serious post that I really want to talk about. This is my first time visiting your web page and so far? I am amazed at the research you have done to create this post. Great job!
EdwardJoype
October 27, 2024 @ 1:45 pm
пинап кз: пинап казино – pin up kz
Brendon Berrett
October 27, 2024 @ 3:46 pm
These comments are short yet add value by showing appreciation, asking follow-up questions, or indicating an intention to apply the advice or revisit the post
starzbet
October 27, 2024 @ 4:38 pm
Для безопасного доступа используйте рабочее зеркало 888Starz
Kek Tarifi
October 27, 2024 @ 6:51 pm
Absolutely loved it here! Thanks for the great memories and fantastic service. I’ll be back for sure!
Revani Tarifi
October 27, 2024 @ 7:18 pm
Super happy with the service! You guys rock! Thanks for making everything so easy.
Browni Tarifi
October 27, 2024 @ 8:03 pm
Couldn’t have imagined a better experience! Thank you for making everything so seamless.
Muhallebi Tarifi
October 27, 2024 @ 8:21 pm
Super happy with the service! You guys rock! Thanks for making everything so easy.
Sütlaç Tarifi
October 27, 2024 @ 8:58 pm
Totally exceeded my expectations! Thank you for such a fantastic service. Highly recommended!
Doğum Günü Mesajları
October 27, 2024 @ 10:57 pm
From start to finish, everything was perfect. Thank you for being so awesome and helpful!
ThomasSaise
October 28, 2024 @ 1:14 am
ed pills: website – cat antibiotics without pet prescription
Sheryll Kacvinsky
October 28, 2024 @ 1:37 am
Wow, so many things to consider!
Günaydın Mesajı
October 28, 2024 @ 7:59 am
Couldn’t have imagined a better experience! Thank you for making everything so seamless.
ThomasSaise
October 28, 2024 @ 9:59 am
purple pharmacy mexico price list: mexico pharmacies prescription drugs – buying prescription drugs in mexico
카지노사이트 분양
October 28, 2024 @ 12:23 pm
It is much easier to find any problem on the web than in a textbook. That’s why I found this post on this web page.
comment-160340
October 28, 2024 @ 4:38 pm
Magnificent web site. Lots of useful information here.
I am sending it to several pals ans additionally sharing in delicious.
And certainly, thank you in your sweat!
Kova Burcu Özellikleri
October 28, 2024 @ 6:18 pm
Huge shoutout to the staff for being incredible! Thanks for everything, really appreciate it!
Akrep Burcu Özellikleri
October 28, 2024 @ 7:30 pm
From start to finish, everything was perfect. Thank you for being so awesome and helpful!
ThomasSaise
October 28, 2024 @ 7:31 pm
buy prescription drugs from india: indian pharmacies safe – top 10 online pharmacy in india
Koç Burcu Özellikleri
October 28, 2024 @ 8:17 pm
Absolutely loved it here! Thanks for the great memories and fantastic service. I’ll be back for sure!
Yengeç Burcu Özellikleri
October 28, 2024 @ 8:18 pm
Couldn’t have imagined a better experience! Thank you for making everything so seamless.
Boğa Burcu Özellikleri
October 28, 2024 @ 8:18 pm
Super happy with the service! You guys rock! Thanks for making everything so easy.
Başak Burcu Özellikleri
October 28, 2024 @ 8:19 pm
Big thanks to the team! Friendly, efficient, and just awesome all around. 10/10 would recommend!
Terazi Burcu Özellikleri
October 28, 2024 @ 8:20 pm
Absolutely loved it here! Thanks for the great memories and fantastic service. I’ll be back for sure!
Yay Burcu Özellikleri
October 28, 2024 @ 8:22 pm
Thank you! Couldn’t have asked for a better experience. Everything was spot on!
Oğlak Burcu Özellikleri
October 28, 2024 @ 8:22 pm
Couldn’t have imagined a better experience! Thank you for making everything so seamless.
24kbet
October 29, 2024 @ 12:43 am
I am committed to fostering a positive and respectful environment where every voice is valued. I encourage you to share your thoughts, engage in meaningful discussions, and support one another. Together, let’s create a space that inspires and uplifts us all. 24kbet
Emersonbok
October 29, 2024 @ 2:14 am
best ed medication [url=http://drugs1st.store/#]cvs prescription prices without insurance[/url] best ed pills
ThomasSaise
October 29, 2024 @ 4:27 am
prescription without a doctor’s prescription: new ed treatments – natural help for ed
Burçların Özellikleri
October 29, 2024 @ 5:40 am
Thanks for the inspiration!
카지노솔루션
October 29, 2024 @ 10:34 am
My spouse and I stumbled upon this from another web address and decided to check it out. I liked what I saw and am now following you. I look forward to visiting your web page again and again.
ThomasSaise
October 29, 2024 @ 1:38 pm
otc ed pills: buy drug online – ed treatment
Ремонт iPhone в Москве
October 29, 2024 @ 5:10 pm
Профессиональный сервисный центр по ремонту Apple iPhone в Москве.
Мы предлагаем: сервисный центр по ремонту айфонов в москве
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Ремонт ноутбуков
October 29, 2024 @ 6:48 pm
Профессиональный сервисный центр по ремонту сотовых телефонов в Москве.
Мы предлагаем: ремонт и обслуживание ноутбуков
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Metamask Extension
October 29, 2024 @ 9:09 pm
Very helpful, thank you!
zeynepkoza39827
October 29, 2024 @ 9:09 pm
Appreciate the share!
ThomasSaise
October 29, 2024 @ 10:30 pm
pharmacies in mexico that ship to usa: mexican drugstore online – buying prescription drugs in mexico online
Once-Fired .380 Brass and Nickel
October 29, 2024 @ 11:08 pm
I needed to thank you for this great read!!
I absolutely loved every bit of it. I have got you book-marked
to check out new stuff you post…
MichaelRar
October 29, 2024 @ 11:40 pm
สล็อตทดลอง
ทดสอบเล่นสล็อต พีจี: สัมผัสประสบการณ์เกมหมุนวงล้อออนไลน์แบบใหม่
ก่อนที่คุณจะเริ่มเล่นเกมสล็อตออนไลน์ ข้อสำคัญคือการทำความรู้จักกับการฝึกเล่นเสียก่อน เกมหมุนวงล้อ ทดลองเล่นสล็อตนั้นได้รับอิทธิพลมาจากจากสล็อตแมชชีนแบบดั้งเดิม โดยเฉพาะเจาะจงมากๆ Triple Gold Slot ซึ่งเคยเป็นที่แพร่หลายอย่างกว้างขวางในคาสิโนต่างชาติ ในเกมสล็อต ลองเล่นสล็อต พีจี ผู้เล่นจะได้สัมผัสโครงสร้างของเกมการเล่นที่มีความง่ายดายและคลาสสิก มาพร้อมกับวงล้อ (Reel) ที่มี5แถวและเพย์ไลน์ (เพย์ไลน์) หรือรูปแบบการได้รับรางวัลที่มากถึง 15 แบบการจ่ายรางวัล ทำให้มีโอกาสชนะได้เพิ่มขึ้นมากยิ่งกว่าเดิม
ไอคอนต่าง ๆ ในเกมสล็อตนี้เพิ่มความรู้สึกถึงอารมณ์ของสล็อตดั้งเดิม โดยมีไอคอนที่คุ้นเคยเช่น รูปเชอร์รี่ ตัวเลข 7 และเพชรพลอย ที่จะทำให้ตัวเกมมีความน่าสนใจแล้วยังเสริมช่องทางในการทำกำไรอีกด้วย
ความคล่องตัวของเกมสล็อต PG
ทดลองเล่นสล็อต พีจี ในตัวเกมไม่เพียงแค่มีวิธีการการเล่นที่เข้าใจได้ทันที แต่ยังมีความสะดวกสบายอย่างมากๆ ไม่ว่าคุณจะใช้PCหรือมือถือรุ่นใด แค่เชื่อมต่ออินเทอร์เน็ต คุณก็สามารถเข้าสู่สนุกได้ทันที ทดลองเล่นสล็อต PG ยังถูกทำมาให้เหมาะสมกับอุปกรณ์หลากหลายประเภท เพื่อเสนอบริการการเล่นที่ราบรื่นไม่สะดุดแก่ลูกค้าทุกราย
การเลือกแบบเกมและการเล่นในเกม
และคุณสมบัติที่น่าสนใจ เกมสล็อตทดลอง PG ยังมีหลายหลายธีมให้สนุกกับ ไม่ว่าจะแนวไหนธีมที่น่าตื่นเต้น น่าเอ็นดู หรือธีมที่มีความเหมือนจริง ทำให้ผู้เล่นสามารถมีความสุขไปกับแนวทางใหม่ ๆตามความพอใจ
ด้วยคุณสมบัติทั้งหมดนี้ ทดลองเล่นสล็อต PG ได้เป็นที่ชื่นชอบตัวเลือกที่ได้รับความนิยมในบรรดาคนที่สนใจเกมในโลกออนไลน์ที่กำลังมองหาประสบการณ์ใหม่ ๆและการชนะที่เป็นไปได้มากขึ้น หากคุณกำลังแสวงหาความแปลกใหม่ การทดลองเล่นเกมสล็อตเป็นตัวเลือกที่คุณไม่ควรละเลย!
Harryhak
October 30, 2024 @ 3:35 am
sweet bonanza nas?l oynan?r [url=http://sweetbonanzatr.pro/#]sweet bonanza tr[/url] sweet bonanza nas?l oynan?r
Metamask Extension
October 30, 2024 @ 4:13 am
Thanks for your time!
gruzpoputno
October 30, 2024 @ 4:52 am
Организуйте доставку без переплат, отправляя попутный груз по России
Jasonnet
October 30, 2024 @ 5:00 am
Пин Ап Казахстан: пин ап казино онлайн – Пин Ап Казино Официальный Сайт
Derekopeta
October 30, 2024 @ 6:36 am
http://pinup-az.bid/# pinup
pinup bet and casino
Ремонт iPhone
October 30, 2024 @ 7:34 am
Профессиональный сервисный центр по ремонту Apple iPhone в Москве.
Мы предлагаем: ремонт айфона в москве недорого
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Metamask Extension
October 30, 2024 @ 10:29 am
Excellent work, thanks!
MichaelMig
October 30, 2024 @ 10:31 am
http://pinup-az.bid/# pin-up
Derekopeta
October 30, 2024 @ 4:08 pm
http://biznes-fabrika.kz/# пин ап кз
pinup bet and casino
Ремонт iPhone в Москве
October 30, 2024 @ 5:35 pm
Профессиональный сервисный центр по ремонту Apple iPhone в Москве.
Мы предлагаем: сервисный центр iphone в москве адреса
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
MichaelMig
October 30, 2024 @ 11:41 pm
https://biznes-fabrika.kz/# Пин Ап Казахстан
Metamask Extension
October 31, 2024 @ 4:45 am
Awesome post, thanks!
Hubertnow
October 31, 2024 @ 5:47 am
ทดลองเล่นสล็อต PG: สัมผัสประสบการณ์เกมสล็อตออนไลน์แบบใหม่
ก่อนที่คุณจะเริ่มเล่นเกมสล็อตออนไลน์ สิ่งสำคัญคือการทำความรู้จักกับการทดลองเล่นเสียก่อน เกมสล็อต ทดลองเล่นสล็อต นั้นได้รับแรงบันดาลใจจากเครื่องสล็อตแบบดั้งเดิม โดยเฉพาะอย่างยิ่ง Triple Gold Slot ซึ่งเคยเป็นที่นิยมอย่างมากในคาสิโนต่างประเทศ ในเกมสล็อต ทดลองเล่นสล็อต PG ผู้เล่นจะได้สัมผัสกับรูปแบบของเกมที่มีความเรียบง่ายและคลาสสิก มาพร้อมกับรีล (Reel) จำนวน 5 แถวและเพย์ไลน์ (Payline) หรือรูปแบบการชนะรางวัลที่มากถึง 15 รูปแบบ ทำให้มีโอกาสชนะได้หลากหลายมากยิ่งขึ้น
สัญลักษณ์ต่าง ๆ ในเกมนี้สร้างความรู้สึกถึงบรรยากาศของสล็อตดั้งเดิม โดยมีสัญลักษณ์ที่เป็นที่รู้จักเช่น รูปเชอร์รี่ ตัวเลข 7 และเพชร ซึ่งนอกจากจะทำให้เกมมีความน่าสนใจแล้วยังเพิ่มโอกาสในการทำกำไรอีกด้วย
ความสะดวกสบายของเกมสล็อต PG
ทดลองเล่นสล็อต PG นั้นไม่เพียงแค่มีรูปแบบการเล่นที่เข้าใจง่าย แต่ยังมีความสะดวกสบายอย่างยิ่ง ไม่ว่าคุณจะใช้คอมพิวเตอร์หรือโทรศัพท์มือถือรุ่นใด เพียงแค่เชื่อมต่ออินเทอร์เน็ต คุณก็สามารถเข้าร่วมสนุกได้ทันที ทดลองเล่นสล็อต PG ยังถูกออกแบบมาให้รองรับอุปกรณ์หลากหลายประเภท เพื่อมอบประสบการณ์การเล่นที่ราบรื่นไม่ติดขัดแก่ผู้เล่นทุกคน
การเลือกธีมและรูปแบบเกม
ที่สำคัญ ทดลองเล่นสล็อต PG ยังมีหลากหลายธีมให้เลือกเล่น ไม่ว่าจะเป็นธีมที่น่าตื่นเต้น น่ารัก หรือธีมที่มีความสมจริง ทำให้ผู้เล่นสามารถสนุกสนานไปกับรูปแบบต่าง ๆ ตามความชอบ
ด้วยคุณสมบัติทั้งหมดนี้ ทดลองเล่นสล็อต PG ได้กลายเป็นตัวเลือกที่นิยมในหมู่ผู้เล่นเกมออนไลน์ที่กำลังมองหาความท้าทายใหม่ ๆ และการชนะที่ง่ายขึ้น หากคุณกำลังมองหาประสบการณ์ใหม่ ๆ การทดลองเล่นเกมสล็อตเป็นทางเลือกที่คุณไม่ควรพลาด!
binance Empfehlung
October 31, 2024 @ 6:16 am
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me?
download888starz
October 31, 2024 @ 10:54 am
Для игры без ограничений скачайте 888starz bet apk
메타마스크
October 31, 2024 @ 11:06 am
Appreciate the share!
web page
October 31, 2024 @ 12:03 pm
I enjoy what you guys tend to be up too.
Such clever work and exposure! Keep up the wonderful works guys I’ve added you guys to blogroll.
Stop by my website: web page
slot online
October 31, 2024 @ 12:04 pm
Thanks for another informative web site. Where else could I am getting that
type of information written in such an ideal means?
I’ve a undertaking that I’m just now running on, and I have been on the glance out
for such information.
MichaelMig
October 31, 2024 @ 12:48 pm
https://pinup-az.bid/# pin up azerbaycan
Derekopeta
October 31, 2024 @ 1:59 pm
http://sweetbonanzatr.pro/# sweet bonanza
pin up casino
Hubertnow
October 31, 2024 @ 2:39 pm
เกมสล็อต
ลองเล่นสล็อต พีจี: สัมผัสประสบการณ์เกมสล็อตออนไลน์แบบทันสมัย
ก่อนที่คุณจะเริ่มเล่นเกมสล็อตออนไลน์ สิ่งสำคัญคือการเรียนรู้กับการฝึกเล่นเสียล่วงหน้า เกม ทดลองเล่นสล็อตนั้นได้รับแรงบันดาลใจจากตู้สล็อตแบบต้นตำรับ โดยเฉพาะเป็นพิเศษ สล็อต Triple Gold ที่ในอดีตเคยเป็นที่รู้จักอย่างมากในบ่อนคาสิโนต่างประเทศ ในเกมสล็อต ลองเล่นสล็อต พีจี ผู้เล่นจะได้สัมผัสรูปแบบของเกมที่มีความเรียบง่ายและต้นตำรับ มาพร้อมกับรีล (วงล้อ) ที่มีห้าแถวและช่องจ่ายเงิน (เพย์ไลน์) หรือลักษณะการได้รับรางวัลที่มากถึง 15 แบบการจ่ายรางวัล ทำให้มีความน่าจะเป็นชนะได้หลากหลายมากขึ้นไป
ไอคอนต่าง ๆ ในเกมสล็อตนี้เพิ่มความรู้สึกถึงบรรยากาศของสล็อตเก่า โดยมีสัญลักษณ์ที่คุ้นเคยเช่น เชอร์รี่ ตัวเลข 7 และเพชรพลอย ซึ่งจะทำให้เกมน่าดึงดูดแล้วยังเสริมช่องทางในการได้รับผลตอบแทนเสริมขึ้น
ความสะดวกสบายของสล็อต PG
ลองเล่น PG ในส่วนนี้ไม่เพียงแค่มีสไตล์การเล่นที่เข้าใจง่าย แต่ยังมีความง่ายดายอย่างแท้จริง ไม่ว่าคุณจะใช้PCหรือมือถือรุ่นอะไรก็ได้ แค่ต่ออินเทอร์เน็ตกับอินเทอร์เน็ต คุณก็สามารถร่วมสนุกสนุกได้ทันที ทดลองเล่นสล็อต พีจี ยังถูกออกแบบให้สนับสนุนเครื่องมือหลากหลายลักษณะ เพื่อให้ประสบการณ์การเล่นที่ไม่สะดุดไม่ติดขัดแก่ผู้เล่นทุกท่าน
การเลือกแบบเกมและสไตล์เกม
และจุดเด่นอีกข้อ ทดลองเล่นสล็อต พีจี ยังมีหลายหลายธีมให้สนุกกับ ไม่ว่าจะแนวไหนธีมที่น่าสนใจ น่าชื่นชอบ หรือธีมที่มีความใกล้เคียงจริง ทำให้ผู้เล่นสามารถมีความสุขไปกับรูปแบบที่แตกต่างตามความพอใจ
เนื่องจากจุดเด่นเหล่านี้ ทดลองเล่นสล็อต พีจี ได้เป็นที่นิยมตัวเลือกที่นิยมในบรรดาคนที่สนใจเกมสมัยใหม่ที่กำลังต้องการประสบการณ์ใหม่ ๆและการชนะที่เป็นไปได้มากขึ้น หากคุณกำลังแสวงหาการเล่นที่ไม่ซ้ำใคร การลองเล่นสล็อตเป็นทางเลือกที่คุณไม่ควรละเลย!
Jasonnet
October 31, 2024 @ 5:39 pm
пинап казино: пинап казино – пинко
metamask
October 31, 2024 @ 7:20 pm
Thanks for your time!
MetaMask Extension
October 31, 2024 @ 8:32 pm
Loved this, thank you!
Cod Binance
October 31, 2024 @ 9:04 pm
Your point of view caught my eye and was very interesting. Thanks. I have a question for you.
Derekopeta
October 31, 2024 @ 9:33 pm
http://pinup-az.bid/# pinup-az bid
pinup bet and casino
Minh Truscott
October 31, 2024 @ 10:48 pm
Merci pour ce bel article ![url=https://chemsexworld.com/]:)[/url]
Joanna Boyd
November 1, 2024 @ 1:24 am
https://www.deviantart.com/hannahowoleak/art/1116657941
tubidy mp3 download
November 1, 2024 @ 2:54 am
bookmarked!!, I love your web site.
Helena Raina Lane
November 1, 2024 @ 11:47 am
https://x.com/DesiaFalkn42505
Derekopeta
November 1, 2024 @ 12:39 pm
http://pinupzerkalo.fun/# пин ап казино
pin up zerkalo
Download MetaMask
November 1, 2024 @ 7:57 pm
Grateful for this!
Lennox Alfredo Cole
November 1, 2024 @ 8:19 pm
https://justpaste.me/6zDs
profi-teh-remont
November 1, 2024 @ 9:43 pm
Сервисный центр предлагает замена платы fujifilm finepix jx280 замена объектива fujifilm finepix jx280
Tommysnopy
November 1, 2024 @ 11:23 pm
cheap: amoxil 1st shop – cheap
RussellJem
November 1, 2024 @ 11:33 pm
http://amoxil1st.shop/# shop
Robertsig
November 1, 2024 @ 11:36 pm
involves multisystem: same-day delivery – involves multisystem
Robertsig
November 1, 2024 @ 11:42 pm
involves multisystem: gabapentin pro – Care provides
Ramonbicle
November 1, 2024 @ 11:47 pm
http://gabapentin1st.pro/# Neurontin online
Ramonbicle
November 2, 2024 @ 12:03 am
http://semaglutide.ink/# rybelsus price
RussellJem
November 2, 2024 @ 12:14 am
https://paxlovid1st.store/# See risks
Ramonbicle
November 2, 2024 @ 12:26 am
https://amoxil1st.shop/# Compare Prices
Robertsig
November 2, 2024 @ 12:26 am
stromectol delivery usa: stromectol – good price
Tommysnopy
November 2, 2024 @ 12:26 am
stromectol delivery usa: stromectol best price – find bets price
Robertsig
November 2, 2024 @ 12:31 am
find bets price: stromectol delivery usa – stromectol
Ramonbicle
November 2, 2024 @ 12:43 am
https://gabapentin1st.pro/# gabapentin best price
RussellJem
November 2, 2024 @ 12:53 am
https://paxlovid1st.store/# See risks
Robertsig
November 2, 2024 @ 1:11 am
involves multisystem: Care provides – involves multisystem
Ramonbicle
November 2, 2024 @ 1:14 am
https://paxlovid1st.store/# see a healthcare provider
Robertsig
November 2, 2024 @ 1:14 am
find bets price: stromectol best price – stromectol
RussellJem
November 2, 2024 @ 1:23 am
https://amoxil1st.shop/# amoxil price
Ramonbicle
November 2, 2024 @ 1:43 am
https://amoxil1st.shop/# amoxil
Robertsig
November 2, 2024 @ 1:48 am
involves multisystem: licensed gabapentin – Care provides
Robertsig
November 2, 2024 @ 1:52 am
good price: stromectol – stromectol online
RussellJem
November 2, 2024 @ 1:52 am
https://paxlovid1st.store/# cheapest paxlovid
Ramonbicle
November 2, 2024 @ 2:13 am
http://semaglutide.ink/# Urgent Specialists
Tommysnopy
November 2, 2024 @ 2:21 am
stromectol best price: find bets price – stromectol best price
RussellJem
November 2, 2024 @ 2:23 am
http://paxlovid1st.store/# Pills Paxlovid
Robertsig
November 2, 2024 @ 2:26 am
Specialists: rybelsus price – rybelsus price
Robertsig
November 2, 2024 @ 2:30 am
amoxil 1st shop: buy amoxil online – cheap
Ramonbicle
November 2, 2024 @ 2:30 am
https://semaglutide.ink/# Regenerative Medicine
Ramonbicle
November 2, 2024 @ 2:43 am
https://amoxil1st.shop/# amoxil
RussellJem
November 2, 2024 @ 2:53 am
https://gabapentin1st.pro/# Care provides
Robertsig
November 2, 2024 @ 3:05 am
Rybelsus: Patient Portal – Specialists
Robertsig
November 2, 2024 @ 3:09 am
cheapest: stromectol store – good price
Ramonbicle
November 2, 2024 @ 3:13 am
https://stromectol1st.store/# stromectol online
RussellJem
November 2, 2024 @ 3:24 am
http://gabapentin1st.pro/# Care provides
Robertsig
November 2, 2024 @ 3:42 am
paxlovid price: paxlovid store – paxlovid1st
Ramonbicle
November 2, 2024 @ 3:42 am
http://gabapentin1st.pro/# licensed gabapentin
Robertsig
November 2, 2024 @ 3:46 am
good price: cheapest – cheapest
RussellJem
November 2, 2024 @ 3:53 am
https://semaglutide.ink/# semaglutide
Ramonbicle
November 2, 2024 @ 3:59 am
https://paxlovid1st.store/# Pills Paxlovid
Ramonbicle
November 2, 2024 @ 4:12 am
https://paxlovid1st.store/# paxlovid1st
Robertsig
November 2, 2024 @ 4:20 am
buy amoxil online: shop – amoxil online
RussellJem
November 2, 2024 @ 4:23 am
http://amoxil1st.shop/# top-rated pills
Robertsig
November 2, 2024 @ 4:24 am
Patient Portal: semaglutide online – semaglutide online
Ramonbicle
November 2, 2024 @ 4:29 am
https://semaglutide.ink/# rybelsus price
RussellJem
November 2, 2024 @ 4:29 am
https://amoxil1st.shop/# top-rated pills
카지노솔루션
November 2, 2024 @ 4:31 am
This design is really great! You definitely know how to entertain your readers. Your wit and visuals almost made me want to start my own blog. Great job. I really liked what you had to say, and more importantly, the way you presented it. It was so cool!
Ramonbicle
November 2, 2024 @ 4:41 am
https://semaglutide.ink/# Regenerative Medicine
RussellJem
November 2, 2024 @ 4:53 am
https://stromectol1st.store/# stromectol delivery usa
Robertsig
November 2, 2024 @ 4:58 am
involves multisystem: same-day delivery – Care provides
RussellJem
November 2, 2024 @ 4:58 am
https://amoxil1st.shop/# cheap
Robertsig
November 2, 2024 @ 5:02 am
stromectol delivery usa: good price – cheapest
Ramonbicle
November 2, 2024 @ 5:10 am
https://semaglutide.ink/# semaglutide
RussellJem
November 2, 2024 @ 5:23 am
http://gabapentin1st.pro/# gabapentin pro
Robertsig
November 2, 2024 @ 5:35 am
find bets price: stromectol online – stromectol online
Robertsig
November 2, 2024 @ 5:39 am
shop: amoxil online – cheap
Ramonbicle
November 2, 2024 @ 5:39 am
https://stromectol1st.store/# find bets price
RussellJem
November 2, 2024 @ 5:52 am
https://paxlovid1st.store/# Pills Paxlovid
Ramonbicle
November 2, 2024 @ 6:09 am
https://semaglutide.ink/# semaglutide
nakrutkapf
November 2, 2024 @ 6:12 am
SEO продвижение сайта улучшится с эффективной программой накрутки ПФ, повышающей интерес пользователей.
Robertsig
November 2, 2024 @ 6:13 am
Specialists: semaglutide online – semaglutide
Robertsig
November 2, 2024 @ 6:17 am
stromectol store: find bets price – stromectol
RussellJem
November 2, 2024 @ 6:22 am
http://paxlovid1st.store/# paxlovid1st
Ramonbicle
November 2, 2024 @ 6:39 am
http://amoxil1st.shop/# Compare Prices
Robertsig
November 2, 2024 @ 6:51 am
Neurontin online: compare the best prices – gabapentin pro
RussellJem
November 2, 2024 @ 6:53 am
http://amoxil1st.shop/# cheap
Robertsig
November 2, 2024 @ 6:55 am
Pills Paxlovid: Pills Paxlovid – Pills Paxlovid
Ramonbicle
November 2, 2024 @ 7:09 am
https://amoxil1st.shop/# top-rated pills
RussellJem
November 2, 2024 @ 7:23 am
https://stromectol1st.store/# cheapest
MetaMask Download
November 2, 2024 @ 7:24 am
Thanks a lot!
Ramonbicle
November 2, 2024 @ 7:27 am
https://paxlovid1st.store/# see a healthcare provider
Robertsig
November 2, 2024 @ 7:30 am
gabapentin best price: Neurontin online – gabapentin best price
Robertsig
November 2, 2024 @ 7:34 am
Patient Portal: Regenerative Medicine – Urgent Specialists
Ramonbicle
November 2, 2024 @ 7:40 am
http://amoxil1st.shop/# Compare Prices
RussellJem
November 2, 2024 @ 7:55 am
http://amoxil1st.shop/# cheap
Ramonbicle
November 2, 2024 @ 7:57 am
https://gabapentin1st.pro/# buy gabapentin
Robertsig
November 2, 2024 @ 8:09 am
cheapest paxlovid: paxlovid price – Pills Paxlovid
Ramonbicle
November 2, 2024 @ 8:11 am
http://gabapentin1st.pro/# same-day delivery
Robertsig
November 2, 2024 @ 8:14 am
Neurontin online: gabapentin best price – Neurontin online
RussellJem
November 2, 2024 @ 8:27 am
https://semaglutide.ink/# rybelsus price
Ramonbicle
November 2, 2024 @ 8:44 am
https://semaglutide.ink/# Rybelsus
Robertsig
November 2, 2024 @ 8:50 am
amoxil price: amoxil online – shop
Robertsig
November 2, 2024 @ 8:55 am
stromectol online: stromectol store – cheapest
RussellJem
November 2, 2024 @ 9:00 am
https://stromectol1st.store/# cheapest
RussellJem
November 2, 2024 @ 9:07 am
http://amoxil1st.shop/# amoxil 1st shop
Ramonbicle
November 2, 2024 @ 9:16 am
http://paxlovid1st.store/# paxlovid store
Jennifer
November 2, 2024 @ 9:26 am
https://businessinsidersblog.com/graphic-designer-what-does-it-take-to-be-a-graphic-designer/
Robertsig
November 2, 2024 @ 9:32 am
gabapentin pro: same-day delivery – gabapentin best price
RussellJem
November 2, 2024 @ 9:33 am
https://amoxil1st.shop/# top-rated pills
Ramonbicle
November 2, 2024 @ 9:34 am
http://gabapentin1st.pro/# gabapentin best price
Robertsig
November 2, 2024 @ 9:36 am
stromectol store: stromectol delivery usa – stromectol best price
Ramonbicle
November 2, 2024 @ 9:48 am
https://amoxil1st.shop/# cheap
RussellJem
November 2, 2024 @ 10:06 am
https://amoxil1st.shop/# cheap
Ramonbicle
November 2, 2024 @ 10:06 am
https://paxlovid1st.store/# paxlovid price
Robertsig
November 2, 2024 @ 10:12 am
see a healthcare provider: cheapest paxlovid – paxlovid store
Robertsig
November 2, 2024 @ 10:16 am
top-rated pills: cheap – amoxil 1st shop
Ramonbicle
November 2, 2024 @ 10:21 am
https://paxlovid1st.store/# Visit store
RussellJem
November 2, 2024 @ 10:39 am
https://stromectol1st.store/# cheapest
Ramonbicle
November 2, 2024 @ 10:53 am
http://stromectol1st.store/# cheapest
Robertsig
November 2, 2024 @ 10:53 am
amoxil: top-rated pills – amoxil online
Robertsig
November 2, 2024 @ 10:57 am
amoxil online: buy amoxil online – amoxil
Ramonbicle
November 2, 2024 @ 11:12 am
https://semaglutide.ink/# Patient Portal
RussellJem
November 2, 2024 @ 11:13 am
https://amoxil1st.shop/# top-rated pills
Callum Bradley Jacobs
November 2, 2024 @ 11:24 am
https://www.deviantart.com/hannahowoleak/art/1117270611
Ramonbicle
November 2, 2024 @ 11:26 am
https://stromectol1st.store/# good price
Robertsig
November 2, 2024 @ 11:36 am
paxlovid store: cheapest paxlovid – Visit store
Robertsig
November 2, 2024 @ 11:41 am
paxlovid1st: Visit store – Visit store
Ramonbicle
November 2, 2024 @ 11:46 am
https://amoxil1st.shop/# shop
RussellJem
November 2, 2024 @ 11:47 am
http://gabapentin1st.pro/# licensed gabapentin
Ramonbicle
November 2, 2024 @ 12:01 pm
https://stromectol1st.store/# find bets price
Robertsig
November 2, 2024 @ 12:18 pm
Urgent Specialists: Regenerative Medicine – Urgent Specialists
RussellJem
November 2, 2024 @ 12:22 pm
https://gabapentin1st.pro/# same-day delivery
Robertsig
November 2, 2024 @ 12:24 pm
amoxil 1st shop: buy amoxil online – shop
Ramonbicle
November 2, 2024 @ 12:34 pm
https://stromectol1st.store/# stromectol best price
RussellJem
November 2, 2024 @ 12:56 pm
https://paxlovid1st.store/# paxlovid store
Robertsig
November 2, 2024 @ 1:01 pm
buy amoxil online: amoxil online – cheap
Robertsig
November 2, 2024 @ 1:06 pm
gabapentin best price: licensed gabapentin – Care provides
Ramonbicle
November 2, 2024 @ 1:07 pm
https://stromectol1st.store/# cheapest
Ramonbicle
November 2, 2024 @ 1:27 pm
https://semaglutide.ink/# Urgent Specialists
RussellJem
November 2, 2024 @ 1:31 pm
https://paxlovid1st.store/# Visit store
Snasdxxxax
November 2, 2024 @ 1:39 pm
Snasdxxxax.Snasdxxxax
Ramonbicle
November 2, 2024 @ 1:41 pm
http://semaglutide.ink/# Patient Portal
Robertsig
November 2, 2024 @ 1:45 pm
buy gabapentin: Care provides – gabapentin best price
Robertsig
November 2, 2024 @ 1:50 pm
amoxil 1st shop: top-rated pills – cheap
RussellJem
November 2, 2024 @ 2:07 pm
https://paxlovid1st.store/# Pills Paxlovid
Ramonbicle
November 2, 2024 @ 2:16 pm
https://stromectol1st.store/# good price
Robertsig
November 2, 2024 @ 2:29 pm
top-rated pills: top-rated pills – amoxil online
Robertsig
November 2, 2024 @ 2:34 pm
amoxil price: top-rated pills – buy amoxil online
Ramonbicle
November 2, 2024 @ 2:36 pm
https://stromectol1st.store/# stromectol store
RussellJem
November 2, 2024 @ 2:42 pm
http://semaglutide.ink/# semaglutide
Ramonbicle
November 2, 2024 @ 2:51 pm
http://stromectol1st.store/# stromectol delivery usa
Robertsig
November 2, 2024 @ 3:15 pm
rybelsus price: Specialists – semaglutide online
RussellJem
November 2, 2024 @ 3:18 pm
https://gabapentin1st.pro/# buy gabapentin
Robertsig
November 2, 2024 @ 3:19 pm
Neurontin online: involves multisystem – buy gabapentin
Ramonbicle
November 2, 2024 @ 3:26 pm
https://semaglutide.ink/# Patient Portal
RussellJem
November 2, 2024 @ 3:53 pm
http://amoxil1st.shop/# buy amoxil online
Robertsig
November 2, 2024 @ 3:58 pm
Visit store: cheapest paxlovid – cheapest paxlovid
Ramonbicle
November 2, 2024 @ 4:01 pm
https://paxlovid1st.store/# Visit store
Robertsig
November 2, 2024 @ 4:03 pm
stromectol online: stromectol store – cheapest
RussellJem
November 2, 2024 @ 4:28 pm
https://stromectol1st.store/# stromectol best price
Ramonbicle
November 2, 2024 @ 4:36 pm
https://amoxil1st.shop/# amoxil
Robertsig
November 2, 2024 @ 4:43 pm
involves multisystem: Care provides – gabapentin pro
Robertsig
November 2, 2024 @ 4:48 pm
amoxil price: amoxil price – top-rated pills
Ramonbicle
November 2, 2024 @ 4:56 pm
http://stromectol1st.store/# stromectol delivery usa
RussellJem
November 2, 2024 @ 5:03 pm
https://paxlovid1st.store/# see a healthcare provider
Ramonbicle
November 2, 2024 @ 5:12 pm
https://amoxil1st.shop/# cheap
Robertsig
November 2, 2024 @ 5:29 pm
Patient Portal: semaglutide – Specialists
Ramonbicle
November 2, 2024 @ 5:32 pm
https://paxlovid1st.store/# Visit store
Robertsig
November 2, 2024 @ 5:34 pm
stromectol store: cheapest – find bets price
RussellJem
November 2, 2024 @ 5:40 pm
http://stromectol1st.store/# stromectol online
RussellJem
November 2, 2024 @ 5:47 pm
https://paxlovid1st.store/# Visit store
Ramonbicle
November 2, 2024 @ 5:48 pm
http://stromectol1st.store/# cheapest
Ramonbicle
November 2, 2024 @ 6:09 pm
http://semaglutide.ink/# Patient Portal
Robertsig
November 2, 2024 @ 6:15 pm
amoxil price: amoxil price – amoxil online
RussellJem
November 2, 2024 @ 6:18 pm
http://semaglutide.ink/# Specialists
Robertsig
November 2, 2024 @ 6:20 pm
Urgent Specialists: Rybelsus – Specialists
Ramonbicle
November 2, 2024 @ 6:24 pm
https://gabapentin1st.pro/# gabapentin pro
Ramonbicle
November 2, 2024 @ 6:52 pm
https://paxlovid1st.store/# See risks
RussellJem
November 2, 2024 @ 6:56 pm
https://stromectol1st.store/# stromectol store
Robertsig
November 2, 2024 @ 7:03 pm
paxlovid1st: Pills Paxlovid – Pills Paxlovid
Robertsig
November 2, 2024 @ 7:09 pm
paxlovid store: Visit store – paxlovid store
RussellJem
November 2, 2024 @ 7:39 pm
https://semaglutide.ink/# Urgent Specialists
Robertsig
November 2, 2024 @ 7:56 pm
buy amoxil online: amoxil 1st shop – amoxil online
Robertsig
November 2, 2024 @ 8:01 pm
top-rated pills: Compare Prices – amoxil 1st shop
Ramonbicle
November 2, 2024 @ 8:17 pm
http://stromectol1st.store/# stromectol best price
RussellJem
November 2, 2024 @ 8:21 pm
http://paxlovid1st.store/# see a healthcare provider
RussellJem
November 2, 2024 @ 8:29 pm
https://semaglutide.ink/# Urgent Specialists
Robertsig
November 2, 2024 @ 8:49 pm
see a healthcare provider: Pills Paxlovid – paxlovid1st
Robertsig
November 2, 2024 @ 8:55 pm
stromectol store: find bets price – stromectol delivery usa
RussellJem
November 2, 2024 @ 9:06 pm
https://stromectol1st.store/# stromectol online
RussellJem
November 3, 2024 @ 12:27 am
https://gabapentin1st.pro/# gabapentin pro
MetaMask Extension
November 3, 2024 @ 4:35 am
Thank you so much for your help! I truly appreciate your support and guidance. https://metamask-extension-1059.blogspot.com/2024/11/how-to-download-and-set-up-metamask.html
Percy Clemente
November 3, 2024 @ 6:56 am
Merci pour ce bel article 🙂 !
Ramonbicle
November 3, 2024 @ 7:52 am
https://semaglutide.ink/# semaglutide online
Robertsig
November 3, 2024 @ 8:13 am
rybelsus price: Rybelsus – rybelsus price
RussellJem
November 3, 2024 @ 10:02 am
http://paxlovid1st.store/# see a healthcare provider
Michael Pao
November 3, 2024 @ 11:28 am
Merci pour ce bel article 🙂 !
MetaMask Extension
November 3, 2024 @ 12:12 pm
Thanks, I guess. Appreciate it.
Download MetaMask Extension
November 3, 2024 @ 12:27 pm
A rare moment of appreciation from me. Thanks.
profi teh remont
November 3, 2024 @ 6:11 pm
Сервисный центр предлагает починка посудомоечных машин fagor ремонт посудомоечной машины fagor в москве
Download MetaMask Extension
November 3, 2024 @ 7:56 pm
Much obliged. Don’t expect a parade.
RussellJem
November 3, 2024 @ 8:20 pm
https://paxlovid1st.store/# See risks
Robertsig
November 3, 2024 @ 10:59 pm
amoxil online: amoxil online – shop
tubidy mp3 download
November 3, 2024 @ 11:27 pm
I could not refrain from commenting. Exceptionally well written!
카지노솔루션
November 4, 2024 @ 2:24 am
Hi! I’m working on your blog from my new iPhone! I just wanted to say that I love reading your blog and look forward to all your posts!
Keep up the great work!
Download MetaMask Extension
November 4, 2024 @ 3:47 am
Noted. Thanks.
Ramonbicle
November 4, 2024 @ 5:14 am
https://amoxil1st.shop/# Compare Prices
RussellJem
November 4, 2024 @ 5:48 am
https://semaglutide.ink/# rybelsus price
WallaceRut
November 4, 2024 @ 7:29 am
https://pharm24.pro/# drug prices
drugs for ed
WallaceRut
November 4, 2024 @ 8:34 am
https://pharm24.pro/# medicines for ed
homepage
DavidDAX
November 4, 2024 @ 8:57 am
how can i order prescription drugs without a doctor: low cost prescription – mens erections
WallaceRut
November 4, 2024 @ 9:23 am
https://mexicanpharm24.cheap/# mexican rx online
what type of medicine is prescribed for allergies
DavidDAX
November 4, 2024 @ 9:29 am
medication from mexico pharmacy: mexican pharm 24 – buying prescription drugs in mexico online
DavidDAX
November 4, 2024 @ 10:08 am
purple pharmacy mexico price list: Mexican pharmacy ship US – mexican online pharmacies prescription drugs
WallaceRut
November 4, 2024 @ 10:29 am
http://indianpharm24.pro/# indian pharmacies safe
prescription drugs online without
DavidDAX
November 4, 2024 @ 10:48 am
drugs and medications: cheap drugs – prescription drugs online without doctor
DavidDAX
November 4, 2024 @ 12:11 pm
world pharmacy india: India pharmacy delivery – best online pharmacy india
DavidDAX
November 4, 2024 @ 1:36 pm
cheap erectile dysfunction pills online: cheap medication – online drug store
DavidDAX
November 4, 2024 @ 2:27 pm
best online pharmacy india: medicines from India – best india pharmacy
Download MetaMask Extension
November 4, 2024 @ 2:53 pm
That’ll do. Thanks.
DavidDAX
November 4, 2024 @ 3:02 pm
medications list: cheap prescription drugs – erection pills
DavidDAX
November 4, 2024 @ 3:10 pm
indian pharmacy: Best Indian pharmacy – indian pharmacy online
WallaceRut
November 4, 2024 @ 3:18 pm
http://pharm24.pro/# do i have ed
best pill for ed
Extension
November 4, 2024 @ 3:32 pm
Consider this my ‘thanks.’ Use it wisely.
WallaceRut
November 4, 2024 @ 3:41 pm
http://pharm24.pro/# over the counter erectile dysfunction pills
generic ed drugs
DavidDAX
November 4, 2024 @ 3:55 pm
buying prescription drugs in mexico online: Mexican pharmacy ship US – mexico pharmacies prescription drugs
DavidDAX
November 4, 2024 @ 4:06 pm
best canadian online pharmacy: cheap pharmacy – the best ed pills
WallaceRut
November 4, 2024 @ 4:26 pm
https://indianpharm24.pro/# top 10 online pharmacy in india
buying ed pills online
WallaceRut
November 4, 2024 @ 4:48 pm
http://mexicanpharm24.cheap/# medication from mexico pharmacy
ed aids
DavidDAX
November 4, 2024 @ 4:54 pm
medication from mexico pharmacy: Mexican pharmacy ship US – buying prescription drugs in mexico
DavidDAX
November 4, 2024 @ 5:05 pm
buy anti biotics without prescription: cheap medication – medication for ed dysfunction
WallaceRut
November 4, 2024 @ 5:35 pm
http://indianpharm24.pro/# india pharmacy
ed cures that work
WallaceRut
November 4, 2024 @ 5:56 pm
https://pharm24.pro/# treatment of ed
ed medicine online
DavidDAX
November 4, 2024 @ 6:00 pm
reputable indian online pharmacy: Indian pharmacy to USA – india online pharmacy
DavidDAX
November 4, 2024 @ 6:49 pm
indian pharmacy online: Indian pharmacy to USA – online shopping pharmacy india
DavidDAX
November 4, 2024 @ 7:29 pm
indian pharmacy online: medicines from India – pharmacy website india
DavidDAX
November 4, 2024 @ 7:39 pm
drugs online: buy drugs – canadian pharmacy
WallaceRut
November 4, 2024 @ 7:41 pm
https://pharm24.pro/# viagra without a doctor prescription walmart
online ed medications
WallaceRut
November 4, 2024 @ 8:03 pm
http://mexicanpharm24.cheap/# mexico pharmacies prescription drugs
online drugs
DavidDAX
November 4, 2024 @ 8:22 pm
top 10 online pharmacy in india: indian pharmacy purchase online – reputable indian online pharmacy
WallaceRut
November 4, 2024 @ 8:25 pm
http://indianpharm24.pro/# india online pharmacy
medicine erectile dysfunction
DavidDAX
November 4, 2024 @ 8:32 pm
reputable indian online pharmacy: India pharmacy delivery – best india pharmacy
JorgeFuG
November 5, 2024 @ 12:29 am
erectile dysfunction remedies: cheaper medications – pet meds without vet prescription canada
binance
November 5, 2024 @ 4:33 am
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
gruzoperevozki54
November 5, 2024 @ 7:01 am
Для удобной доставки по СФО выбирайте грузоперевозки попутный груз и отправляйте груз по выгодным условиям.
WilliamCew
November 5, 2024 @ 7:51 am
ed pills that work quickly http://pharm24.pro/# ed drugs online
DevonTum
November 5, 2024 @ 8:03 am
https://indianpharm24.pro/# buy prescription drugs from india
ed in young men
MetaMask Extension
November 5, 2024 @ 9:59 am
That’ll do. Thanks.
Download MetaMask Extension
November 5, 2024 @ 9:59 am
Consider this my ‘thanks.’ Use it wisely.
카지노솔루션
November 5, 2024 @ 1:25 pm
Thank you for the great post! I really enjoyed reading it. You might be a great writer. I will bookmark your blog and come back later. I hope you continue your great posts. Good morning!
WilliamCew
November 5, 2024 @ 5:34 pm
drug store online https://mexicanpharm24.cheap/# buying from online mexican pharmacy
DevonTum
November 5, 2024 @ 5:46 pm
https://pharm24.pro/# buying pills online
online medications
Kairi P. R. Washington
November 5, 2024 @ 6:52 pm
https://s3.amazonaws.com/textstory/index.html?kaksdalle
GregoryTes
November 5, 2024 @ 7:21 pm
เว็บสล็อตโดยตรงคือแพลตฟอร์มการให้บริการเกมผ่านเน็ตที่ให้ผู้เล่นเข้าสู่เกมสล็อตแมชชีนได้โดยตรงจากเว็บ โดยไม่ต้องผ่านเอเย่นต์หรือตัวกลางใดๆ ข้อดีของสล็อตเว็บตรงคือความปลอดภัยเพิ่มขึ้น เนื่องจากผู้เล่นไม่ต้องกังวลเรื่องความเสี่ยงจากการใช้บริการผ่านเอเย่นต์ อีกทั้งยังมีการมอบรางวัลที่มากกว่าและโปรโมชั่นมากมาย เนื่องจากไม่มีค่านายหน้าจากเอเย่นต์ ทำให้นักเล่นเข้าถึงเกมสปินได้อย่างไม่ยุ่งยากและรวดเร็ว พร้อมรับประสบการณ์การเล่นเกมที่ดีขึ้นและไม่สะดุด
การเล่น สล็อตตรง ต่างจาก สล็อตปกติอย่างไร?
สล็อตไม่ผ่านเอเย่นต์เป็นตัวเลือกที่ดีที่ไม่มีการผ่านผู้แทน ทำให้นักเดิมพันสามารถเข้าถึง เกมและเงินรางวัลได้โดยตรงจากเจ้าของเว็บ ลดความเสี่ยงในการโดนโกงหรือถูกหักค่าใช้จ่ายสูง นอกจากนี้ สล็อตตรงยังมีหลายแบบของเกมสล็อตให้เลือกมากกว่าในสล็อตดั้งเดิม เนื่องจากสล็อตตรงมักจะได้รับการอัปเดตและเพิ่มเกมหลากหลายอย่างต่อเนื่อง อัตราการมอบเงิน (เรทการจ่าย) ของสล็อตเว็บตรงมักจะเหนือกว่าสล็อตแบบดั้งเดิม เนื่องจากไม่มีค่าธรรมเนียมพิเศษ ทำให้ผู้เล่นได้รับผลตอบแทนที่สูงขึ้น และยังมี ข้อเสนอและรางวัลที่ดีกว่า โดยสล็อตเว็บตรงมักมีข้อเสนอเพิ่มเติมและการสะสมแต้มที่ดึงดูดใจมากขึ้น
โปรโมชันและโปรโมชั่นพิเศษในเว็บสล็อตตรงที่ไม่ควรพลาด
เว็บสล็อตโดยตรงมักมีโปรโมชันและโปรโมชั่นพิเศษที่น่าดึงดูดสำหรับผู้เล่น เริ่มตั้งแต่โบนัสสมาชิกใหม่สำหรับผู้เล่นใหม่ เงินเพิ่มในการฝาก เครดิตเล่นฟรี รวมถึงการสะสมคะแนนที่สามารถแลกแต้มหรือรางวัลอื่นๆได้ ทำให้ผู้เล่นได้รับความคุ้มค่าและสิทธิประโยชน์มากมาย การมีโปรโมชั่นที่น่าสนใจช่วยให้ผู้เล่นสามารถเพิ่มโอกาสในการชนะและลดค่าใช้จ่ายในการเล่น นอกจากนี้ยังมีโปรโมชันเสริมเช่นคืนเงินบางส่วนจากยอดเสียและการแจกของรางวัลตามวันสำคัญอีกด้วย
สรุปแล้ว สล็อตเว็บตรงเป็นทางเลือกที่เหมาะสมสำหรับผู้ที่ต้องการ ความสะดวกและการรักษาความปลอดภัยในการเล่นเกม มีการจ่ายเงินที่มากกว่า โปรโมชั่นมากมาย และประสบการณ์การเล่นที่ยอดเยี่ยมโดยไม่มีการผ่านเอเย่นต์
profi-teh-remont
November 5, 2024 @ 7:59 pm
Сервисный центр предлагает ремонт moto g31 цены ремонт moto g31 цены
JorgeFuG
November 5, 2024 @ 9:37 pm
mexican drugstore online: mexican pharm 24 – reputable mexican pharmacies online
WilliamCew
November 6, 2024 @ 3:06 am
ed drugs over the counter http://pharm24.pro/# ed meds online
DevonTum
November 6, 2024 @ 3:23 am
http://indianpharm24.pro/# buy medicines online in india
generic ed pills
GregoryTes
November 6, 2024 @ 6:58 am
สล็อตเว็บตรง
สล็อตเว็บตรงคือแพลตฟอร์มการให้บริการการเล่นผ่านเน็ตที่ให้นักเล่นเกมเข้าสู่เกมสล็อตได้โดยตรงจากเว็บ โดยไม่ต้องใช้บริการจากผู้แทนหรือตัวกลางใดๆ ข้อดีของเว็บสล็อตตรงคือการรักษาความปลอดภัยที่มากขึ้น เนื่องจากนักเล่นไม่ต้องกังวลใจเรื่องการเสี่ยงจากการใช้บริการผ่านตัวแทน อีกทั้งยังมีการมอบรางวัลที่เหนือกว่าและโบนัสมากมาย เนื่องจากไม่มีค่านายหน้าจากตัวแทน ทำให้นักเดิมพันเข้าถึงเกมแมชชีนได้อย่างไม่ยุ่งยากและรวดเร็ว พร้อมรับประสบการณ์การเล่นเกมที่มีคุณภาพและไม่สะดุด
การหมุน สล็อตตรง แตกต่างจาก สล็อตทั่วไปอย่างไร?
สล็อตไม่ผ่านเอเย่นต์เป็นตัวเลือกที่ดีที่ไม่มีการผ่านเอเย่นต์ ทำให้นักเดิมพันสามารถเข้าถึง เกมสล็อตและผลตอบแทนได้โดยตรงจากผู้ให้บริการ ลดโอกาสเสี่ยงในการโดนโกงหรือจ่ายค่าธรรมเนียมสูง นอกจากนี้ เว็บสล็อตโดยตรงยังมีความหลากหลายของเกมสปินให้เลือกมากกว่าในสล็อตแบบเดิม เนื่องจากสล็อตเว็บตรงมักจะได้รับการปรับปรุงและเพิ่มเกมใหม่อย่างสม่ำเสมอ อัตราการให้รางวัล (RTP) ของสล็อตเว็บตรงมักจะเหนือกว่าสล็อตดั้งเดิม เนื่องจากไม่มีค่าธรรมเนียมพิเศษ ทำให้นักเดิมพันได้รับรางวัลที่มีมูลค่ามากกว่า อีกทั้งยัง ข้อเสนอและโปรโมชั่นพิเศษที่ดีกว่า โดยสล็อตเว็บตรงมักมีข้อเสนอพิเศษและโปรแกรมคะแนนที่ให้ความคุ้มค่ามากขึ้น
ข้อเสนอพิเศษและโปรโมชั่นพิเศษในเว็บสล็อตตรงที่ไม่ควรพลาด
สล็อตเว็บตรงมักมีโปรโมชั่นและโปรโมชั่นพิเศษที่ดีมากสำหรับนักเดิมพัน เริ่มตั้งแต่โบนัสสมาชิกใหม่สำหรับผู้เล่นใหม่ โบนัสเงินฝากเพิ่ม โบนัสฟรี รวมถึงการสะสมคะแนนที่สามารถแลกเป็นเงินหรือสิทธิประโยชน์ต่างๆได้ ทำให้ผู้เล่นได้รับความคุ้มค่าและสิทธิประโยชน์มากมาย การมีข้อเสนอที่น่าสนใจช่วยให้นักเล่นเกมสามารถมีโอกาสชนะมากขึ้นและลดเงินลงทุนในการเล่น นอกจากนี้ยังมีโปรโมชั่นพิเศษเช่นคืนเงินบางส่วนจากยอดเสียและรางวัลพิเศษตามช่วงเทศกาลอีกด้วย
สรุปว่า สล็อตตรงเป็นทางเลือกที่คุ้มค่าสำหรับผู้ที่ต้องการ ความสะดวกสบายและความปลอดภัยในการเดิมพัน มีการจ่ายเงินที่สูงกว่าปกติ โปรโมชันดีๆ และการเล่นเกมที่ยอดเยี่ยมโดยไม่มีการพึ่งพาตัวกลาง
JorgeFuG
November 6, 2024 @ 7:27 am
drug medication: low cost prescription – legal to buy prescription drugs from canada
Lavabo
November 6, 2024 @ 10:32 am
As a new reader, I am blown away by the quality and depth of your content I am excited to explore your past posts and see what else you have to offer
WilliamCew
November 6, 2024 @ 11:52 am
ed causes and cures https://indianpharm24.pro/# online shopping pharmacy india
JorgeFuG
November 6, 2024 @ 5:29 pm
cheapest online pharmacy india: India pharmacy international – buy prescription drugs from india
MetaMask Extension
November 6, 2024 @ 8:19 pm
Grateful for the insights, thanks again. For additional support, visit https://docs.extension.support/.
Keithrem
November 6, 2024 @ 9:46 pm
demo pg slot
Jelajahi dunia fantasi dan keamanan di mesin slot PG Soft
Slot online telah menjadi salah satu hiburan paling populer di kalangan para gamer. Dengan banyaknya pilihan yang tersedia, PG Soft Slots menonjol sebagai salah satu penyedia terkemuka yang tidak hanya menawarkan pengalaman bermain game yang mengasyikkan, tetapi juga mengutamakan keamanan dan keadilan dalam setiap permainan. Pada artikel ini kita akan melihat beberapa versi demo menarik dari mesin slot PG Soft dan menekankan pentingnya memiliki sistem keamanan.
1. Versi demo Luke Dragon
Dragon Hatch mengajak pemain untuk terjun ke dunia fantasi yang kaya akan kekuatan sihir dan mistik. Namun, fokus utama dari game ini adalah sistem keamanan yang kompleks. Dengan teknologi canggih yang melindungi setiap sesi permainan, pemain bisa menang tanpa khawatir akan gangguan.
2. Slot demo Perjalanan Menuju Kekayaan Hal
Seperti namanya, Journey to Wealth adalah perjalanan menuju kekayaan tanpa akhir. Game ini menawarkan fitur bonus yang menggiurkan dan RTP yang kompetitif. Kegembiraan dan keadilan adalah dua hal yang saling melengkapi di setiap putaran, memastikan bahwa setiap kemenangan diperoleh secara adil.
3. Versi demo slot Bikini Paradise Pg
Di balik keindahan dan pesona tropis Bikini Paradise terdapat protokol keamanan yang ketat. Game ini tidak hanya menawarkan gameplay yang seru namun juga memberikan ketenangan pikiran bagi para pemainnya. Berkat sistem yang terintegrasi, setiap transaksi dan hasil permainan dijamin terlindungi dari penipuan.
4. Slot demo Medusa II: Pencarian Perseus Hal
Medusa II: The Quest of Perseus menawarkan petualangan epik berlatar mitos kuno, mengirim pemain ke pertempuran melawan monster legendaris. Namun perhatian terhadap keselamatan tidak pernah dilupakan. Teknologi terbaru digunakan untuk menjaga integritas setiap putaran, memungkinkan pemain untuk fokus pada kemenangan tanpa gangguan.
5. Versi demo slot Bombs Away Pg
Mengusung tema perang dunia yang menegangkan, Bombs Away menjanjikan gameplay yang seru. Meski demikian, PG Soft tetap mengutamakan keselamatan. Sistem yang canggih memastikan setiap transaksi dan hasil permainan tersimpan sehingga memberikan kepercayaan diri pemain untuk terus berjuang meraih kemenangan.
6. Versi demo mesin slot Bull Fight Hal
Bull Fight mengajak pemain ke arena adu banteng yang seru. Di sini, keberanian dan keterampilan adalah kunci kemenangan. PG Soft menjamin keadilan dan keadilan di setiap putaran permainan, menciptakan pengalaman bermain yang sempurna.
7. Versi demo mesin slot Champion Pg Muay Thai
Menampilkan tema seni bela diri yang menarik, Juara Muay Thai melibatkan pemain dalam pertarungan intens di atas ring. Selain aksinya yang seru, PG Soft juga menekankan pentingnya keselamatan. Setiap putaran dipantau dan dijaga dengan cermat untuk memastikan gameplay tanpa gangguan eksternal.
Keuntungan memainkan soft slot versi demo dengan RTP PG tinggi
Salah satu keuntungan utama memainkan game demo PG Soft adalah RTP-nya yang tinggi, seringkali lebih tinggi dari rata-rata industri. Ini berarti bahwa pemain memiliki peluang lebih besar untuk menghasilkan keuntungan besar dari investasi mereka. Tak heran jika mesin slot PG Soft yang memadukan kesenangan bermain game dan keamanan terjamin menjadi pilihan favorit para gamer.
Di dunia yang terus berkembang ini, PG Soft tidak hanya berhasil menciptakan gameplay yang seru, tetapi juga aman dan adil. Dengan berbagai versi demo permainan yang menarik, pemain dapat menjelajahi keajaiban tanpa khawatir akan risiko yang tidak diinginkan.
Jefferycof
November 6, 2024 @ 10:06 pm
Hercule99: Kumpulan 8 buah Permainan Uji Coba PGS Bebas Curang Dapat Dipercaya di Tanah Air
PGS sudah menjadi favorit pilihan utama bagi banyak pecinta slot di Indonesia, menyuguhkan ragam permainan slot yang memikat dengan keasyikan dan kesenangan berkelanjutan. Di saat popularitasnya semakin tinggi, ada juga kecemasan seputar ketidakjujuran atau gangguan dalam permainan judi online. Tetapi, PG Slot telah berhasil mengatasi masalah ini dengan menciptakan game slot tanpa manipulasi yang terjamin serta dapat dipercaya.
Dengan teknologi keamanan terbaru dan fitur inovatif, Permainan PG Slot memastikan bahwa game yang disediakan aman dan terkontrol untuk para pemain. Di bawah ini daftar 8 slot percobaan PG tanpa manipulasi yang gacor dan aman di Nusantara:
1. Demo Fortune Mouse PG Slot
Tikur Keberuntungan adalah gim slot yang penuh daya tarik dengan visual yang indah dan tema yang menggemaskan. Di luar visual yang cantik, permainan ini menggunakan enkripsi modern agar melindungi semua data pemain. Setiap putaran dijamin bebas dari manipulasi, agar permainan tetap nyaman.
2. Slot Demo Dragon Hatch
Menetas Naga mengajak pemain ke semesta fiksi yang dilengkapi kemegahan dan kekuatan mistis. Namun, kekuatan utama game ini terdapat pada fitur keamanannya. Dilengkapi proteksi anti-kecurangan, game ini mewujudkan keadilan dalam kemenangan tanpa gangguan tanpa campur tangan pihak luar.
3. Versi Demo Journey to the Wealth Slot PG
Slot Journey Wealth mengundang pemain dalam perjalanan mencari kekayaan tanpa batas. Berbekal bonus melimpah dan Return to Player yang tinggi, slot ini memberikan kegembiraan berlipat dan kejujuran di setiap perputaran. Pengguna merasakan serunya bermain tanpa kekhawatiran mengenai manipulasi.
4. Demo Bikini Tropis Slot PG
Permainan ini mengundang player dalam suasana pulau tropis yang cantik dan penuh sukacita. Di balik keindahan tropisnya, Bikini Paradise dibekali sistem keamanan yang andal, dan menjadikannya salah satu game Slot PG anti-rungkad yang aman. User bisa menikmati slot tanpa khawatir akan adanya kecurangan.
5. Percobaan Medusa II: Petualangan Perseus Pocket Games Slot
Di dalam game Medusa II, user terjun dalam kisah epik menghadapi makhluk legendaris. Game ini dilengkapi dengan keamanan mutakhir yang menjaga kejujuran tiap giliran. Dengan demikian, pengguna bisa fokus mengejar kemenangan tanpa terganggu campur tangan luar.
6. Uji Coba Bombs Away Slot PG
Mengusung tema perang yang mendalam, Bombs memicu semangat para pemain. Saat permainan memanas, keamanan tetap menjadi prioritas utama. Proteksi dari PG Soft mengamankan semua transaksi dan hasil game, menjamin keamanan bagi pengguna saat mengejar kemenangan.
7. Percobaan Adu Banteng PGS
Slot Bull Fight mengajak pemain ke arena banteng yang seru. Di dalam game ini, PG Soft memastikan keadilan dalam setiap giliran. Pemain bisa menikmati sensasi permainan yang adil dan aman tanpa takut akan kecurangan.
8. Uji Coba Juara Muay Thai Slot PG
Game Muay Thai Champion bertema seni bela diri menarik, memasukkan pengguna dalam duel di atas ring. Slot ini tidak hanya menarik dari segi tema, namun juga di segi keamanan. Setiap putaran dipantau dengan ketat, memastikan pengalaman main tanpa gangguan atau curang.
Keuntungan Bermain Demo Slot PG Soft dengan RTP Tinggi
Demo game PG Soft anti-kecurangan dikenal dengan RTP besar yang di atas standar industri. Memberikan peluang lebih besar kepada pengguna agar bisa meraih kembali modal mereka. Memilih slot PG Soft tidak sekadar hiburan, namun juga potensi untung yang lebih besar.
Melalui list game yang tersedia di atas, pemain dapat menikmati pengalaman bermain slot yang adil, aman, dan menyenangkan. Teknologi canggih yang diterapkan oleh PG Slot menjaga kelancaran dalam setiap permainan, memberikan rasa percaya bagi pengguna.
WilliamCew
November 6, 2024 @ 10:35 pm
ed vacuum pumps http://pharm24.pro/# how to cure ed
JorgeFuG
November 7, 2024 @ 5:06 am
buy prescription drugs from india: cheaper medications – drugs and medications
Sugar defender
November 7, 2024 @ 7:44 am
Including Sugar Defender into my everyday regimen general wellness.
As somebody who prioritizes healthy and balanced consuming,
I appreciate the additional protection this supplement supplies.
Since beginning to take it, I have actually noticed a significant renovation in my energy levels and a substantial decrease in my need for harmful snacks
such a such a profound influence on my every day
life.
WilliamCew
November 7, 2024 @ 8:30 am
sildenafil without a doctor’s prescription http://indianpharm24.pro/# india pharmacy
MetaMask Extension
November 7, 2024 @ 8:36 am
Thanks, you saved me time and trouble! For further help, check out https://download.extension.support/
DevonTum
November 7, 2024 @ 9:25 am
https://mexicanpharm24.cheap/# п»їbest mexican online pharmacies
natural herbs for ed
JorgeFuG
November 7, 2024 @ 2:41 pm
mail order pharmacy india: indian pharm 24 – top online pharmacy india
WilliamCew
November 7, 2024 @ 6:02 pm
mexican pharmacy without prescription https://mexicanpharm24.cheap/# buying from online mexican pharmacy
DevonTum
November 7, 2024 @ 7:09 pm
http://pharm24.pro/# cheap erectile dysfunction
herbal ed treatment
Charlesjip
November 7, 2024 @ 8:39 pm
สล็อตเว็บตรงคือแพลตฟอร์มการให้บริการการเล่นเกมออนไลน์ที่ให้นักเล่นเกมเชื่อมต่อเกมสล็อตแมชชีนได้โดยตรงจากเว็บ โดยไม่ต้องผ่านผู้แทนหรือตัวกลางใดๆ ข้อดีของสล็อตเว็บตรงคือการป้องกันสูงขึ้น เนื่องจากนักเล่นไม่ต้องกังวลเรื่องโอกาสเสี่ยงจากการใช้บริการผ่านเอเย่นต์ อีกทั้งยังมีการมอบเงินรางวัลที่มากกว่าและโปรโมชั่นมากมาย เนื่องจากไม่มีค่าบริการจากตัวแทน ทำให้ผู้เล่นเข้าถึงเกมแมชชีนได้อย่างไม่ยุ่งยากและรวดเร็ว พร้อมรับการเล่นที่ดีขึ้นและไม่สะดุด
การเดิมพัน สล็อตเว็บตรง ไม่เหมือนกับ สล็อตแบบดั้งเดิมอย่างไร?
เว็บสล็อตตรงเป็นตัวเลือกที่ดีที่ไม่มีการผ่านตัวแทน ทำให้นักเดิมพันสามารถเข้าถึงเกมได้ทันที เกมสปินและผลตอบแทนได้โดยตรงจากผู้ดำเนินการ ลดการเสี่ยงในการเสียเปรียบหรือถูกหักค่าธรรมเนียมสูง นอกจากนี้ สล็อตตรงยังมีหลายแบบของเกมสล็อตให้เลือกเล่นมากกว่าในสล็อตดั้งเดิม เนื่องจากสล็อตตรงมักจะอัปเดตบ่อยและเพิ่มเกมใหม่อย่างต่อเนื่อง อัตราการมอบเงิน (RTP) ของสล็อตเว็บตรงมักจะสูงกว่าสล็อตแบบดั้งเดิม เนื่องจากไม่มีค่าใช้จ่ายเพิ่มเติม ทำให้นักเดิมพันได้ผลตอบแทนผลตอบแทนที่มีมูลค่ามากกว่า ทั้งยัง โปรโมชั่นและรางวัลที่ดีกว่า โดยเว็บสล็อตตรงมักมีข้อเสนอเพิ่มเติมและโปรแกรมคะแนนที่ดึงดูดใจมากขึ้น
โปรโมชั่นและโบนัสในสล็อตตรงที่น่าสนใจ
เว็บสล็อตโดยตรงมักมีข้อเสนอและโปรโมชั่นพิเศษที่น่าดึงดูดสำหรับผู้เล่น เริ่มตั้งแต่โบนัสต้อนรับสำหรับผู้เล่นใหม่ โบนัสเติมเงิน เครดิตฟรี รวมถึงโปรแกรมสะสมแต้มที่สามารถแลกแต้มหรือรางวัลอื่นๆได้ ทำให้ผู้เล่นมีความคุ้มค่าและข้อดีมากมาย การมีโปรโมชั่นที่คุ้มค่าช่วยให้นักเล่นเกมสามารถมีโอกาสชนะมากขึ้นและลดเงินลงทุนในการเล่น นอกจากนี้ยังมีโปรโมชันเสริมเช่นเงินคืนบางส่วนและรางวัลพิเศษตามเทศกาลต่างๆอีกด้วย
สรุปว่า สล็อตเว็บตรงเป็นตัวเลือกที่ดีสำหรับคนที่มองหา ความสะดวกสบายและการรักษาความปลอดภัยในการเดิมพัน มีการจ่ายเงินที่สูงกว่า โปรโมชั่นมากมาย และประสบการณ์การเล่นที่ดีโดยไม่มีการใช้ตัวแทน
JorgeFuG
November 8, 2024 @ 1:28 am
canadian online pharmacy: cheaper medications – ed medications
Derekfet
November 8, 2024 @ 5:09 am
Casino Siteleri: casino siteleri win – Casino Siteleri
LarryPab
November 8, 2024 @ 5:10 am
matadorbet.bid [url=https://matadorbet.bid/#]matadorbet giris[/url] matadorbet giris
ClydeJix
November 8, 2024 @ 5:11 am
matadorbet bid: matadorbet giris – matadorbet giris
ClydeJix
November 8, 2024 @ 5:16 am
ultrabet giris: ultrabet giris – ultrabet giris
Hassanagize
November 8, 2024 @ 5:40 am
https://denemebonusuverensiteler.top/# deneme bonusu veren siteler denemebonusu2026.com
deneme bonusu veren siteler mycbet.com
Derekfet
November 8, 2024 @ 5:40 am
ultrabet yeni giris 1125: ultrabet tr online – ultrabet bonus
Derekfet
November 8, 2024 @ 5:48 am
slot oyunlar? puf noktalar?: slot siteleri – en cok kazand?ran slot oyunlar?
ClydeJix
November 8, 2024 @ 5:57 am
slot oyunlar?: slot oyunlar? – en cok kazand?ran slot oyunlar?
Hassanagize
November 8, 2024 @ 6:02 am
https://ultrabet-tr.online/# ultrabet giris
deneme bonusu veren siteler mycbet.com
ClydeJix
November 8, 2024 @ 6:02 am
Casino Siteleri: Casino Siteleri – Deneme Bonusu Veren Siteler
Hassanagize
November 8, 2024 @ 6:18 am
https://casinositeleri.win/# guvenilir casino siteleri
deneme bonusu veren siteler mycbet.com
Derekfet
November 8, 2024 @ 6:20 am
Deneme Bonusu Veren Siteler: Casino Siteleri – Deneme Bonusu Veren Siteler
Hassanagize
November 8, 2024 @ 6:39 am
https://matadorbet.bid/# matadorbet.bid
deneme bonusu veren siteler mycbet.com
ClydeJix
November 8, 2024 @ 6:42 am
Deneme Bonusu Veren Siteler: casino siteleri win – guvenilir casino siteleri
ClydeJix
November 8, 2024 @ 6:46 am
deneme bonusu veren siteler 2024: deneme bonusu veren siteler 2024 – deneme bonusu veren siteler yeni
Derekfet
November 8, 2024 @ 6:58 am
deneme bonusu veren siteler mycbet.com: deneme bonusu veren siteler – deneme bonusu veren yeni siteler
ClydeJix
November 8, 2024 @ 7:27 am
matadorbet.bid: matadorbet giris – matadorbet.bid
ClydeJix
November 8, 2024 @ 7:31 am
Canl? Casino Siteleri: casino siteleri win – Casino Siteleri
Derekfet
November 8, 2024 @ 7:37 am
deneme bonusu veren siteler: deneme bonusu veren yeni siteler – deneme bonusu veren siteler denemebonusu2026.com
Hassanagize
November 8, 2024 @ 7:54 am
http://casinositeleri.win/# Deneme Bonusu Veren Siteler
deneme bonusu veren siteler
Hassanagize
November 8, 2024 @ 8:10 am
https://slot-tr.online/# slot tr online
deneme bonusu veren siteler yerliarama.org
Derekfet
November 8, 2024 @ 8:15 am
slot tr online: slot siteleri – az parayla cok kazandiran slot oyunlar?
Derekfet
November 8, 2024 @ 9:30 am
ultrabet bonus: ultrabet guncel – ultrabet bonus
ClydeJix
November 8, 2024 @ 10:21 am
ultrabet tr online: ultrabet guncel – ultrabet giris
MetaMask Extension
November 8, 2024 @ 6:37 pm
Thanks. https://sites.google.com/view/metamask-extension-62165/metamask-extension-for-chrome-firefox-and-brave-a-comprehensive-guide
сейф для оружия
November 8, 2024 @ 6:43 pm
Тут можно преобрести купить сейф с доставкой в москве сейф для ружья
onexslotsfun
November 8, 2024 @ 7:50 pm
Для игроков на Android 1xslots скачать бесплатно и получите фриспины за регистрацию.
Derekfet
November 8, 2024 @ 9:25 pm
slot tr online: en kazancl? slot oyunlar? – slot siteleri
купить сейф москва для дома
November 8, 2024 @ 9:45 pm
Тут можно купить сейфы для дома купить домашний сейф в москве
огнестойкие сейфы
November 9, 2024 @ 2:12 am
Тут можно преобрести купить огнестойкий сейф несгораемые сейфы
MetaMask Extension
November 9, 2024 @ 2:54 am
Thank you for taking the time to read! https://sites.google.com/view/metamask-extension-detail-1450/
카지노솔루션
November 9, 2024 @ 6:16 am
Hi! I got some really helpful advice from this article! It’s the little changes that make the biggest difference. Thanks for sharing!
MetaMask Extension for Chrome
November 9, 2024 @ 8:09 am
Thanks for stopping by and reading! https://docs.webstore.it.com/
купить сейф для оружия
November 9, 2024 @ 1:26 pm
Тут можно преобрести купить сейф с доставкой в москве цена сейфа для оружия
메타마스크
November 9, 2024 @ 3:49 pm
Heartfelt thanks for being here! https://sites.google.com/view/download-metamask-wallet/
Hassanagize
November 9, 2024 @ 7:10 pm
http://denemebonusuverensiteler.top/# deneme bonusu veren siteler
deneme bonusu veren siteler
슬롯솔루션
November 10, 2024 @ 2:14 am
I used to read small articles or reviews that clearly stated their motivations, and this post you are reading now is no exception.
купить сейф для охотничьего ружья
November 10, 2024 @ 5:40 am
Тут можно преобрести оружейные сейфы купить оружейный сейф купить
luckyjetraketa
November 10, 2024 @ 6:36 am
Начните игру с бонусом, используя промокод лаки джет.
tubidy
November 10, 2024 @ 5:20 pm
Good post. I learn something totally new and challenging on blogs I stumbleupon everyday. It’s always helpful to read articles from other writers and practice a little something from their websites.
xxxtubebest.com/xp7RRsktWWcnk
November 10, 2024 @ 5:22 pm
Everthing said wwas actually very reasonable. However, connsider this, suppose yoou composd a cstchier ost title?
I mean, I don’t want to twll yoou how to ruun yohr website, butt supppose youu added a tittle thast grabbed people’s attention? I mean Understanding
spit is a little plain. Youu could lolk at Yahoo’s ome pag and see how thhey creae adticle
totles to geet people interested. Youu mitht add a video orr a ppicture or twwo to
gab readers intterested bout what you’ve gott
too say. In mmy opinion, iit could brng yor boog a liittle
bitt mokre interesting.
메타마스크
November 10, 2024 @ 10:24 pm
Thank you for reading and sharing your thoughts! https://webstore.builders
RobertAtose
November 11, 2024 @ 9:37 am
http://erepharm.com/# ed pills
Dennisinamy
November 11, 2024 @ 11:52 am
cheapest ed pills ere pharm: ED pills non prescription – best ed pill ere pharm
warm blankets
November 11, 2024 @ 1:23 pm
Hey! Do you know if they make any plugins to assist with Search Engine
Optimization? I’m trying to get my site to rank for some targeted
keywords but I’m not seeing very good success. If you know of any please share.
Thanks! You can read similar art here: Blankets
Lloydwaf
November 11, 2024 @ 2:45 pm
https://gabapharm.com/# cheapest Gabapentin GabaPharm
огнестойкие сейфы
November 11, 2024 @ 6:21 pm
Тут можно преобрести сейф пожаростойкий купить сейф несгораемый
Curtisspeve
November 11, 2024 @ 6:56 pm
erepharm.com [url=http://erepharm.com/#]ED meds online with insurance[/url] ere pharm
купить сейф для охотничьего ружья
November 11, 2024 @ 9:17 pm
Тут можно преобрести сейфы оружейные магазин сейфы для оружия
abdul acosta
November 11, 2024 @ 9:32 pm
Hello everyone, here everyone is sharing this kind of intimacy. So
it is difficult to read this webpage, and I
used to see this webpage all the time.
pupis
November 12, 2024 @ 12:07 am
Чтобы начать выигрывать, просто скачайте приложение БК и делайте ставки на любые спортивные события
Lloydwaf
November 12, 2024 @ 1:34 am
https://furpharm.com/# furosemide furpharm.com
RobertAtose
November 12, 2024 @ 1:57 am
https://rybpharm.com/# rybpharm
Curtisspeve
November 12, 2024 @ 5:36 am
buy rybelsus [url=http://rybpharm.com/#]rybpharm rybelsus[/url] semaglutide
Dennisinamy
November 12, 2024 @ 9:24 am
rybpharm: buy rybelsus rybpharm – semaglutide
카지노솔루션
November 12, 2024 @ 11:57 am
Hei, alt går bra her og selvfølgelig deler alle data. Det er virkelig flott. Fortsett å skrive.
JamesBer
November 12, 2024 @ 12:56 pm
Ритуальная служба https://pohoronnoe-agentstvo.kz ритуальные услуги. Похоронные услуги по государственным ценам. Более 15 лет занимаемся организацией похорон
JamesBer
November 12, 2024 @ 1:08 pm
Ритуальная служба https://pohoronnoe-agentstvo.kz ритуальные услуги. Похоронные услуги по государственным ценам. Более 15 лет занимаемся организацией похорон
Snaptik.icu
November 12, 2024 @ 1:34 pm
Excellent post. I’m facing some of these issues as well..
RobertAtose
November 12, 2024 @ 4:51 pm
http://furpharm.com/# lasix
Jefferysoync
November 12, 2024 @ 6:40 pm
магазин кейсов кс го кейсы кс го
Jasoncah
November 12, 2024 @ 6:46 pm
Слот Sugar Rush https://sugar-rush-play.ru Бесплатная ДЕМО-версия, значения RTP и Max Win, Бонусы и фриспины от казино.
JaredUrivy
November 12, 2024 @ 7:29 pm
главные новости спорта https://beachsoccer.com.ua фото, видео и инфографика, аналитика и блоги от экспертов и известных спортсменов
profi teh remont
November 12, 2024 @ 7:29 pm
Сервисный центр предлагает ремонт телефонов senseit адреса ремонт телефонов senseit адреса
Lloydwaf
November 12, 2024 @ 9:56 pm
https://gabapharm.com/# gabapentin
огнестойкий сейф
November 12, 2024 @ 10:17 pm
Тут можно преобрести купить противопожарный сейф сейф несгораемый
비아그라구매
November 13, 2024 @ 12:54 am
Энэ сайхан нийтлэлд баярлалаа. Өөр нэг дурдах зүйл бол олон тооны дижитал камерууд шинэ томруулах линзтэй байдаг бөгөөд энэ нь танд “томруулж”, “томруулж” ямар ч дүр зургийг бага багаар харуулах боломжийг олгодог.
Curtisspeve
November 13, 2024 @ 2:13 am
kamagra [url=https://kampharm.shop/#]kamagra[/url] kamagra
ThomasBap
November 13, 2024 @ 5:20 am
оценка условий труда цена оценка соут москва
Dennisinamy
November 13, 2024 @ 6:29 am
rybpharm canada: buy rybelsus rybpharm – buy rybelsus online usa
Danielkag
November 13, 2024 @ 7:10 am
секс шоп каталог sex shop
sünnet organizasyonu
November 13, 2024 @ 7:18 am
Your blog post had me hooked from the very beginning!
bandar togel online
November 13, 2024 @ 9:44 am
Hi to every one, it’s genuinely a fastidious for me to pay
a quick visit this web page, it contains precious Information.
купить сейф для охотничьего ружья
November 13, 2024 @ 12:32 pm
Тут можно преобрести купить шкаф для ружья сейф для оружие
Curtisspeve
November 13, 2024 @ 12:56 pm
buy gabapentin [url=http://gabapharm.com/#]Buy gabapentin for humans[/url] Buy gabapentin for humans
raketaigra
November 13, 2024 @ 3:59 pm
Lucky Jet игра — это шанс для тех, кто хочет заработать на азарте.
сейф
November 14, 2024 @ 1:24 am
Здесь можно преобрести сейф купить купить сейф оптом
Michaelbip
November 14, 2024 @ 3:10 am
Потрясающий игровой сайт Lev открывает для вас потрясающие опции для крупных выигрышей!
На онлайн-казино Lev вас встречают акционные игры и разнообразные игры. Каждый раз здесь вы попробуете завоевать колоссальные суммы с легкостью и удовольствием!
Сайт Lev предлагает ежедневные бонусы, которые улучшают ваш настроение. Примите участие в турнирах, чтобы достичь успеха.
Казино Лев предлагает максимально удобный доступ, что делает игру комфортной на любой платформе.
На игровом доме Лев вас встретят не только видеослоты, но и живые дилеры, которые дадут вам возможность насладиться реальным игровым процессом из любой точки мира.
Кроме того, наш бонусная система дадут вам бесплатные вращения каждый день.
Играй с наслаждением, побеждай с игровым домом Лев, и получай славы каждый день!
На площадке Lev вас ждут щедрые бонусы и разнообразные игры. Всегда здесь вы успеете завоевать огромные джекпоты.
Зарегистрируйтесь на нашем сайте и запустите игру [url=https://vostok-sk.ru/igrovie-avtomati/]игровые автоматы[/url] уже сейчас! [url=https://vostok-sk.ru/]казино лев[/url], [url=https://vostok-sk.ru/faq/]faq[/url], [url=https://vostok-sk.ru/rules/]правила[/url], [url=https://vostok-sk.ru/bonus/]бонус[/url]
казино лев 2024
Lloydwaf
November 14, 2024 @ 4:34 am
https://rybpharm.com/# rybpharm canada
Nicholas A. T. Robertson
November 14, 2024 @ 5:20 am
This blog has become a part of my daily routine I start my mornings with a cup of coffee and your latest post
Curtisspeve
November 14, 2024 @ 7:49 am
rybpharm cheap semaglutide [url=https://rybpharm.com/#]semaglutide[/url] buy rybelsus rybpharm
купить сейф для охотничьего ружья
November 14, 2024 @ 9:51 am
Тут можно преобрести шкаф для оружия оружейный сейф для ружья
RobertAtose
November 14, 2024 @ 11:10 am
https://furpharm.com/# furpharm
огнестойкие сейфы
November 14, 2024 @ 12:34 pm
Тут можно преобрести купить несгораемый сейф сейф несгораемый
LesleyLof
November 14, 2024 @ 12:57 pm
компании специальная оценка условий труда стоимость специальной оценки условий труда
Romina C. C. Galvan
November 14, 2024 @ 9:43 pm
Your writing has a way of resonating with me and making me feel understood Thank you for being a relatable and authentic voice
슬롯솔루션
November 14, 2024 @ 10:49 pm
Bonjour, c’est un bon article sur l’impression multimédia. Nous savons tous que les médias constituent une fantastique source de données.
Dennisinamy
November 15, 2024 @ 12:07 am
furosemide furpharm.com: lasix – buy lasix fur pharm
social media girls forums
November 15, 2024 @ 6:40 am
social media girls forums
Davidtam
November 15, 2024 @ 9:43 am
Overview of Crypto Transfer Check and Conformity Solutions
In contemporary crypto market, ensuring deal transparency and compliance with Anti-Money Laundering (AML) and Customer Identification regulations is vital. Here is an overview of well-known sites that provide tools for crypto transfer surveillance, verification, and fund safety.
1. Token Metrics
Description: Tokenmetrics delivers cryptocurrency evaluation to examine possible scam threats. This solution lets individuals to check coins prior to purchase to avoid possibly scam holdings. Features:
– Risk analysis.
– Suitable for holders aiming to bypass questionable or fraud assets.
2. Metamask Monitor Center
Overview: Metamask.Monitory.Center enables individuals to review their cryptocurrency assets for questionable actions and standard conformity. Benefits:
– Verifies coins for legitimacy.
– Provides warnings about possible asset blockages on specific exchanges.
– Delivers comprehensive reports after address sync.
3. Bestchange.com
Overview: Bestchange.ru is a platform for monitoring and verifying crypto transaction deals, ensuring transparency and transaction safety. Highlights:
– Transaction and account monitoring.
– Compliance checks.
– Internet interface; compatible with BTC and various different coins.
4. Bot amlchek
Description: AMLchek is a portfolio tracker and AML tool that employs machine learning methods to find dubious activity. Features:
– Deal observation and user verification.
– Offered via web version and Telegram.
– Supports digital assets like BSC, BTC, DOGE, and other types.
5. Alfabit AML
Summary: AlfaBit offers thorough AML services specifically made for the digital currency field, supporting firms and banks in maintaining compliance compliance. Advantages:
– Extensive AML options and evaluations.
– Complies with modern protection and conformity requirements.
6. AMLNode
Summary: AML Node provides AML and identification tools for digital currency businesses, which includes transaction observing, compliance checks, and risk assessment. Features:
– Threat analysis options and restriction screenings.
– Useful for ensuring protected company processes.
7. Btrace.io
Overview: Btrace.AMLcrypto.io specializes in resource validation, providing transaction monitoring, compliance evaluations, and help if you are a affected by theft. Benefits:
– Effective help for resource recovery.
– Transfer tracking and safety features.
Dedicated USDT Verification Services
Our platform also reviews multiple services providing validation solutions for Tether transfers and holdings:
– **USDT TRC20 and ERC20 Validation:** Many sites provide comprehensive evaluations for USDT deals, assisting in the identification of doubtful activity.
– **AML Validation for USDT:** Solutions are available for observing for fraudulent actions.
– **“Cleanliness” Checks for Holdings:** Validation of transfer and account legitimacy is available to detect likely threats.
**Wrap-up**
Selecting the suitable platform for validating and monitoring crypto deals is important for providing protection and standard conformity. By viewing our evaluations, you can select the ideal solution for transfer observation and fund protection.
огнестойкие сейфы
November 15, 2024 @ 12:10 pm
Тут можно преобрести сейф огнестойкий цена купить огнеупорный сейф
swift quilting company
November 15, 2024 @ 12:15 pm
Check out the following page for some recommendation.
BradleyJopay
November 15, 2024 @ 1:05 pm
https://indianpharmacyeasy.com/# best india pharmacy
Leo Hall
November 15, 2024 @ 1:10 pm
훌륭한 포스팅 감사합니다! 정말 재미있게 읽었습니다. 당신은 훌륭한 작가가 될 수 있습니다.
블로그를 북마크하고 언젠가 다시 방문하겠습니다.
훌륭한 포스팅을 계속하시기를 바랍니다. 좋은 오후 보내세요!
Larryglops
November 15, 2024 @ 4:21 pm
indian pharmacy online: Indian pharmacy international shipping – buy medicines online in india
сейф для оружия
November 15, 2024 @ 5:48 pm
Тут можно преобрести где купить сейф для оружия сейф для ружья
JefferyHot
November 15, 2024 @ 6:29 pm
ed pills that work quickly: canadiandrugsgate – buy prescription drugs from canada
Davidtam
November 15, 2024 @ 9:06 pm
Overview of Cryptocurrency Transfer Verification and Compliance Services
In the current cryptocurrency sector, ensuring transfer clarity and adherence with Anti-Money Laundering (AML) and Know Your Customer (KYC) standards is vital. Following is an summary of well-known services that provide services for cryptocurrency deal surveillance, validation, and resource security.
1. Token Metrics Platform
Overview: Tokenmetrics provides digital asset analysis to assess likely fraud threats. This platform enables investors to review coins ahead of buying to evade potentially fraudulent assets. Highlights:
– Threat assessment.
– Perfect for investors looking to steer clear of risky or scam ventures.
2. Metamask.Monitory.Center
Summary: Metamask Monitor Center enables users to review their crypto holdings for suspicious actions and regulatory compliance. Benefits:
– Checks assets for legitimacy.
– Provides warnings about likely resource restrictions on specific platforms.
– Gives detailed insights after account sync.
3. Best Change
Summary: Bestchange.ru is a site for tracking and validating digital exchange deals, guaranteeing openness and transfer safety. Benefits:
– Transaction and wallet tracking.
– Sanctions checks.
– Web-based interface; compatible with BTC and multiple additional coins.
4. AML Bot
Overview: AMLCheck Bot is a holding tracker and anti-money laundering service that utilizes machine learning models to identify questionable actions. Features:
– Transaction tracking and identity check.
– Offered via web version and chat bot.
– Compatible with coins including BSC, BTC, DOGE, and other types.
5. Alfabit AML
Summary: AlphaBit offers complete anti-money laundering solutions customized for the cryptocurrency field, supporting businesses and banks in maintaining compliance compliance. Advantages:
– Extensive compliance features and checks.
– Complies with modern safety and regulatory standards.
6. AMLNode
Description: AML Node offers anti-money laundering and KYC services for cryptocurrency businesses, such as deal monitoring, compliance validation, and evaluation. Benefits:
– Threat analysis tools and restriction screenings.
– Important for guaranteeing protected business operations.
7. Btrace.AMLcrypto.io
Description: Btrace.AMLcrypto.io specializes in asset verification, delivering deal observation, sanctions screenings, and support if you are a target of theft. Highlights:
– Useful support for asset recovery.
– Deal tracking and security options.
Specialized USDT Check Options
Our website also provides information on various services that offer check tools for USDT deals and wallets:
– **USDT TRC20 and ERC20 Validation:** Many services offer thorough evaluations for USDT transactions, helping in the detection of doubtful transactions.
– **AML Screening for USDT:** Options are provided for tracking for money laundering actions.
– **“Cleanliness” Validation for Holdings:** Validation of transaction and account “cleanliness” is provided to detect possible risks.
**Summary**
Selecting the right tool for checking and monitoring crypto transfers is crucial for providing security and standard compliance. By consulting our recommendations, you can find the best solution for transaction tracking and resource safety.
Glennlashy
November 15, 2024 @ 10:18 pm
cheapest online pharmacy india [url=http://indianpharmacyeasy.com/#]indianpharmacyeasy[/url] top 10 online pharmacy in india
how to describe foamy ocean waves in writing
November 16, 2024 @ 3:17 am
how to describe foamy ocean waves in writing
i dont have school spirit
November 16, 2024 @ 4:28 am
i dont have school spirit
JefferyHot
November 16, 2024 @ 6:10 am
mexico pharmacies prescription drugs: mexican drugstore online – mexico drug stores pharmacies
сейф огнестойкий
November 16, 2024 @ 7:00 am
Тут можно преобрести пожаростойкие сейфы купить противопожарный сейф
Davidtam
November 16, 2024 @ 7:04 am
CFT compliance check for cryptocurrency wallets
Overview of Cryptocurrency Transaction Validation and Conformity Solutions
In contemporary cryptocurrency industry, ensuring transfer openness and conformity with AML and Customer Identification rules is crucial. Here is an summary of well-known platforms that deliver services for crypto transfer monitoring, validation, and asset security.
1. Token Metrics Platform
Description: Tokenmetrics offers crypto assessment to evaluate potential risk dangers. This solution allows users to check tokens ahead of buying to prevent likely fraudulent resources. Attributes:
– Threat assessment.
– Perfect for buyers aiming to avoid questionable or scam ventures.
2. Metamask Monitor Center
Summary: Metamask.Monitory.Center allows users to check their cryptocurrency holdings for questionable actions and regulatory compliance. Features:
– Validates assets for legitimacy.
– Provides alerts about likely resource restrictions on particular exchanges.
– Gives thorough insights after account sync.
3. Best Change
Summary: Bestchange.ru is a platform for tracking and verifying cryptocurrency transaction transactions, ensuring openness and transaction security. Benefits:
– Transfer and holding observation.
– Sanctions screening.
– Internet platform; supports BTC and various additional digital assets.
4. AMLCheck Bot
Overview: AMLCheck Bot is a holding observer and compliance service that utilizes artificial intelligence algorithms to detect dubious activity. Advantages:
– Deal observation and identity validation.
– Accessible via internet and Telegram.
– Supports digital assets like BSC, BTC, DOGE, and additional.
5. AlfaBit
Overview: AlfaBit offers comprehensive Anti-Money Laundering (AML) services specifically made for the cryptocurrency field, supporting firms and banks in ensuring compliance conformity. Advantages:
– Comprehensive compliance features and screenings.
– Meets modern safety and conformity guidelines.
6. AMLNode
Overview: AML Node delivers AML and customer identity solutions for digital currency companies, which includes deal monitoring, sanctions checks, and risk assessment. Features:
– Danger analysis options and compliance screenings.
– Useful for maintaining safe business processes.
7. Btrace.io
Summary: Btrace.AMLcrypto.io specializes in asset verification, delivering transfer observation, sanctions screenings, and help if you are a target of theft. Advantages:
– Reliable support for asset retrieval.
– Transaction observation and safety tools.
Specialized USDT Verification Services
Our website also evaluates various sites providing check tools for USDT deals and holdings:
– **USDT TRC20 and ERC20 Verification:** Many sites support thorough evaluations for USDT transactions, aiding in the finding of questionable activity.
– **AML Verification for USDT:** Options are available for monitoring for money laundering activities.
– **“Cleanliness” Validation for Holdings:** Verification of deal and holding “cleanliness” is provided to find potential threats.
**Summary**
Selecting the suitable service for validating and observing digital currency transfers is crucial for guaranteeing protection and compliance compliance. By consulting our recommendations, you can select the most suitable solution for transfer observation and fund security.
profi-teh-remont
November 16, 2024 @ 8:56 am
Сервисный центр предлагает ремонт samsung galaxy j2 2018 в петербурге ремонт samsung galaxy j2 2018 цены
BradleyJopay
November 16, 2024 @ 11:48 am
http://indianpharmacyeasy.com/# online shopping pharmacy india
купить сейф для охотничьего ружья
November 16, 2024 @ 12:06 pm
Тут можно преобрести оружейный сейф цена сейф для оружие
aloetopakistan
November 16, 2024 @ 1:52 pm
Experience high-quality gaming with 888Starz Pakistan and access top bonuses.
Glennlashy
November 16, 2024 @ 2:27 pm
drugs prices [url=http://canadiandrugsgate.com/#]Canadian pharmacy online[/url] otc ed drugs
sugar defender official website
November 16, 2024 @ 3:50 pm
sugar defender official website As a person who’s constantly been cautious concerning
my blood glucose, finding Sugar Defender has actually been a relief.
I really feel a lot a lot more in control, and my recent exams
have revealed positive improvements. Understanding I have a trustworthy supplement to
support my routine gives me assurance. I’m so happy for Sugar Defender’s impact on my
wellness!
BradleyJopay
November 16, 2024 @ 9:50 pm
http://canadiandrugsgate.com/# ed dysfunction
Jackson Martinez
November 16, 2024 @ 9:50 pm
Hello everyone, the content on this website is really amazing for people’s experience. I hope you keep up the good work.
Larryglops
November 16, 2024 @ 11:24 pm
medicine in mexico pharmacies: mexican pharmacy online – best online pharmacies in mexico
сейф для оружия
November 17, 2024 @ 7:24 am
Тут можно преобрести купить оружейный сейф для пистолета охотничьи сейфы
огнестойкие сейфы
November 17, 2024 @ 7:38 am
Тут можно преобрести сейф огнеупорный купить сейф огнестойкий в москве
BradleyJopay
November 17, 2024 @ 7:49 am
http://canadiandrugsgate.com/# cheap online pharmacy
Larryglops
November 17, 2024 @ 12:09 pm
pain medications without a prescription: Canada pharmacy – cheap ed medication
JefferyHot
November 17, 2024 @ 1:12 pm
п»їbest mexican online pharmacies: mexican pharmacy online medications – pharmacies in mexico that ship to usa
сейф
November 17, 2024 @ 4:06 pm
Здесь можно преобрести купить сейф интернете купить сейф
joker123
November 17, 2024 @ 4:28 pm
Superb post however I was wondering if you could write a litte more on this
topic? I’d be very thankful if you could
elaborate a little bit further. Many thanks!
BradleyJopay
November 17, 2024 @ 5:50 pm
https://canadiandrugsgate.com/# legal to buy prescription drugs without prescription
indirim hesaplama
November 17, 2024 @ 11:56 pm
It is a great website and a nice share.
ihow much does it cost to start a skateshop
November 18, 2024 @ 2:28 am
ihow much does it cost to start a skateshop
is instagram good for storing photos and memories
November 18, 2024 @ 3:28 am
is instagram good for storing photos and memories
love poems that metaphors in them
November 18, 2024 @ 3:54 am
love poems that metaphors in them
Stevenjag
November 18, 2024 @ 4:23 am
30mg prednisone: generic Prednisone – order prednisone
past lives and trine aspects
November 18, 2024 @ 5:23 am
past lives and trine aspects
BrandenJew
November 18, 2024 @ 5:52 am
prednisone tabs 20 mg: raypharm – prednisone 60 mg tablet
people love selectivity and exclusivity.
November 18, 2024 @ 5:54 am
people love selectivity and exclusivity.
planet meaning past lives astrology
November 18, 2024 @ 6:36 am
planet meaning past lives astrology
planet meaning past lives astrology
November 18, 2024 @ 7:14 am
planet relationship past lives astrology
descargar1xslots
November 18, 2024 @ 9:01 am
Juega en 1xslots casino y disfruta de una experiencia de casino completa.
MetaMask Extension
November 18, 2024 @ 10:16 am
Good. https://docs.webstore.guru/
сейф для оружия
November 18, 2024 @ 12:40 pm
Тут можно преобрести сейфы для оружия москва оружейные сейфы и шкафы для охотничьего ружья
Casibom
November 18, 2024 @ 1:33 pm
Thanks. https://casibom.agency/
Dereksleen
November 18, 2024 @ 2:15 pm
amoxicillin online without prescription: Amoxicillin Com Pharm – amoxil generic
Stevenjag
November 18, 2024 @ 3:03 pm
cost cheap clomid: rex pharm – can i purchase clomid tablets
степени варикоцеле
November 18, 2024 @ 4:27 pm
Узнай все о варикоцеле 1 степени варикоцеле 2 степени
BrandenJew
November 18, 2024 @ 4:56 pm
buy amoxicillin from canada: buy amoxil online – amoxicillin 500 mg tablet price
BradleyJopay
November 18, 2024 @ 6:47 pm
buying prednisone [url=https://prednisoneraypharm.com/#]ray pharm[/url] purchase prednisone
Casibom
November 18, 2024 @ 7:11 pm
Thanks. https://casiboms.it.com/
Stevenjag
November 19, 2024 @ 2:39 am
amoxicillin capsule 500mg price: buy amoxil online – amoxicillin buy online canada
prayer after dream
November 19, 2024 @ 4:28 am
prayer after dream
BrandenJew
November 19, 2024 @ 4:39 am
prednisone 20mg: cheap prednisone – can i buy prednisone online without prescription
settling or ungrateful
November 19, 2024 @ 5:15 am
settling or ungrateful
BradleyJopay
November 19, 2024 @ 6:00 am
amoxicillin price canada [url=http://amoxilcompharm.com/#]com pharm[/url] amoxicillin 500 mg where to buy
варикоцеле у мужчин
November 19, 2024 @ 6:02 am
Узнай все о варикоцеле диагностика варикоцеле 3 степени
sun rising from the horizon wording
November 19, 2024 @ 6:30 am
sun rising from the horizon wording
Telegram中文
November 19, 2024 @ 7:00 am
Telegram provides an excellent platform for researchers to share breakthrough findings like this! The discovery that nematodes produce cytokinin to hijack plant root cells is groundbreaking and could open new doors for combating agricultural losses caused by these parasites. It’s fascinating how these tiny worms manipulate plant hormones in such a sophisticated way.
Telegram
November 19, 2024 @ 7:01 am
Telegram groups are a great way for plant scientists to discuss studies like this! The revelation that an animal can synthesize cytokinin to facilitate parasitism is a unique discovery. It underscores the complex interactions between parasites and hosts, offering hope for innovative solutions to protect crops like soybeans from nematode damage.
Telegram中文版下载
November 19, 2024 @ 7:02 am
Telegram channels dedicated to plant science would find this research intriguing. Understanding how nematodes exploit cytokinin signaling to create feeding sites is crucial for developing resistant crops. The interdisciplinary collaboration between MU and the University of Bonn highlights the power of teamwork in addressing global agricultural challenges.
Michaelbip
November 19, 2024 @ 7:16 am
Фантастический игровой сайт игорный дом Лев дарит для вас незабываемые опции для ярких побед!
На площадке игрового дома Лев вас радуют удивительные акции и богатый выбор слотов. Только здесь вы откроете для себя выиграть невероятные призы с легкостью и удовольствием!
Наше казино предлагает регулярные акции, которые поднимут ваш игровой процесс. Примите участие в лотереях, чтобы выиграть.
Казино Лев обеспечивает простой и удобный интерфейс, обеспечивает легкость использования на смартфоне, планшете или ПК.
На игровом доме Лев вас встретят не только рулетка и покер, но и живые игры, которые обеспечат вам насладиться реальным игровым процессом прямо у себя дома.
Кроме того, наш кэшбэк помогут вам бонусные кредиты каждый день.
Играй с наслаждением, побеждай с Lev, и завоевывай богатства каждый день!
На платформе Lev вас ждут щедрые бонусы и огромный выбор игровых автоматов. Только здесь вы получите возможность выиграть крупные выигрыши.
Зарегистрируйтесь на платформе Лев и погрузитесь в азарт [url=https://byteschool.ru/igrovie-avtomati/]игровые автоматы[/url] уже без промедлений! [url=https://byteschool.ru/]казино лев[/url], [url=https://byteschool.ru/faq/]faq[/url], [url=https://byteschool.ru/rules/]правила[/url], [url=https://byteschool.ru/bonus/]бонус[/url]
казино лев 2024
Michaelbip
November 19, 2024 @ 10:33 am
Фантастический игровой сайт игорный дом Лев предлагает для вас захватывающие шансы для успешной игры!
На нашей платформе Lev вас ожидают акционные игры и множество игровых автоматов. Всегда здесь вы откроете для себя завоевать огромные суммы с легкостью и удовольствием!
Площадка Лев предлагает ежедневные бонусы, которые поднимут ваш опыт игры. Примите участие в турнирах, чтобы оказаться среди лидеров.
Наш сайт предлагает эргономичный интерфейс, обеспечивает легкость использования на смартфоне, планшете или ПК.
На Лев вас встретят не только слоты, но и столы с живыми дилерами, которые позволят вам испытать настоящее казино в любое время.
Кроме того, наш программа лояльности порадуют вас дополнительные средства на каждом шагу.
Играй с азартом, побеждай с игровым домом Лев, и получай признания каждый день!
На площадке Lev вас ожидают щедрые бонусы и разнообразные игры. Каждый раз здесь вы успеете завоевать колоссальные суммы.
Зарегистрируйтесь на платформе Лев и начните побеждать [url=https://cementm.ru/igrovie-avtomati/]игровые автоматы[/url] уже прямо сейчас! [url=https://cementm.ru/]казино лев[/url], [url=https://cementm.ru/faq/]faq[/url], [url=https://cementm.ru/rules/]правила[/url], [url=https://cementm.ru/bonus/]бонус[/url]
казино лев 2024
Сервисный центр Xiaomi
November 19, 2024 @ 11:31 am
Предлагаем услуги профессиональных инженеров офицальной мастерской.
Еслли вы искали сервисный центр xiaomi, можете посмотреть на сайте: сервисный центр xiaomi
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Stevenjag
November 19, 2024 @ 1:31 pm
buy priligy: Priligy tablets – cheap priligy
Michaelbip
November 19, 2024 @ 1:45 pm
Лучший игровой сайт Lev предоставляет для вас невероятные шансы для крупных выигрышей!
На онлайн-казино игрового дома Лев вас ожидают щедрые бонусы и богатый выбор слотов. Всегда здесь вы откроете для себя завоевать невероятные призы с легкостью и удовольствием!
Площадка Лев предлагает регулярные акции, которые увеличат ваш игровой процесс. Примите участие в игровых мероприятиях, чтобы выиграть.
Казино Лев предлагает эргономичный интерфейс, обеспечивает легкость использования на вашем мобильном телефоне.
На игровом доме Лев вас приветствуют не только видеослоты, но и живые дилеры, которые дадут вам возможность ощутить азарт вживую из любой точки мира.
Кроме того, наш щедрый бонусный план помогут вам дополнительные шансы на выигрыш на каждом шагу.
Играй с фантастическим азартом, побеждай с Lev, и завоевывай признания каждый день!
На площадке Lev вас радуют выгодные предложения и разнообразные игры. Всегда здесь вы откроете выиграть множество призов.
Зарегистрируйтесь на игровом доме Лев и начните побеждать [url=https://www.wellkhomeimmobilier.fr/igrovie-avtomati/]игровые автоматы[/url] уже сейчас! [url=https://www.wellkhomeimmobilier.fr/]казино лев[/url], [url=https://www.wellkhomeimmobilier.fr/faq/]faq[/url], [url=https://www.wellkhomeimmobilier.fr/rules/]правила[/url], [url=https://www.wellkhomeimmobilier.fr/bonus/]бонус[/url]
казино лев 2024
BrandenJew
November 19, 2024 @ 3:48 pm
priligy: cheap priligy – Priligy tablets
MetaMask Extension
November 19, 2024 @ 4:36 pm
Thanks. https://extensions.work/
BradleyJopay
November 19, 2024 @ 4:54 pm
rx clomid [url=http://clomidrexpharm.com/#]rex pharm[/url] where can i get clomid tablets
варикоцеле
November 19, 2024 @ 9:00 pm
Узнай все о варикоцеле 2 степени варикоцеле 1 степени
BrandenJew
November 20, 2024 @ 1:55 am
dapoxetine online: priligy – buy priligy
купить сейф для охотничьего ружья
November 20, 2024 @ 1:56 am
Тут можно преобрести оружейный сейф цена ящик для оружия купить
vincent van gogh famous failures
November 20, 2024 @ 2:42 am
vincent van gogh famous failures
what are some stereotypes about vietnamese nail salons
November 20, 2024 @ 3:07 am
what are some stereotypes about vietnamese nail salons
what ever happened to blogging for fun
November 20, 2024 @ 3:40 am
what ever happened to blogging for fun
can rabbits eat grapes
November 20, 2024 @ 4:28 am
can rabbits eat grapes
can rabbits eat grapes
November 20, 2024 @ 4:43 am
can rabbits eat grapes
why are autistic people creepy
November 20, 2024 @ 4:57 am
why are autistic people creepy
geek bar flavors ranked
November 20, 2024 @ 5:42 am
geek bar flavors ranked
GeorgeMoiny
November 20, 2024 @ 6:11 am
онлайн курсы по охране труда москва курсы по охране труда дистанционное обучение в москве
can be applied to limerence fans
November 20, 2024 @ 7:07 am
can be applied to limerence fans
GeorgeJat
November 20, 2024 @ 7:52 am
items4games
Discover a Universe of Gaming Chances with ItemsforGames
At Items4Play, we deliver a active marketplace for gamers to buy or sell profiles, items, and options for some of the most popular titles. Whether you’re looking to enhance your gaming inventory or interested in selling your account, our platform delivers a easy, safe, and rewarding journey.
Reasons to Select Items4Play?
**Extensive Title Catalog**: Browse a large selection of titles, from action-packed games such as Battleground and War Call to captivating RPGs such as ARK: Survival Evolved and Genshin Impact. We have everything, guaranteeing no player is excluded.
**Range of Services**: Our products include game account acquisitions, credits, exclusive goods, milestones, and mentoring sessions. If you want guidance gaining levels or obtaining exclusive rewards, we’ve got you covered.
**Ease of Use**: Browse easily through our systematized platform, arranged by order to find exactly the item you need fast.
**Protected Deals**: We prioritize your safety. All exchanges on our platform are conducted with the utmost safeguarding to protect your confidential and financial information.
**Standouts from Our Offerings**
– **Adventure and Survival**: Games ARK and Survival Day let you enter exciting settings with high-quality gear and passes available.
– **Adventure and Questing**: Elevate your experience in games such as Clash Royale and Wonders Age with virtual money and options.
– **Professional Play**: For eSports enthusiasts, enhance your skills with coaching and profile boosts for Val, Dota, and LoL.
**A Marketplace Designed for Players**
Backed by Apex Technologies, a established business officially recognized in Kazakhstan, Items4Games is a market where playtime dreams become real. From purchasing advance passes for the latest titles to locating hard-to-find in-game treasures, our marketplace fulfills every gaming requirement with professionalism and efficiency.
Become part of the community right away and upgrade your gaming play!
For questions or guidance, contact us at **support@items4games.com**. Let’s improve the game, as one!
GeorgeJat
November 20, 2024 @ 1:07 pm
Discover a Realm of Virtual Opportunities with ItemsforGames
At Items4Play, we provide a vibrant marketplace for players to purchase or sell gaming accounts, goods, and features for widely played games. Whether you’re seeking to enhance your gaming inventory or wanting to profit from your account, our platform delivers a seamless, protected, and valuable process.
Reasons to Choose ItemsforGames?
**Extensive Game Library**: Discover a large selection of games, from intense titles such as Battlefield and COD to captivating RPGs such as ARK: Survival Evolved and Genshin. We include everything, ensuring no gamer is excluded.
**Range of Options**: Our products include profile buys, virtual money, rare collectibles, trophies, and mentoring options. Whether you want assistance improving or getting premium rewards, we have it all.
**Ease of Use**: Explore effortlessly through our well-organized marketplace, arranged alphabetically to find precisely what you are looking for quickly.
**Safe Exchanges**: We prioritize your security. All trades on our marketplace are handled with the top protection to guard your private and monetary information.
**Key Points from Our Collection**
– **Action and Adventure**: Games ARK and Survival Day allow you to explore tough settings with premium goods and passes on offer.
– **Strategy and Questing**: Boost your experience in titles such as Clash Royale and Age of Wonders 4 with virtual money and options.
– **eSports Competitions**: For competitive players, boost your abilities with coaching and level-ups for Valored, DotA, and League of Legends.
**A Platform Built for Gamers**
Backed by ApexTech Innovations, a reliable company registered in Kazakh Republic, ItemsforGames is a market where playtime dreams come true. From purchasing advance keys for the latest games to getting rare in-game treasures, our marketplace meets every player’s requirement with expertise and speed.
Sign up for the gaming family right away and elevate your gaming play!
For questions or assistance, contact us at **support@items4games.com**. Let’s enjoy gaming, together!
Dereksleen
November 20, 2024 @ 1:45 pm
priligy max pharm: buy priligy – Priligy tablets
Casibom
November 20, 2024 @ 2:14 pm
Thanks. https://casibom.works/
MetaMask Extension
November 20, 2024 @ 9:51 pm
MetaMask Extension provides secure wallet integration, dApp connectivity, and seamless access to DeFi platforms. Start exploring Web3 today! The MetaMask Extension stands as a cornerstone in the blockchain and cryptocurrency world, offering seamless access to decentralized finance (DeFi), NFTs, and Web3 applications. https://webstore.work/
BrandenJew
November 20, 2024 @ 10:01 pm
buy dapoxetine online: priligy maxpharm – dapoxetine online
Stevenjag
November 21, 2024 @ 5:49 am
buy amoxicillin 500mg uk: cheap amoxil – where can i buy amoxicillin over the counter uk
apkportalnik
November 21, 2024 @ 8:46 am
скачать приложения казино http://sspolytechnic.co.in/prilozhenie-wix-owner-upravljajte-sajtom-s/
Darrenmow
November 21, 2024 @ 11:50 am
https://lisinopril1st.com/# buy Lisinopril online
гистероскопия удаление полипа
November 21, 2024 @ 12:25 pm
Узнай все о удалить полип в матке ценаоперация по удалению полипа в матке
Micheal Suarez
November 21, 2024 @ 12:28 pm
I am truly thankful to the owner of this web site who has shared this fantastic piece of writing at at this place.
BrandenJew
November 21, 2024 @ 1:41 pm
amoxicillin 500 mg price: amoxicillin 500 mg tablet price – antibiotic amoxicillin
Chainlist
November 21, 2024 @ 3:50 pm
Chainlist lets you easily connect to multiple blockchain networks. Simplify wallet setups, save time, and boost your crypto management! https://chainlist.cam/
한국 드라마 리뷰
November 21, 2024 @ 11:44 pm
이 글이 정말 많은 도움이 되었습니다. 감사합니다! 앞으로도 유익한 정보 많이 공유해 주세요.
한국 드라마 리뷰
Bernardwet
November 22, 2024 @ 1:21 am
plavix best price [url=http://plavixclo.com/#]Clopidogrel Best Prices[/url] cheap plavix antiplatelet drug
why does exercising self discipline tre me out
November 22, 2024 @ 3:31 am
why does exercising self discipline tre me out
wnt bible translation problems
November 22, 2024 @ 3:57 am
wnt bible translation problems
BrandenJew
November 22, 2024 @ 4:18 am
purchase amoxicillin online: amoxil – amoxicillin 500mg capsules uk
септопластика носа
November 22, 2024 @ 9:39 am
Узнай все о пластика перегородки носасептопластика в москве
카지노솔루션
November 22, 2024 @ 12:03 pm
안녕하세요 여러분, 모든 것이 잘 되어가고 계신가요? 그리고 이 글에 대해 어떻게 생각하시나요? 제 생각에는
정말로 저를 위해 디자인된 멋진 글입니다.
Burç Özellikleri
November 22, 2024 @ 1:08 pm
Burç özellikleri hakkında merak edilen her şey! Tüm burçların genel ve özel karakteristik özelliklerini keşfetmek için bu rehberi inceleyin. https://tedez.com/burc-ozellikleri/
Darrenmow
November 22, 2024 @ 1:17 pm
https://cytpremium.com/# buy cytotec online
AlbertNef
November 22, 2024 @ 3:58 pm
buy Lisinopril online: zestril 5 mg tablet – buy Lisinopril online
BrandenJew
November 22, 2024 @ 4:46 pm
buy amoxicillin online cheap: amoxil – prescription for amoxicillin
MetaMask Extension
November 22, 2024 @ 7:07 pm
Download MetaMask Extension for Chrome to securely manage your crypto wallet. Follow this easy guide to install and start using MetaMask in minutes! https://sites.google.com/view/download-metamask-extension-41/home
Bernardwet
November 22, 2024 @ 8:31 pm
buy plavix [url=https://plavixclo.com/#]buy plavix online[/url] buy clopidogrel bisulfate
hornyxxx.win/hornyvid844536313
November 22, 2024 @ 11:09 pm
I ave beenn surfing online more than three hours today, yet I never ound anny
interestinng arrticle likke yours. It’s prety wortgh enough for me.
In my opinion, iff all webmazsters and bbloggers mawde goold content as you did, the
internet wikl bbe muhh moree useful tan ver before.
GeorgeJat
November 23, 2024 @ 4:03 am
Discover a World of Video Game Possibilities with Items4Games
At ItemsforGames, we provide a vibrant hub for enthusiasts to acquire or sell accounts, virtual items, and options for widely played video games. Whether you’re looking to upgrade your game arsenal or wanting to profit from your profile, our service delivers a seamless, protected, and valuable journey.
Why Choose Items4Games?
**Extensive Game Catalog**: Discover a extensive array of games, from thrilling titles like Battlefield and War Call to captivating adventure games such as ARK and Genshin. We have all games, making sure no enthusiast is excluded.
**Selection of Services**: Our products include account purchases, credits, exclusive goods, achievements, and mentoring options. If you want guidance gaining levels or obtaining exclusive rewards, we have it all.
**Ease of Use**: Navigate effortlessly through our well-organized site, arranged by order to locate just the item you need fast.
**Secure Exchanges**: We focus on your safety. All transactions on our site are processed with the top safeguarding to guard your private and financial details.
**Key Points from Our Collection**
– **Survival and Exploration**: Games like ARK and Day-Z give you the chance to explore tough environments with premium items and accesses available.
– **Adventure and Exploration**: Boost your performance in adventures like Clash Royale and Age of Wonders 4 with in-game currencies and features.
– **eSports Play**: For competitive fans, improve your technique with coaching and level-ups for Val, Dota 2, and League of Legends.
**A Hub Designed for Fans**
Supported by Apex Technologies, a trusted company officially recognized in Kazakh Republic, Items4Games is a place where gaming aspirations are realized. From buying advance passes for the latest titles to finding rare game items, our site caters to every gamer’s requirement with skill and reliability.
Join the community today and boost your gaming play!
For support or guidance, email us at **support@items4games.com**. Let’s game better, together!
BrandenJew
November 23, 2024 @ 5:01 am
amoxicillin 500 mg price: Amoxicillin for sale – buy amoxicillin without prescription
Darrenmow
November 23, 2024 @ 5:28 am
https://cytpremium.com/# Cytotec 200mcg price
увеличение полового члена
November 23, 2024 @ 9:08 am
Узнай все о увеличение размера полового члена увеличение полового члена в москве
Online Marketing Social Media
November 23, 2024 @ 10:53 am
Hey there! Just wanted dropping by to inform you how much I admire your blog. Your expertise on making money online are genuinely noteworthy. Earning an income from home has never been easier thanks to affiliate promotion. It’s all about identifying the perfect products to promote and establishing connections with your audience. Your blog is a treasure trove of information for emerging online entrepreneurs. Keep on the great work!
드라마 다시보기
November 23, 2024 @ 2:35 pm
저는 그때마다 작은 기사나 리뷰를 읽곤 했는데, 그 기사나 리뷰는
그들의 동기를 분명히 했고, 지금 읽고 있는 이
게시물도 마찬가지입니다.
Bernardwet
November 23, 2024 @ 3:26 pm
plavix best price [url=https://plavixclo.com/#]PlavixClo[/url] Plavix generic price
Affiliate Marketing Amazon
November 23, 2024 @ 4:28 pm
Hey there! Just wanted to remind you that every day is a new chance to chase your aspirations and make them a reality. Keep moving forward and never give up!
BrandenJew
November 23, 2024 @ 6:15 pm
buy clomid pills: cheap clomid – order generic clomid without rx
xxxtubebest.com/xp15a11fK1Vbk
November 23, 2024 @ 6:26 pm
Why userrs still maske use off to ead news papers when inn this technoloogical world aall
is existing on web?
Darrenmow
November 23, 2024 @ 10:42 pm
https://lisinopril1st.com/# lisinopril 40 mg india
Frankcix
November 24, 2024 @ 1:26 am
dj88 slot login
MetaMask Extension
November 24, 2024 @ 4:12 am
Download MetaMask extension today for secure crypto wallet access. Follow our step-by-step guide to install it effortlessly on your browser. https://extension.wtf/
Сервисный центр LG Москва
November 24, 2024 @ 6:06 am
Предлагаем услуги профессиональных инженеров офицальной мастерской.
Еслли вы искали сервисный центр lg, можете посмотреть на сайте: официальный сервисный центр lg
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Jeffry Tisa
November 24, 2024 @ 8:15 am
Merci j’ai appris beaucoup avec ton article 🙂 !
BrandenJew
November 24, 2024 @ 8:18 am
how to buy amoxicillin online: amoxil com pharm – amoxicillin medicine over the counter
카지노솔루션
November 24, 2024 @ 10:49 am
귀하의 게시물을 읽는 것을 즐깁니다. 귀하를 지원하기 위해 짧은 댓글을 쓰고 싶습니다. 이렇게 좋은 아이디어를 공유해 주셔서 감사합니다.
MetaMask Extension
November 24, 2024 @ 11:14 am
MetaMask is a browser extension and mobile application designed to simplify the interaction between users and blockchain-based applications (dApps). https://sites.google.com/view/metamask-extension-guide-39/home
Bernardwet
November 24, 2024 @ 12:49 pm
buy minocycline 50 mg for humans [url=https://iverfast.com/#]IverFast[/url] ivermectin generic cream
Affiliate Marketing Motivation Quotes
November 24, 2024 @ 2:31 pm
Hi friends! Embrace the path of self-discovery and self-improvement. Each step brings you closer to revealing your true potential.
Darrenmow
November 24, 2024 @ 3:04 pm
https://lisinopril1st.com/# buy Lisinopril 1st
bokep
November 24, 2024 @ 6:55 pm
Hi there, I want to subscribe for this webpage to get most up-to-date updates, thus where can i do it please assist.
Personalized keto meal guide 2024
November 24, 2024 @ 7:19 pm
Hello beautiful souls! Trust in the wonders of new beginnings. Each day is a fresh start.
Keto weight loss success stories
November 25, 2024 @ 2:09 am
Hi friends! Grasp opportunities to feed your soul and refresh your spirit. Self-love is crucial for peace of mind.
Dariodig
November 25, 2024 @ 4:09 am
вавада казино зеркало: вавада казино онлайн – вавада казино зеркало
Josephneach
November 25, 2024 @ 6:23 am
https://pinup-kazi.kz/# пин ап казино
Free customer growth
November 25, 2024 @ 6:35 am
Hello beautiful souls! Take a moment today to appreciate how far you’ve come on your journey. You are capable of accomplishing greatness, so keep shining bright!
USA Pro Leggings
November 25, 2024 @ 7:45 am
USA Pro Leggings
Сервисный центр Huawei
November 25, 2024 @ 7:45 am
Предлагаем услуги профессиональных инженеров офицальной мастерской.
Еслли вы искали сервисный центр huawei, можете посмотреть на сайте: сервисный центр huawei
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Dariodig
November 25, 2024 @ 10:06 am
pinup kazi: pin up казино – pinup
CRM software free
November 25, 2024 @ 12:19 pm
Hey there! Believe in your inner strength. You have the boldness to overcome any difficulty.
Jamesinfup
November 25, 2024 @ 1:04 pm
пин ап вход: pinup – пинап казино
WilliamHaith
November 25, 2024 @ 4:06 pm
THA娛樂城與九州娛樂簡介
THA娛樂城是九州娛樂旗下的一個知名線上娛樂平台,致力於為玩家提供多元化且優質的娛樂服務。九州娛樂是亞洲地區領先的線上娛樂品牌,長期專注於打造穩定、公平且充滿樂趣的遊戲環境,旗下擁有多個知名娛樂平台,而THA娛樂城便是其中的一個旗艦產品。以下將詳細介紹THA娛樂城的特色與服務。
THA娛樂城的遊戲種類
THA娛樂城為玩家提供了多樣化的遊戲選擇,滿足不同玩家的需求與興趣,包括以下幾大類別:
1. 體育投注
體育迷可以在THA娛樂城享受豐富的體育賽事投注,涵蓋足球、籃球、網球等多項國際體育比賽。平台提供即時賠率和多種投注選項,讓玩家隨時掌握比賽動態並進行下注。
2. 真人娛樂
真人娛樂區提供即時互動的遊戲體驗,玩家可透過直播技術與真人荷官互動,遊玩如百家樂、輪盤、骰寶等經典遊戲。這不僅增添了真實感,更讓玩家有身臨其境的感受。
3. 電子老虎機
THA娛樂城匯集了多款高人氣的電子老虎機遊戲,從經典水果機到新潮主題機,皆配備精美的畫面與流暢的操作介面,為玩家帶來無限樂趣。
4. 捕魚遊戲
捕魚遊戲是許多玩家的最愛,THA娛樂城提供多種精美場景和豐富玩法的捕魚遊戲,玩家可以挑戰高額獎勵,享受射擊與策略結合的獨特樂趣。
5. 彩票與其他遊戲
除了以上幾類,平台還提供彩票遊戲及其他創新型娛樂遊戲,適合喜歡嘗試新鮮玩法的玩家。
九州娛樂的技術支持
作為THA娛樂城的母公司,九州娛樂以其強大的技術實力與嚴謹的管理體系為平台提供支持。九州娛樂採用國際領先的安全加密技術,確保玩家的個人資訊與交易數據得到妥善保護。此外,平台的遊戲結果皆經過嚴格的隨機性測試與第三方機構認證,保證公平公正。
優質的會員服務
THA娛樂城致力於打造一個高品質的會員體驗,其服務特色包括:
– 優惠活動
平台定期推出多種促銷活動,如新會員註冊禮金、存款返利、週週回饋等,讓玩家享受更多的遊戲資金。
– 快速出入金
平台提供便捷且快速的存提款服務,玩家可以安心進行資金操作,且無需擔心延遲問題。
– 24小時客戶服務
為了確保玩家的需求能即時被解決,THA娛樂城提供全年無休的客服支援,無論是遊戲問題還是技術諮詢,客服團隊都能快速回應。
與其他平台的區別
THA娛樂城之所以能在眾多線上娛樂平台中脫穎而出,不僅因其豐富的遊戲種類與高品質服務,更在於它所帶來的獨特價值:
1. 品牌信譽
作為九州娛樂旗下的品牌,THA娛樂城擁有長期積累的良好信譽,是許多玩家的首選。
2. 創新與多樣性
平台不斷推出新遊戲與創意玩法,讓玩家始終保持新鮮感。
3. 本地化服務
THA娛樂城深諳玩家需求,提供多語言支援與符合本地習慣的服務,提升玩家的使用便利性。
未來展望
隨著線上娛樂行業的快速發展,THA娛樂城也在持續進步與創新,力求為玩家帶來更多驚喜。無論是透過引進新的遊戲技術、擴展遊戲種類,還是改善服務品質,THA娛樂城都希望成為每位玩家的最佳娛樂夥伴。
總結來說,THA娛樂城憑藉其豐富的遊戲內容、可靠的技術支持以及優質的會員服務,已在亞洲娛樂市場佔有一席之地。如果您正在尋找一個結合刺激與信賴的娛樂平台,那麼THA娛樂城絕對值得一試。
Dariodig
November 25, 2024 @ 4:09 pm
vavada: вавада казино – вавада казино онлайн
Сервисный центр Philips
November 25, 2024 @ 6:23 pm
Предлагаем услуги профессиональных инженеров офицальной мастерской.
Еслли вы искали сервисный центр philips в москве, можете посмотреть на сайте: сервисный центр philips
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
HenryLix
November 25, 2024 @ 6:54 pm
pinup kazi [url=http://pinup-kazi.kz/#]пин ап кз[/url] пин ап казино онлайн
Dariodig
November 25, 2024 @ 9:55 pm
вавада казино онлайн: vavada – vavada
Dariodig
November 26, 2024 @ 3:30 am
пин ап зеркало: pinup – пин ап зеркало
9th grade freshman high school swim team
November 26, 2024 @ 4:28 am
9th grade freshman high school swim team
utility vs beauty
November 26, 2024 @ 4:45 am
utility vs beauty
Curtispoilm
November 26, 2024 @ 4:52 am
пинап казино: pinup kazi – пинап казино
behavioral questions are asking you to lie
November 26, 2024 @ 5:29 am
behavioral questions are asking you to lie
why do trolls criticize people in person
November 26, 2024 @ 5:31 am
why do trolls criticize people in person
behavioral questions are asking you to lie
November 26, 2024 @ 5:44 am
behavioral questions are asking you to lie
behavioral questions are stupid if you havent exprienced the question
November 26, 2024 @ 6:05 am
behavioral questions are stupid if you havent exprienced the question
why don't girls like the younger sibling at school
November 26, 2024 @ 6:25 am
why don’t girls like the younger sibling at school
SEM growth free
November 26, 2024 @ 6:56 am
Hi friends! Decide contentment and thankfulness as your constant companions. These feelings will light up your path.
a dad is supposed to teach his son
November 26, 2024 @ 6:56 am
a dad is supposed to teach his son
best paint for plastic
November 26, 2024 @ 7:12 am
best paint for plastic
can i call myself fullstack software engineer intern
November 26, 2024 @ 7:38 am
can i call myself fullstack software engineer intern
MetaMask Extension
November 26, 2024 @ 9:57 am
A heartfelt thank you for reading and sharing your thoughts—it’s readers like you that inspire me to keep writing.
HenryLix
November 26, 2024 @ 1:18 pm
vavada-kazi.ru [url=http://vavada-kazi.ru/#]казино вавада[/url] вавада казино
Sign up to get 100 USDT
November 26, 2024 @ 1:45 pm
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me?
Dariodig
November 26, 2024 @ 3:31 pm
pinup-kazi.kz: pinup-kazi.kz – пин ап кз
Jamesinfup
November 26, 2024 @ 4:11 pm
pinup kazi: пинап казино – пин ап казино официальный сайт
Curtispoilm
November 26, 2024 @ 6:19 pm
казино вавада: vavada – vavada kazi
Сервисный центр Apple Москва
November 26, 2024 @ 8:20 pm
Предлагаем услуги профессиональных инженеров офицальной мастерской.
Еслли вы искали сервисный центр apple, можете посмотреть на сайте: официальный сервисный центр apple
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Dariodig
November 26, 2024 @ 9:21 pm
пин ап казино онлайн: пин ап кз – пинап казино
porn
November 26, 2024 @ 11:08 pm
Hi there! I could have sworn I’ve visited this blog before but after browsing through many of the articles I realized it’s new to me. Nonetheless, I’m certainly pleased I came across it and I’ll be bookmarking it and checking back regularly.
Josephneach
November 26, 2024 @ 11:43 pm
https://pinup-kazi.kz/# pinup-kazi.kz
bad to not go out in 20s
November 27, 2024 @ 2:23 am
bad to not go out in 20s
can i use microwavable heating pad for hissing cockroach
November 27, 2024 @ 3:07 am
can i use microwavable heating pad for hissing cockroach
Dariodig
November 27, 2024 @ 3:10 am
пин ап казино официальный сайт: пин ап вход – пин ап казино официальный сайт
can rabbits have grapes
November 27, 2024 @ 4:14 am
can rabbits have grapes
choosing people to be your friends
November 27, 2024 @ 5:57 am
choosing people to be your friends
Jamesinfup
November 27, 2024 @ 6:17 am
pinup: пин ап вход – пин ап казино официальный сайт
choosing people to be your friends
November 27, 2024 @ 6:24 am
choosing people to be your friends
demonstration speech ideas
November 27, 2024 @ 7:11 am
demonstration speech ideas
demonstration speech ideas
November 27, 2024 @ 7:53 am
demonstration speech ideas
Dariodig
November 27, 2024 @ 9:21 am
vavada-kazi.ru: вавада казино – vavada
Josephneach
November 27, 2024 @ 1:13 pm
http://pinup-kazi.kz/# pinup-kazi.kz
Curtispoilm
November 27, 2024 @ 3:13 pm
вавада казино: вавада казино зеркало – вавада онлайн казино
Dariodig
November 27, 2024 @ 3:19 pm
vavada-kazi.ru: вавада казино – вавада казино зеркало
Jamesinfup
November 27, 2024 @ 4:00 pm
пинап казино: пинап казино – пин ап казино онлайн
ErnestoGer
November 27, 2024 @ 4:51 pm
THA娛樂城
THA娛樂城與九州娛樂簡介
THA娛樂城是九州娛樂旗下的一個知名線上娛樂平台,致力於為玩家提供多元化且優質的娛樂服務。九州娛樂是亞洲地區領先的線上娛樂品牌,長期專注於打造穩定、公平且充滿樂趣的遊戲環境,旗下擁有多個知名娛樂平台,而THA娛樂城便是其中的一個旗艦產品。以下將詳細介紹THA娛樂城的特色與服務。
THA娛樂城的遊戲種類
THA娛樂城為玩家提供了多樣化的遊戲選擇,滿足不同玩家的需求與興趣,包括以下幾大類別:
1. 體育投注
體育迷可以在THA娛樂城享受豐富的體育賽事投注,涵蓋足球、籃球、網球等多項國際體育比賽。平台提供即時賠率和多種投注選項,讓玩家隨時掌握比賽動態並進行下注。
2. 真人娛樂
真人娛樂區提供即時互動的遊戲體驗,玩家可透過直播技術與真人荷官互動,遊玩如百家樂、輪盤、骰寶等經典遊戲。這不僅增添了真實感,更讓玩家有身臨其境的感受。
3. 電子老虎機
THA娛樂城匯集了多款高人氣的電子老虎機遊戲,從經典水果機到新潮主題機,皆配備精美的畫面與流暢的操作介面,為玩家帶來無限樂趣。
4. 捕魚遊戲
捕魚遊戲是許多玩家的最愛,THA娛樂城提供多種精美場景和豐富玩法的捕魚遊戲,玩家可以挑戰高額獎勵,享受射擊與策略結合的獨特樂趣。
5. 彩票與其他遊戲
除了以上幾類,平台還提供彩票遊戲及其他創新型娛樂遊戲,適合喜歡嘗試新鮮玩法的玩家。
九州娛樂的技術支持
作為THA娛樂城的母公司,九州娛樂以其強大的技術實力與嚴謹的管理體系為平台提供支持。九州娛樂採用國際領先的安全加密技術,確保玩家的個人資訊與交易數據得到妥善保護。此外,平台的遊戲結果皆經過嚴格的隨機性測試與第三方機構認證,保證公平公正。
優質的會員服務
THA娛樂城致力於打造一個高品質的會員體驗,其服務特色包括:
– 優惠活動
平台定期推出多種促銷活動,如新會員註冊禮金、存款返利、週週回饋等,讓玩家享受更多的遊戲資金。
– 快速出入金
平台提供便捷且快速的存提款服務,玩家可以安心進行資金操作,且無需擔心延遲問題。
– 24小時客戶服務
為了確保玩家的需求能即時被解決,THA娛樂城提供全年無休的客服支援,無論是遊戲問題還是技術諮詢,客服團隊都能快速回應。
與其他平台的區別
THA娛樂城之所以能在眾多線上娛樂平台中脫穎而出,不僅因其豐富的遊戲種類與高品質服務,更在於它所帶來的獨特價值:
1. 品牌信譽
作為九州娛樂旗下的品牌,THA娛樂城擁有長期積累的良好信譽,是許多玩家的首選。
2. 創新與多樣性
平台不斷推出新遊戲與創意玩法,讓玩家始終保持新鮮感。
3. 本地化服務
THA娛樂城深諳玩家需求,提供多語言支援與符合本地習慣的服務,提升玩家的使用便利性。
未來展望
隨著線上娛樂行業的快速發展,THA娛樂城也在持續進步與創新,力求為玩家帶來更多驚喜。無論是透過引進新的遊戲技術、擴展遊戲種類,還是改善服務品質,THA娛樂城都希望成為每位玩家的最佳娛樂夥伴。
總結來說,THA娛樂城憑藉其豐富的遊戲內容、可靠的技術支持以及優質的會員服務,已在亞洲娛樂市場佔有一席之地。如果您正在尋找一個結合刺激與信賴的娛樂平台,那麼THA娛樂城絕對值得一試。
Georgeenuff
November 27, 2024 @ 5:19 pm
кто проводит спецоценку условий труда москва сделать соут в москве
does doom metal help trauma victims
November 28, 2024 @ 1:33 am
social media girls forums
do hissing cockroaches like sugar
November 28, 2024 @ 2:38 am
do hissing cockroaches like sugar
ScottLiawl
November 28, 2024 @ 3:12 am
Online medicine order: indian pharm – Online medicine home delivery
do the cruson ansestors practice witchcraft
November 28, 2024 @ 3:25 am
do the cruson ansestors practice witchcraft
heaven turned hell and hell turned heaven
November 28, 2024 @ 5:16 am
heaven turned hell and hell turned heaven
FrankNidly
November 28, 2024 @ 5:17 am
otc ed drugs http://mexicanpharmeasy.com/# mexican online pharmacies prescription drugs
how does ambitious go with literature
November 28, 2024 @ 5:49 am
how does ambitious go with literature
how to compliment a craftsman
November 28, 2024 @ 6:17 am
how to compliment a craftsman
how to compliment a craftsman
November 28, 2024 @ 6:19 am
how to compliment a craftsman
ernest hemingway implied meaning
November 28, 2024 @ 7:19 am
ernest hemingway implied meaning
Lutheravami
November 28, 2024 @ 7:31 am
메인 서비스: 간편하고 효율적인 배송 및 구매 대행 서비스
1. 대행 서비스 주요 기능
메인 서비스는 고객이 한 번에 필요한 대행 서비스를 신청할 수 있도록 다양한 기능을 제공합니다.
배송대행 신청: 국내외 상품 배송을 대신 처리하며, 효율적인 시스템으로 신속한 배송을 보장합니다.
구매대행 신청: 원하는 상품을 대신 구매해주는 서비스로, 고객의 수고를 줄입니다.
엑셀 대량 등록: 대량 상품을 엑셀로 손쉽게 등록 가능하여 상업 고객의 편의성을 증대합니다.
재고 관리 신청: 창고 보관 및 재고 관리를 통해 물류 과정을 최적화합니다.
2. 고객 지원 시스템
메인 서비스는 사용자 친화적인 접근성을 제공합니다.
유저 가이드: 대행 서비스를 더욱 합리적으로 사용할 수 있도록 세부 안내서를 제공합니다.
운송장 조회: 일본 사가와 등 주요 운송사의 추적 시스템과 연동하여 운송 상황을 실시간으로 확인 가능합니다.
3. 비용 안내와 부가 서비스
비용 계산기: 예상되는 비용을 간편하게 계산해 예산 관리를 돕습니다.
부가 서비스: 교환 및 반품, 폐기 및 검역 지원 등 추가적인 편의 서비스를 제공합니다.
출항 스케줄 확인: 해외 배송의 경우 출항 일정을 사전에 확인 가능하여 배송 계획을 세울 수 있습니다.
4. 공지사항
기본 검수 공지
무료 검수 서비스로 고객의 부담을 줄이며, 보다 철저한 검수가 필요한 경우 유료 정밀 검수 서비스를 권장합니다.
수출허가서 발급 안내
항공과 해운 수출 건에 대한 허가서를 효율적으로 발급받는 방법을 상세히 안내하며, 고객의 요청에 따라 이메일로 전달됩니다.
노데이터 처리 안내
운송장 번호 없는 주문에 대한 새로운 처리 방안을 도입하여, 노데이터 발생 시 관리비가 부과되지만 서비스 품질을 개선합니다.
5. 고객과의 소통
카카오톡 상담: 실시간 상담을 통해 고객의 궁금증을 해결합니다.
공지사항 알림: 서비스 이용 중 필수 정보를 지속적으로 업데이트합니다.
메인 서비스는 고객 만족을 최우선으로 하며, 지속적인 개선과 세심한 관리를 통해 최상의 경험을 제공합니다.
Scottpyday
November 28, 2024 @ 9:04 am
KU娛樂城與九州娛樂簡介
KU娛樂城是九州娛樂旗下的一個知名線上娛樂平台,致力於為玩家提供安全、創新且多元化的娛樂服務。九州娛樂作為亞洲地區的線上娛樂領導品牌,以其穩定的運營、先進的技術和用戶至上的服務理念聞名於業界。KU娛樂城在九州娛樂的支持下,不僅具備強大的品牌背景,還融入了創新的遊戲設計和完善的會員體系,成為眾多玩家的理想選擇。
KU娛樂城的遊戲種類
KU娛樂城為玩家提供了豐富多樣的遊戲選項,涵蓋多個熱門類別,旨在滿足不同類型玩家的娛樂需求:
1. 體育投注
KU娛樂城提供全面的體育賽事投注服務,覆蓋足球、籃球、棒球等多項主流運動。玩家可以根據實時賠率和賽事數據進行投注,享受觀賽與下注相結合的刺激體驗。
2. 真人娛樂
在真人娛樂區,玩家可以參與百家樂、輪盤、德州撲克等經典遊戲,並與真人荷官進行即時互動。先進的直播技術確保遊戲過程流暢無延遲,讓玩家彷彿置身於真實賭場。
3. 電子老虎機
KU娛樂城集合了數百款精美設計的電子老虎機遊戲,無論是經典三軸機還是具備豐富特效的多軸機,皆可滿足玩家的需求。高額的獎金機制與趣味主題設計,讓每一次旋轉都充滿驚喜。
4. 捕魚遊戲
KU娛樂城的捕魚遊戲區擁有多種玩法,玩家可以透過射擊技巧和策略挑戰不同級別的魚類,贏取豐厚的獎勵。畫面精美的海底場景和豐富的道具選擇,讓捕魚遊戲成為平台上的人氣項目。
5. 彩票與其他遊戲
KU娛樂城還提供豐富的彩票遊戲和創新娛樂玩法,玩家可以選擇自己喜好的遊戲類型,盡享多樣化的娛樂體驗。
九州娛樂的技術支持
九州娛樂作為KU娛樂城的母公司,以其先進的技術實力和多年的行業經驗為平台提供堅實的支持。九州娛樂引入國際頂級的數據加密技術,確保用戶的個人資訊和交易數據的安全性。同時,遊戲結果均經過公平性測試和第三方機構認證,為玩家打造一個公平透明的娛樂環境。
優質的會員服務
KU娛樂城始終以玩家為中心,提供一系列貼心的服務與福利:
– 優惠活動
平台定期推出豐富的促銷活動,包括新會員首存禮金、充值返利以及抽獎活動,讓玩家的每一次參與都更有價值。
– 快速出入金
KU娛樂城支持多種主流支付方式,並保證存提款的快速處理,讓玩家無需等待即可享受遊戲樂趣。
– 全天候客戶服務
KU娛樂城提供24小時在線客服支援,隨時解答玩家的疑問,確保每位玩家都能獲得最好的服務體驗。
KU娛樂城的競爭優勢
KU娛樂城之所以能在眾多線上娛樂平台中脫穎而出,除了其豐富的遊戲種類與頂級服務外,還有以下幾個顯著的競爭優勢:
1. 品牌信譽
作為九州娛樂旗下的品牌,KU娛樂城憑藉多年的穩定運營,已成為亞洲玩家心目中值得信賴的娛樂平台。
2. 持續創新
KU娛樂城不斷推出新遊戲和新功能,為玩家帶來更多元化和現代化的娛樂體驗。
3. 本地化服務
平台針對不同地區的玩家提供本地化的界面與支付選項,提升了用戶的便利性與滿意度。
未來展望
KU娛樂城將繼續秉承九州娛樂的核心理念,致力於成為亞洲線上娛樂市場的佼佼者。無論是透過升級遊戲技術、推出更多創新玩法,還是提升服務品質,KU娛樂城都希望為玩家帶來更加豐富的娛樂體驗。
總而言之,KU娛樂城在九州娛樂的支持下,憑藉其出色的遊戲內容和優質的服務,已成為線上娛樂行業中的一顆璀璨明珠。如果您正在尋找一個結合信譽、創新與趣味的娛樂平台,KU娛樂城將是不二之選。
ScottLiawl
November 28, 2024 @ 4:21 pm
purple pharmacy mexico price list: mexican pharm easy – pharmacies in mexico that ship to usa
AdolfoSmobe
November 28, 2024 @ 5:50 pm
reputable mexican pharmacies online: Pharm Easy – best online pharmacies in mexico
CarlosTib
November 28, 2024 @ 7:40 pm
ed remedies that really work: canadian pharm 1st – the canadian drugstore
Suzi Kadelak
November 28, 2024 @ 7:50 pm
URL News 7
AdolfoSmobe
November 28, 2024 @ 11:46 pm
cheap drugs: canadian pharmacy – buy prescription drugs online legally
ScottLiawl
November 29, 2024 @ 1:51 am
buy medicines online in india: indian pharmacy – buy medicines online in india
Jordon Haapala
November 29, 2024 @ 3:49 am
https://bzmos.hashnode.dev/pornhun-live
ernest hemingway implied meaning
November 29, 2024 @ 4:19 am
ernest hemingway implied meaning
can you condem people to hell
November 29, 2024 @ 4:39 am
can you condem people to hell
cctv monitor side reference
November 29, 2024 @ 5:28 am
cctv monitor side reference
AdolfoSmobe
November 29, 2024 @ 5:52 am
online shopping pharmacy india: indian pharm – reputable indian online pharmacy
computation and data science gmu vs data analytics engineering gmu
November 29, 2024 @ 5:53 am
computation and data science gmu vs data analytics engineering gmu
demon slayer book summary back
November 29, 2024 @ 6:11 am
demon slayer book summary back
does cvs sell postage stamps
November 29, 2024 @ 6:39 am
does cvs sell postage stamps
CarlosTib
November 29, 2024 @ 9:40 am
mail order pharmacy india: indian pharmacy – best india pharmacy
Haiden Gardner
November 29, 2024 @ 10:36 am
https://twitter.com/NeginSolan91252
ScottLiawl
November 29, 2024 @ 11:58 am
top online pharmacy india: indian pharmacy – best india pharmacy
AdolfoSmobe
November 29, 2024 @ 12:05 pm
indian pharmacy: IndianPharmStar – world pharmacy india
FrankNidly
November 29, 2024 @ 1:10 pm
mens ed pills https://indianpharmstar.com/# cheapest online pharmacy india
Michaelten
November 29, 2024 @ 3:15 pm
일본소비세환급
일본 소비세 환급, 네오리아와 함께라면 간편하고 안전하게
일본 소비세 환급은 복잡하고 까다로운 절차로 많은 구매대행 셀러들이 어려움을 겪는 분야입니다. 네오리아는 다년간의 경험과 전문성을 바탕으로 신뢰할 수 있는 서비스를 제공하며, 일본 소비세 환급 과정을 쉽고 효율적으로 처리합니다.
1. 일본 소비세 환급의 필요성과 네오리아의 역할
네오리아는 일본 현지 법인을 설립하지 않아도 합법적인 방식으로 소비세 환급을 받을 수 있는 솔루션을 제공합니다. 이를 통해:
한국 개인사업자와 법인 사업자 모두 간편하게 환급 절차를 진행할 수 있습니다.
일본의 복잡한 서류 심사를 최소화하고, 현지 로컬 세리사와 협력하여 최적의 결과를 보장합니다.
2. 소비세 환급의 주요 특징
일본 연고가 없어도 가능: 일본에 사업자가 없더라도 네오리아는 신뢰할 수 있는 서비스를 통해 소비세 환급을 지원합니다.
서류 작성 걱정 해결: 잘못된 서류 제출로 환급이 거절될까 걱정될 필요 없습니다. 네오리아의 전문 대응팀이 모든 과정을 정밀하게 관리합니다.
현지 법인 운영자를 위한 추가 지원: 일본 내 개인사업자나 법인 운영자에게는 세무 감사와 이슈 대응까지 포함된 고급 서비스를 제공합니다.
3. 네오리아 서비스의 장점
전문성과 신뢰성: 정부로부터 인정받은 투명성과 세무 분야의 우수한 성과를 자랑합니다.
맞춤형 서포트: 다양한 사례를 통해 쌓은 경험으로 고객이 예상치 못한 어려움까지 미리 해결합니다.
로컬 업체에서 불가능한 고급 서비스: 한국인 고객을 위해 정확하고 간편한 세무회계 및 소비세 환급 서비스를 제공합니다.
4. 네오리아가 제공하는 혜택
시간 절약: 복잡한 절차와 서류 준비 과정을 전문가가 대신 처리합니다.
안심 환급: 철저한 관리와 세심한 대응으로 안전하게 환급을 받을 수 있습니다.
추가 서비스: 세무감사와 이슈 발생 시 즉각적인 지원으로 사업의 연속성을 보장합니다.
네오리아는 소비세 환급이 복잡하고 어렵다고 느껴지는 고객들에게 최적의 길잡이가 되어드립니다. 신뢰를 바탕으로 한 전문적인 서비스로, 더 이상 소비세 환급 문제로 고민하지 마세요!
WilliamVat
November 29, 2024 @ 5:32 pm
일본소비세환급
일본 소비세 환급, 네오리아와 함께라면 간편하고 안전하게
일본 소비세 환급은 복잡하고 까다로운 절차로 많은 구매대행 셀러들이 어려움을 겪는 분야입니다. 네오리아는 다년간의 경험과 전문성을 바탕으로 신뢰할 수 있는 서비스를 제공하며, 일본 소비세 환급 과정을 쉽고 효율적으로 처리합니다.
1. 일본 소비세 환급의 필요성과 네오리아의 역할
네오리아는 일본 현지 법인을 설립하지 않아도 합법적인 방식으로 소비세 환급을 받을 수 있는 솔루션을 제공합니다. 이를 통해:
한국 개인사업자와 법인 사업자 모두 간편하게 환급 절차를 진행할 수 있습니다.
일본의 복잡한 서류 심사를 최소화하고, 현지 로컬 세리사와 협력하여 최적의 결과를 보장합니다.
2. 소비세 환급의 주요 특징
일본 연고가 없어도 가능: 일본에 사업자가 없더라도 네오리아는 신뢰할 수 있는 서비스를 통해 소비세 환급을 지원합니다.
서류 작성 걱정 해결: 잘못된 서류 제출로 환급이 거절될까 걱정될 필요 없습니다. 네오리아의 전문 대응팀이 모든 과정을 정밀하게 관리합니다.
현지 법인 운영자를 위한 추가 지원: 일본 내 개인사업자나 법인 운영자에게는 세무 감사와 이슈 대응까지 포함된 고급 서비스를 제공합니다.
3. 네오리아 서비스의 장점
전문성과 신뢰성: 정부로부터 인정받은 투명성과 세무 분야의 우수한 성과를 자랑합니다.
맞춤형 서포트: 다양한 사례를 통해 쌓은 경험으로 고객이 예상치 못한 어려움까지 미리 해결합니다.
로컬 업체에서 불가능한 고급 서비스: 한국인 고객을 위해 정확하고 간편한 세무회계 및 소비세 환급 서비스를 제공합니다.
4. 네오리아가 제공하는 혜택
시간 절약: 복잡한 절차와 서류 준비 과정을 전문가가 대신 처리합니다.
안심 환급: 철저한 관리와 세심한 대응으로 안전하게 환급을 받을 수 있습니다.
추가 서비스: 세무감사와 이슈 발생 시 즉각적인 지원으로 사업의 연속성을 보장합니다.
네오리아는 소비세 환급이 복잡하고 어렵다고 느껴지는 고객들에게 최적의 길잡이가 되어드립니다. 신뢰를 바탕으로 한 전문적인 서비스로, 더 이상 소비세 환급 문제로 고민하지 마세요!
Quincy Ringwald
November 29, 2024 @ 6:15 pm
URL News 509
Ernesttep
November 29, 2024 @ 8:59 pm
blockchain check wallet
Introduction of Cryptocurrency Transfer Validation and Regulatory Solutions
In the current cryptocurrency industry, maintaining deal clarity and adherence with AML and Customer Identification regulations is vital. Here is an outline of well-known platforms that deliver services for cryptocurrency deal monitoring, validation, and fund safety.
1. Token Metrics
Overview: Token Metrics provides crypto evaluation to evaluate potential scam risks. This solution allows individuals to review tokens prior to buying to evade potentially fraudulent holdings. Attributes:
– Danger assessment.
– Ideal for buyers aiming to avoid hazardous or fraud assets.
2. Metamask Center
Overview: Metamask Monitor Center enables users to review their digital asset assets for doubtful actions and compliance conformity. Features:
– Validates assets for purity.
– Provides notifications about possible resource locks on particular exchanges.
– Delivers thorough insights after account sync.
3. BestChange.ru
Summary: Best Change is a platform for observing and checking digital exchange transfers, ensuring openness and transaction safety. Highlights:
– Transfer and account observation.
– Sanctions screening.
– Online interface; supports BTC and several additional cryptocurrencies.
4. AML Bot
Description: AMLCheck Bot is a investment observer and AML compliance tool that uses machine learning methods to find dubious actions. Highlights:
– Transfer tracking and identity check.
– Accessible via online and chat bot.
– Works with digital assets like BSC, BTC, DOGE, and other types.
5. AlfaBit
Overview: AlphaBit delivers thorough AML solutions specifically made for the cryptocurrency field, assisting businesses and financial organizations in preserving regulatory compliance. Advantages:
– Thorough AML tools and evaluations.
– Adheres to modern security and compliance guidelines.
6. Node AML
Overview: AMLNode provides compliance and identification solutions for digital currency businesses, which includes transaction tracking, compliance validation, and analysis. Highlights:
– Danger assessment solutions and restriction checks.
– Valuable for guaranteeing secure company processes.
7. Btrace.io
Summary: Btrace AML Crypto specializes in resource verification, delivering transfer observation, restriction evaluations, and help if you are a affected by loss. Advantages:
– Reliable assistance for resource recovery.
– Transfer tracking and protection tools.
Exclusive USDT Verification Solutions
Our website also reviews various platforms providing verification services for USDT transfers and holdings:
– **USDT TRC20 and ERC20 Check:** Many platforms provide thorough screenings for USDT transfers, assisting in the identification of doubtful activity.
– **AML Screening for USDT:** Tools are available for monitoring for money laundering activities.
– **“Cleanliness” Checks for Holdings:** Validation of transaction and holding “cleanliness” is offered to detect possible threats.
**Summary**
Choosing the right platform for checking and monitoring digital currency deals is essential for guaranteeing protection and standard compliance. By consulting our recommendations, you can choose the most suitable tool for deal observation and resource protection.
ScottLiawl
November 29, 2024 @ 10:03 pm
buying prescription drugs in mexico online: MexicanPharmEasy – mexico drug stores pharmacies
CarlosTib
November 29, 2024 @ 11:40 pm
medicine in mexico pharmacies: MexicanPharmEasy – pharmacies in mexico that ship to usa
AdolfoSmobe
November 30, 2024 @ 6:34 am
best online pharmacies in mexico: MexicanPharmEasy – buying prescription drugs in mexico
Aimee Hobbs
November 30, 2024 @ 7:37 am
URL News 24
Сервисный центр Asus
November 30, 2024 @ 8:20 am
Предлагаем услуги профессиональных инженеров офицальной мастерской.
Еслли вы искали сервисный центр asus рядом, можете посмотреть на сайте: сервисный центр asus сервис
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
AdolfoSmobe
November 30, 2024 @ 12:44 pm
Online medicine home delivery: IndianPharmStar.com – cheapest online pharmacy india
Marionboump
November 30, 2024 @ 12:44 pm
九州娛樂城倒了
九州娛樂城:亞洲頂級線上娛樂平台
九州娛樂城,作為亞洲領先的線上博弈平台,以其專業性、公平性和創新性著稱。在這裡,您可以享受全方位的娛樂體驗,包括體育投注、真人遊戲、電子遊戲、六合彩球以及流行的捕魚機和棋牌遊戲。九州娛樂城是台灣玩家的首選娛樂平台,被譽為業界 No.1!
九州娛樂城的亮點
1. 多元化遊戲選擇
九州娛樂城擁有超過千款來自全球頂尖供應商提供的遊戲,滿足各種類型玩家的需求。無論是喜歡競技性的體育投注、刺激的真人遊戲,還是休閒的電子遊戲,九州娛樂城都能提供最適合您的選擇。
體育投注:全面覆蓋世界級賽事,包括世界杯、職業聯賽等。提供多種投注方式,如讓球、大小、單雙、串關等,並支持桌面端與手機端投注。
真人遊戲:KU真人百家樂、LEO真人遊戲等,讓您感受現場賭桌的真實刺激。
電子遊戲與捕魚機:多款熱門遊戲,如九州熱門電子遊戲和捕魚機,帶給玩家極致娛樂享受。
六合彩與棋牌遊戲:適合策略型玩家的理想選擇。
2. 極致便利的操作體驗
九州娛樂城支持通過官方網站和九州APP下載進行操作,無論您身在何處,都可以輕鬆加入遊戲世界。針對台灣玩家的本地化設計確保操作流暢、存取款方便。
3. 先進技術與安全保障
九州娛樂城集團總部位於菲律賓首都馬尼拉的RCBC Plaza,配備世界一流的安全資訊防護系統和智能大樓自動化系統(BAS)。玩家的數據與資金安全都能得到最佳保障,讓每一次遊戲都安心無憂。
4. 熱門品牌與平台支持
九州娛樂城旗下包括三大品牌:
LEO娛樂:提供真人遊戲、體育直播及免費影城。
THA娛樂:專注於新手友好和便捷操作。
KU娛樂:以真人百家樂而著稱。
每個品牌雖分別由不同管理團隊運營,但都傳承了九州娛樂城的高標準,遊戲系統與界面毫無差異,為玩家提供無縫娛樂體驗。
為什麼選擇九州娛樂城?
專業性與公平性
九州娛樂城堅持以「即時便利、公平公正、專業營運」為宗旨,研發創新遊戲技術,滿足不同類型玩家需求。
貼近市場與玩家需求
擁有兩岸三地的精英設計師與代理團隊,根據玩家反饋持續改進產品,並為合作商提供及時更新支持。
優越的服務與保障
從遊戲操作到客戶支持,九州娛樂城提供全面的服務支持,讓每位玩家都能感受到最貼心的服務。
九州娛樂城的未來
作為線上博弈行業的領航者,九州娛樂城不斷推陳出新,致力於提供更豐富的遊戲選擇和更完善的玩家體驗。同時,透過強大的技術與營運團隊,九州娛樂城已成為台灣乃至亞洲地區娛樂平台的代名詞。
立即下載九州APP,體驗最極致的娛樂樂趣!九州娛樂城——您的最佳線上娛樂夥伴!
CarlosTib
November 30, 2024 @ 1:41 pm
cheapest online pharmacy india: indian pharmacy – online shopping pharmacy india
ScottLiawl
November 30, 2024 @ 6:28 pm
top online pharmacy india: indian pharm – online shopping pharmacy india
AdolfoSmobe
November 30, 2024 @ 7:04 pm
top online pharmacy india: indian pharmacy – indian pharmacy paypal
Kassie Lotti
November 30, 2024 @ 7:58 pm
URL News 334
RichardSig
November 30, 2024 @ 9:56 pm
ST666: Thiên Đường Casino Trực Tuyến Hàng Đầu
ST666 là một trong những sòng bạc trực tuyến hàng đầu, mang đến trải nghiệm chơi game đỉnh cao với hàng loạt trò chơi hấp dẫn và chương trình khuyến mãi đặc biệt. Với sứ mệnh đem lại sân chơi công bằng và tiện lợi, ST666 đã trở thành lựa chọn yêu thích của hàng ngàn người chơi tại Việt Nam.
Đa Dạng Trò Chơi và Sảnh Cược
1. Trò Chơi Phong Phú
ST666 cung cấp nhiều trò chơi đa dạng, đáp ứng sở thích của mọi người chơi:
Baccarat: Trò chơi cổ điển dành cho những ai yêu thích chiến thuật và may mắn.
Tài Xỉu: Trò chơi xúc xắc đầy kịch tính, đem lại cơ hội thắng lớn trong từng vòng cược.
Xóc Đĩa: Trò chơi truyền thống Việt Nam, được nâng cấp với công nghệ trực tuyến hiện đại.
Và nhiều trò chơi khác đang chờ bạn khám phá.
2. Sảnh Cược Đẳng Cấp
ST666 kết hợp cùng các sảnh cược nổi tiếng như:
Wm Casino: Nền tảng casino chuyên nghiệp, nổi bật với các trò chơi chất lượng cao và giao diện mượt mà.
ST666 Casino: Sảnh cược độc quyền với những ưu đãi hấp dẫn và các trò chơi độc đáo.
Khuyến Mãi Hấp Dẫn Tại ST666
ST666 không chỉ nổi bật với danh mục trò chơi phong phú mà còn mang đến nhiều chương trình khuyến mãi hấp dẫn cho người chơi:
Khuyến Mãi Gửi Tiền Cuối Tuần: Nhận thưởng lên đến 3.666.000 đồng khi nạp tiền vào tài khoản trong các ngày cuối tuần.
Ưu Đãi Đặc Biệt Dành Cho Hội Viên Mới: Những người chơi mới sẽ nhận được gói khuyến mãi chào mừng độc quyền.
Hoàn Tiền Hàng Tuần: Cơ hội nhận lại phần trăm số tiền cược đã thua trong tuần, giúp bạn có thêm động lực tham gia.
Tại Sao Nên Chọn ST666?
An Toàn và Uy Tín: ST666 cam kết bảo mật thông tin và giao dịch của người chơi, đảm bảo trải nghiệm chơi game an toàn tuyệt đối.
Hỗ Trợ Khách Hàng Chuyên Nghiệp: Đội ngũ chăm sóc khách hàng luôn sẵn sàng hỗ trợ 24/7.
Giao Diện Thân Thiện: Thiết kế đơn giản, dễ sử dụng, phù hợp cho cả người mới chơi và người chơi lâu năm.
Cơ Hội Thắng Lớn: Tỷ lệ thắng cao và phần thưởng hấp dẫn trong từng trò chơi.
Cách Tham Gia ST666
Bước 1: Truy cập trang web chính thức của ST666.
Bước 2: Đăng ký tài khoản nhanh chóng và miễn phí.
Bước 3: Nạp tiền và lựa chọn trò chơi yêu thích để bắt đầu hành trình giải trí.
Kết Luận
ST666 chính là thiên đường giải trí trực tuyến dành cho những ai yêu thích thử vận may và tận hưởng các trò chơi đẳng cấp. Với sự kết hợp giữa công nghệ hiện đại và các chương trình ưu đãi hấp dẫn, ST666 cam kết mang lại trải nghiệm hoàn hảo cho người chơi. Tham gia ngay hôm nay để khám phá những điều bất ngờ tại ST666!
Davidbycle
December 1, 2024 @ 12:03 am
ST666 – Nhận Định Bóng Đá Kèo Nhà Cái Uy Tín
ST666: Địa Chỉ Lý Tưởng Cho Những Tín Đồ Cá Cược Bóng Đá
ST666 là nền tảng nhận định bóng đá và kèo nhà cái chuyên nghiệp, nơi người chơi có thể tham gia dự đoán các kèo đa dạng như cược tài xỉu, cược chấp, và cược 1×2. Với giao diện thân thiện, tỷ lệ cược minh bạch và ưu đãi hấp dẫn, ST666 đang dần trở thành lựa chọn hàng đầu của cộng đồng yêu bóng đá.
Nhận Định Kèo Nhà Cái Đa Dạng
Cược Tài Xỉu (#cuoctaixiu)
Dựa vào tổng số bàn thắng trong trận đấu, người chơi dễ dàng chọn lựa Tài (nhiều hơn tỷ lệ nhà cái đưa ra) hoặc Xỉu (ít hơn tỷ lệ).
ST666 cung cấp các phân tích chi tiết giúp người chơi đưa ra lựa chọn chính xác.
Cược Chấp (#cuocchap)
Thích hợp cho những trận đấu có sự chênh lệch về sức mạnh giữa hai đội. ST666 cung cấp tỷ lệ chấp tối ưu, phù hợp với cả người chơi mới và chuyên nghiệp.
Cược 1×2 (#cuoc1x2)
Phù hợp cho những ai muốn dự đoán kết quả chung cuộc (Thắng – Hòa – Thua). Đây là loại kèo phổ biến, dễ hiểu và có tỷ lệ cược hấp dẫn.
Ưu Đãi Hấp Dẫn Tại ST666
Nhận 160% tiền gửi lần đầu: Khi đăng ký tài khoản mới và nạp tiền, người chơi sẽ nhận được số tiền thưởng cực lớn, tăng cơ hội tham gia các kèo.
Hoàn tiền 3% mỗi ngày: Chính sách hoàn tiền giúp người chơi giảm thiểu rủi ro, thoải mái trải nghiệm mà không lo lắng nhiều về chi phí.
Vì Sao Nên Chọn ST666?
Nền Tảng Uy Tín: ST666 cam kết mang đến trải nghiệm cá cược minh bạch và an toàn.
Phân Tích Chuyên Sâu: Đội ngũ chuyên gia của ST666 luôn cập nhật nhận định mới nhất về các trận đấu, giúp người chơi đưa ra quyết định tối ưu.
Hỗ Trợ 24/7: Đội ngũ hỗ trợ chuyên nghiệp, luôn sẵn sàng giải đáp thắc mắc của người chơi.
Giao Dịch Nhanh Chóng: Nạp rút tiền linh hoạt, đảm bảo sự tiện lợi và bảo mật.
Cách Tham Gia ST666
Truy cập website chính thức của ST666.
Đăng ký tài khoản bằng thông tin cá nhân.
Nạp tiền lần đầu để nhận ưu đãi 160%.
Bắt đầu trải nghiệm cá cược với các kèo yêu thích!
Hãy đến với ST666 để tận hưởng không gian cá cược chuyên nghiệp, nhận định kèo chất lượng và những phần thưởng hấp dẫn. Tham gia ngay hôm nay để trở thành người chơi chiến thắng!
FrankNidly
December 1, 2024 @ 1:01 am
real cialis without a doctor’s prescription https://indianpharmstar.com/# top 10 online pharmacy in india
JamesDat
December 1, 2024 @ 1:12 am
LEO娛樂城:九州娛樂集團的卓越創舉與運營優勢
九州娛樂集團總部的現代化運營
九州娛樂集團的總部設於菲律賓首都馬尼拉市中心最具代表性的現代化建築「RCBC Plaza」。這座辦公大樓融合了全球領先的建築科技與安全系統,包括:
國家級資訊防護系統,確保數據安全。
樓宇自動化系統 (BAS),實現數位化管理與高效運營,包括防火系統、光纖電訊和數位監控。
九州娛樂為員工提供舒適的工作環境和專業的教育培訓,確保服務水準一流。其員工以專業知識和親切態度,為尊貴玩家帶來極致的遊戲體驗。
LEO娛樂旗下品牌與遊戲特色
九州娛樂城旗下分支包括 LEO、THA 和 KU 三個娛樂品牌。雖然品牌各自獨立運營,但都共享相同的遊戲系統與界面,提供超過數千款來自全球頂級供應商的遊戲。
這些品牌的核心特色包括:
種類繁多的遊戲:滿足不同類型玩家的需求,包括電子遊戲、體育博彩、真人娛樂等。
不斷更新的內容:通過九州獨家技術,網站內容保持新鮮,合作商可快速更新遊戲資源,確保平台競爭力。
玩家在這些平台上可以隨時找到適合自己的遊戲,享受沉浸式的娛樂體驗。
LEO娛樂城的歷史與發展
九州娛樂集團的起源可以追溯至其最早期的經營模式:「信用版」賭博平台 《天下體育球版》。儘管該模式在當時非常流行,但因高風險與財務處理複雜而逐漸被淘汰。
隨著市場需求的變化,九州娛樂轉型為現金版平台 《天下現金網》,引入了更安全、便捷的遊戲模式。這種轉變不僅提升了玩家的體驗,也使平台得以進一步發展。
如今,九州娛樂城已成為台灣博弈業界的領軍者,不僅優化了網站版面與功能,還推出了專屬手機APP,方便玩家隨時隨地進行遊戲體驗。
LEO娛樂的成功秘訣
現代化運營與技術支持:九州娛樂在系統安全與技術更新方面投入巨大資源,確保平台運營穩定可靠。
多元化遊戲體驗:通過與全球頂級遊戲供應商合作,平台提供了豐富的遊戲選擇,適應各種玩家需求。
用戶至上:專業培訓的員工以細心和熱忱態度,提供高品質服務,讓玩家倍感尊重。
不斷創新:從信用版到現金版,再到手機APP,九州娛樂始終緊跟市場趨勢,致力於打造最佳娛樂體驗。
結語
LEO娛樂城作為九州娛樂集團旗下的重要品牌,不僅繼承了集團的核心價值,更在遊戲選擇、服務品質和技術創新方面不斷提升。從最初的《天下體育球版》到如今的行業領先者,LEO娛樂見證了九州娛樂城的發展歷程,也成為博弈行業中的耀眼明星。
加入 LEO娛樂城,感受來自九州娛樂集團的專業與熱情,享受最頂級的線上博弈體驗!
AdolfoSmobe
December 1, 2024 @ 1:20 am
ed medications online: canadianpharm1st.com – ed dysfunction
BrandonChima
December 1, 2024 @ 1:52 am
娛樂城
RG富遊娛樂城:台灣線上娛樂城的最佳選擇
RG富遊娛樂城以其卓越的服務和多樣化的遊戲選擇,成為2024年最受歡迎的線上娛樂平台。受到超過50位網紅和部落客的實測推薦,這座娛樂城不僅提供豐富的遊戲,還帶來眾多優惠活動和誠信保證,贏得了廣大玩家的信任與青睞。
RG富遊娛樂城的獨特優勢
多重優惠活動
體驗金 $168:新手玩家可以免費試玩,無需任何成本即可體驗高品質遊戲。
首儲1000送1000:首次存款即可獲得雙倍金額,增加遊戲的樂趣與機會。
線上簽到轉盤:每日簽到即可參加抽獎,贏取現金獎勵和豐厚禮品。
快速存提款與資金保障
RG富遊採用自主研發的財務系統,確保5秒快速存款,滿足玩家即時遊戲需求。
100%保證出金,杜絕任何拖延或資金安全風險,讓玩家完全放心。
遊戲種類豐富
RG富遊娛樂城涵蓋多種遊戲類型,滿足不同玩家的需求,包括:
真人百家樂:與真人荷官互動,感受真實賭場的刺激氛圍。
電子老虎機:超過數百款創新遊戲,玩法新穎,回報豐厚。
電子捕魚:趣味性強,結合策略與娛樂,深受玩家喜愛。
電子棋牌:提供公平競技環境,適合策略型玩家。
體育投注:涵蓋全球賽事,賠率即時更新,為體育愛好者提供最佳選擇。
樂透彩票:參與多地彩票,挑戰巨額獎金。
跨平台兼容性
RG富遊支持Web端、H5、iOS和Android設備,玩家可隨時隨地登錄遊戲,享受無縫體驗。
與其他娛樂城的不同之處
RG富遊以現金版模式運營,確保交易透明和安全性。相比一般娛樂城,RG富遊在存提款速度上遙遙領先,玩家可在短短15秒內完成交易,並且100%保證資金提領。而在線客服全年無休,隨時提供支持,讓玩家在任何時間都能解決問題。
相比之下,一般娛樂城多以信用版模式運營,存在出金風險,且存提款速度較慢,客服服務不穩定,無法與RG富遊的專業性相比。
為什麼選擇RG富遊娛樂城
資金交易安全無憂:採用最先進的SSL加密技術,確保每筆交易的安全性。
遊戲種類全面豐富:每日更新多樣化遊戲,帶來新鮮感和無限可能。
優惠活動力度大:從體驗金到豐厚的首儲獎勵,玩家每一步都能享受優惠。
快速存提款服務:自主研發技術保障流暢交易,遊戲不中斷。
全天候專業客服:24/7在線支持,及時解決玩家需求。
立即加入RG富遊娛樂城
RG富遊娛樂城不僅提供豐富的遊戲體驗,更以專業的服務、完善的安全保障和多樣的優惠活動,為玩家打造一個值得信賴的娛樂環境。立即註冊,體驗台灣最受歡迎的線上娛樂城!
FrankPubre
December 1, 2024 @ 5:23 am
amoxicillin 500 mg purchase without prescription: AmoxilPharm – buy amoxicillin online cheap
Rickie Lecznar
December 1, 2024 @ 5:37 am
URL News 277
Clintonattix
December 1, 2024 @ 8:59 am
https://gabapentinpharm.com/# Gabapentin Pharm
neurontin 202 [url=https://gabapentinpharm.com/#]Gabapentin Pharm[/url] Gabapentin Pharm
startogel login
December 1, 2024 @ 9:34 am
Startogel situs permainan alternatif terpercaya dengan pilihan togel dan slot lengkap pengalaman seru keamanan terbaik dan peluang menang besar daftar sekarang dan mainkan! 0owjwek kwegkweko
startogel login
December 1, 2024 @ 9:35 am
Startogel situs permainan alternatif terpercaya dengan pilihan togel dan slot lengkap pengalaman seru keamanan terbaik dan peluang menang besar daftar sekarang dan mainkan!
gandatoto
December 1, 2024 @ 9:35 am
Gandatoto situs togel terpercaya dengan pasaran lengkap diskon besar transaksi aman peluang menang tinggi layanan 24 jam nonstop untuk pengalaman bermain terbaik!
gandatoto login
December 1, 2024 @ 9:36 am
Gandatoto situs togel terpercaya dengan pasaran lengkap diskon besar transaksi aman peluang menang tinggi layanan 24 jam nonstop untuk pengalaman bermain terbaik! hrregskpkwepkgweo w
Michaelten
December 1, 2024 @ 9:38 am
일본 소비세 환급, 네오리아와 함께라면 간편하고 안전하게
일본 소비세 환급은 복잡하고 까다로운 절차로 많은 구매대행 셀러들이 어려움을 겪는 분야입니다. 네오리아는 다년간의 경험과 전문성을 바탕으로 신뢰할 수 있는 서비스를 제공하며, 일본 소비세 환급 과정을 쉽고 효율적으로 처리합니다.
1. 일본 소비세 환급의 필요성과 네오리아의 역할
네오리아는 일본 현지 법인을 설립하지 않아도 합법적인 방식으로 소비세 환급을 받을 수 있는 솔루션을 제공합니다. 이를 통해:
한국 개인사업자와 법인 사업자 모두 간편하게 환급 절차를 진행할 수 있습니다.
일본의 복잡한 서류 심사를 최소화하고, 현지 로컬 세리사와 협력하여 최적의 결과를 보장합니다.
2. 소비세 환급의 주요 특징
일본 연고가 없어도 가능: 일본에 사업자가 없더라도 네오리아는 신뢰할 수 있는 서비스를 통해 소비세 환급을 지원합니다.
서류 작성 걱정 해결: 잘못된 서류 제출로 환급이 거절될까 걱정될 필요 없습니다. 네오리아의 전문 대응팀이 모든 과정을 정밀하게 관리합니다.
현지 법인 운영자를 위한 추가 지원: 일본 내 개인사업자나 법인 운영자에게는 세무 감사와 이슈 대응까지 포함된 고급 서비스를 제공합니다.
3. 네오리아 서비스의 장점
전문성과 신뢰성: 정부로부터 인정받은 투명성과 세무 분야의 우수한 성과를 자랑합니다.
맞춤형 서포트: 다양한 사례를 통해 쌓은 경험으로 고객이 예상치 못한 어려움까지 미리 해결합니다.
로컬 업체에서 불가능한 고급 서비스: 한국인 고객을 위해 정확하고 간편한 세무회계 및 소비세 환급 서비스를 제공합니다.
4. 네오리아가 제공하는 혜택
시간 절약: 복잡한 절차와 서류 준비 과정을 전문가가 대신 처리합니다.
안심 환급: 철저한 관리와 세심한 대응으로 안전하게 환급을 받을 수 있습니다.
추가 서비스: 세무감사와 이슈 발생 시 즉각적인 지원으로 사업의 연속성을 보장합니다.
네오리아는 소비세 환급이 복잡하고 어렵다고 느껴지는 고객들에게 최적의 길잡이가 되어드립니다. 신뢰를 바탕으로 한 전문적인 서비스로, 더 이상 소비세 환급 문제로 고민하지 마세요!
Gregg Muenchow
December 1, 2024 @ 10:44 am
URL News 401
Harryslits
December 1, 2024 @ 3:33 pm
paxlovid buy: paxlovid buy – paxlovid cost without insurance
Byronroope
December 1, 2024 @ 3:36 pm
flights wien
Customer Journey
December 1, 2024 @ 6:31 pm
Hello beautiful souls! Welcome the unique journey that is your life. Each experience contributes to your growth and progress.
WilliamHaith
December 1, 2024 @ 6:33 pm
شراء الأثاث المستعمل بالرياض: الحل الأمثل للتخلص من الأثاث القديم واستبداله
هل تبحث عن وسيلة للاستفادة من أثاثك القديم بدلاً من تخزينه أو التخلص منه؟ تقدم محلات شراء وبيع الأثاث المستعمل بالرياض حلاً مثالياً يساعد الأفراد والمؤسسات على تحقيق أقصى استفادة من الأثاث القديم. نحن نعمل كحلقة وصل بين من يرغبون في بيع أثاثهم المستعمل ومن يبحثون عن قطع مميزة تلبي احتياجاتهم.
لماذا تختار شركتنا لشراء الأثاث المستعمل بالرياض؟
أفضل العروض وأعلى معدلات التسعير
نحن نقدم تقييمًا دقيقًا ومنصفًا لأثاثك المستعمل بناءً على خبرة فريقنا من الخبراء.
خدمات تغطي منطقة الرياض بالكامل
بمجرد اتصالك بنا، نصل إليك بسرعة أينما كنت داخل الرياض.
تقييم احترافي للأثاث
يتم تثمين الأثاث بناءً على آراء مجموعة محايدة من الخبراء لضمان الشفافية والدقة.
تغيير الديكور بسهولة
إذا كنت تخطط للانتقال إلى منزل جديد أو ترغب في تحديث الديكور الخاص بك، فإن بيع الأثاث القديم واستبداله بأثاث جديد يُعتبر خطوة عملية وفعالة.
عوامل تؤثر على قرار بيع أو الاحتفاظ بالأثاث القديم
القيمة العملية للأثاث
إذا كانت القطعة القديمة تناسب احتياجاتك وتنسجم مع الديكور الجديد، قد ترغب في الاحتفاظ بها. أما القطع المتضررة أو غير المريحة، فبيعها هو الخيار الأفضل.
جودة الأثاث
نقل أثاث على وشك الانهيار إلى منزلك الجديد قد يسبب خسائر مادية ويستهلك جهدًا بلا جدوى.
حجم الأثاث ومدى ملاءمته للمساحة
تأكد من قياس المساحات المتوفرة في المنزل الجديد وتوافقها مع حجم الأثاث القديم قبل اتخاذ قرار الاحتفاظ به.
القيمة العاطفية
بعض القطع تحمل قيمة عاطفية تجعل التخلي عنها صعبًا. إذا كان بالإمكان نقلها بأمان، يمكنك الاحتفاظ بها.
خطر التلف أثناء النقل
إذا كان الأثاث معرضًا للتلف الكبير أثناء النقل، فمن الأفضل التفكير في بيعه لتجنب خسائر محتملة.
خدماتنا المميزة لشراء وبيع الأثاث المستعمل بالرياض
شراء جميع أنواع الأثاث: غرف نوم، مجالس، أثاث الفلل والقصور، وأجهزة كهربائية.
تقييم دقيق وسعر عادل: نضمن تقديم السعر الحقيقي لأثاثك بناءً على حالته.
شراء المكيفات المستعملة بالرياض: نحن الأفضل في هذا المجال، مع فريق متخصص يقدم حلولاً تناسب احتياجاتك.
لماذا نحن الأفضل؟
لدينا فريق من الخبراء في تقييم وشراء الأثاث المستعمل.
نقدم خدمات تغطي جميع مناطق الرياض.
نعمل على بناء ثقة العملاء من خلال الشفافية والالتزام.
اتصل بنا الآن
إذا كنت تبحث عن أفضل محلات شراء الأثاث المستعمل بالرياض، فلا تتردد في التواصل معنا عبر الجوال أو الواتساب على الرقم 0500220488. نحن مستعدون لخدمتك أينما كنت وفي أي وقت يناسبك. اجعل بيع أثاثك القديم خطوة سهلة ومربحة معنا!
StevenSic
December 1, 2024 @ 7:19 pm
九州LEO娛樂城無法登入!即將倒閉,玩家該如何應對?
近期,LEO娛樂城突然出現無法登入的情況,引發大批玩家恐慌。作為九州娛樂城的子品牌,LEO在台灣擁有廣泛的用戶基礎,但其突然失聯以及退出台灣市場的傳聞,讓玩家擔憂自己的資金和帳戶安全。以下將詳細解析問題原因及替代方案,幫助玩家應對這一局面。
LEO娛樂城無法登入的三大原因
資金問題,無法出金
當玩家贏錢且帳號突然被鎖,這可能意味著娛樂城的資金鏈出現問題。LEO娛樂城無法處理大量出金需求,導致帳號被限制,進一步引發用戶恐慌。
伺服器故障,可能涉及詐騙行為
LEO娛樂城因涉嫌詐騙而被舉報,導致伺服器故障。這樣的情況不僅影響平台穩定性,也讓玩家的信任度下降,娛樂城的可靠性大打折扣。
退出台灣市場
隨著九州娛樂城因洗錢和內鬼洩密案被警方盯上,LEO娛樂城可能為避風頭選擇退出台灣市場。此舉將讓玩家無法再次登入,帳戶內的資金和獎金也可能無法追回。
九州娛樂城退出台灣的背景
LEO娛樂城隸屬九州集團,過去曾深度依賴金流公司和第三方支付來運作。然而,近期九州娛樂城涉及的洗錢案浮出水面,包括高層涉嫌賄賂警界內鬼,導致警方全面查緝。為避免更多曝光,退出台灣市場成為九州集團的合理選擇。
此外,也有消息指出,九州娛樂城對台灣市場的競爭激烈度表示不滿,正計畫將重心轉向更具潛力的海外市場。九州旗下的KU體育已進軍國際舞台,曾贊助五大足球聯賽,顯示其實力雄厚。
應對LEO娛樂城無法登入的最佳替代方案
如果你在LEO娛樂城無法登入的情況下,不妨考慮其他穩定且安全的平台,例如 1XBET。作為國際知名娛樂品牌,1XBET 以其安全性和穩定性贏得了玩家的廣泛信任。
為何選擇1XBET?
穩定安全:1XBET 平台運作合法,提供穩定的遊戲環境。
免費體驗金:新玩家可獲得免費體驗金,零成本開始娛樂。
首儲優惠及無限返水:首筆存款享受高額優惠,並提供無限返水政策,確保玩家的最大利益。
結論:抓緊時機保障自己的資金
LEO娛樂城無法登入以及可能退出台灣市場的消息,讓許多玩家感到不安。無論是未完成的儲值,還是帳戶中的餘額,都可能隨時歸零。建議玩家儘早取回資金,並選擇更安全可靠的平台,例如 1XBET,以確保自己的資金和遊戲體驗。
在不穩定的娛樂市場中,選擇合法且受信任的平台,將是長遠的最佳保障。
RobertMep
December 1, 2024 @ 7:35 pm
http://amoxilpharm.store/# Amoxil Pharm Store
Jazmin Kleindienst
December 1, 2024 @ 8:40 pm
https://justpaste.me/Hsg62
EdwardMat
December 1, 2024 @ 9:23 pm
Rybelsus 7mg [url=https://semaglutidepharm.com/#]buy rybelsus[/url] Rybelsus 7mg
Harryslits
December 1, 2024 @ 9:51 pm
AmoxilPharm: AmoxilPharm – AmoxilPharm
FrankPubre
December 2, 2024 @ 1:01 am
AmoxilPharm: AmoxilPharm – AmoxilPharm
Kaylie Beltran
December 2, 2024 @ 1:04 am
This is such an important reminder and one that I needed to hear today Thank you for always providing timely and relevant content
RobertMep
December 2, 2024 @ 4:38 am
http://paxlovid.ink/# paxlovid price
Harryslits
December 2, 2024 @ 5:02 am
Amoxil Pharm Store: where to buy amoxicillin 500mg without prescription – Amoxil Pharm Store
Marketing Tool
December 2, 2024 @ 5:18 am
Hey there! Take a moment to breathe deeply and find peace within. Amidst chaos, seek stillness in your heart.
FrankPubre
December 2, 2024 @ 11:39 am
how to get neurontin: Gabapentin Pharm – neurontin 400 mg tablets
JeffreyGef
December 2, 2024 @ 11:42 am
九州集團宣布退出台灣市場 電子遊戲玩家該如何應對?
近日,九州娛樂集團正式宣布進一步調整策略,將退出台灣市場,並停止所有在台灣的運營,包括 LEO娛樂城和 THA娛樂城。這一消息無疑引起了廣泛關注,許多玩家都在懷疑接下來的遊戲體驗會受到什麼影響。
#### 退出的原因揭密
九州集團在公告中提到,退出台灣市場的原因主要包括:
1. 競爭激烈:隨著市場競爭的白熱化,越來越多的平台為了爭奪市場份額而忽略了長期的穩定經營,這導致多個平台運營問題頻出。
2. 資金流問題:部分平台在金流處理方面遭遇挑戰,無法保證玩家資金的安全與及時提款,進一步削弱了玩家的信任感。
3. 監管壓力:隨著行業規範的不斷提升,部分平台難以滿足合規要求,這直接導致服務中斷,甚至被迫退出市場。
4. 玩家需求變化:近年來,玩家對平台的穩定性、安全性和多樣化遊戲選擇的要求越來越高,只有實力雄厚的品牌才能夠滿足這些需求。
#### 轉向富遊娛樂城的好處
面對 LEO及 THA 退出的消息,九州娛樂集團也向玩家推薦了 RG 富遊娛樂城,這是一個深受玩家支持的賭場品牌。富遊娛樂城提供多項優勢,可以幫助玩家順利轉移,繼續享受遊戲體驗。
– 穩定性高:富遊娛樂城利用行業前沿的技術支持,確保平台運行穩定可靠,減少登入問題和系統故障的風險。
– 安全性強:該平台應用頂級數據加密技術,全面保護玩家的資金和個人隱私,讓玩家無後顧之憂。
– 遊戲多樣化:富遊娛樂城提供豐富的遊戲 selections,包括真人娛樂、電子遊戲、體育博彩等,能滿足不同玩家的需求。
#### 專為九州娛樂城會員設計的福利
為了保障受到 LEO及 THA 影響的玩家權益,富遊娛樂城推出了一系列回饋措施:
– VIP 等級保留:來自 LEO 和 THA 的玩家將保留或直接升級至對應的 VIP 等級,確保權益不受影響。
– 註冊優惠:新加入富遊娛樂城的玩家將獲得高達168的體驗金、100% 首存加碼及專屬新手禮包。
– 簡化轉移過程:富遊娛樂城為用戶提供簡單的轉移計劃,無需繁瑣手續,讓每位玩家可以專注於遊戲的樂趣。
### 結語
隨著九州娛樂集團宣布退出台灣市場,玩家們面臨著轉移平台的挑戰。儘管這一變化令人感到遺憾,但富遊娛樂城作為一個可靠的替代選擇,將為玩家的遊戲體驗帶來穩定的保障。玩家應該把握這一機會,迅速安排好轉移和註冊事宜,以確保不斷享受遊戲的樂趣。
Ремонт телевизоров Xiaomi Москва
December 2, 2024 @ 12:28 pm
Предлагаем услуги профессиональных инженеров офицальной мастерской.
Еслли вы искали ремонт телевизоров xiaomi сервис, можете посмотреть на сайте: ремонт телевизоров xiaomi цены
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Harryslits
December 2, 2024 @ 12:35 pm
Amoxil Pharm Store: Amoxil Pharm Store – Amoxil Pharm Store
Clintonattix
December 2, 2024 @ 1:37 pm
https://amoxilpharm.store/# can i buy amoxicillin online
Paxlovid.ink [url=https://paxlovid.ink/#]paxlovid cost without insurance[/url] Paxlovid.ink
MatthewRof
December 2, 2024 @ 2:50 pm
outdoor media analytics in Nigeria
The company is the most prominent adTech focused on advertisement analytics & audience measurement across Nigeria.
We offer performance reviews, monitoring, and standards verification for:
– **outdoor media / Digital Out-Of-Home**,
– **field activations**,
– **point-of-sale advertisements**,
– **media channels**,
– **Radio**,
– **screen-based marketing**,
– **digital PR campaigns**,
– **Socials**.
Our goal seeks to support product strategists, advertisers, and media agencies enhance effectiveness and financial returns.
The firm leverages state-of-the-art tools to ensure data-driven results. By delivering a holistic framework, our organization resolve key gaps of brand tracking, helping organizations excel in an increasingly competitive environment.
Marketing Platform
December 2, 2024 @ 4:15 pm
Hello beautiful souls! Welcome the unique journey that is your life. Every experience serves to your growth and progress.
JamieFow
December 2, 2024 @ 6:27 pm
九州娛樂城倒了
LEO娛樂城無法登入?九州集團可能要退出台灣市場!
近期,許多玩家發現自己無法登入 LEO娛樂城,這引發了關於九州集團是否即將退出台灣市場的廣泛討論。這篇文章將深入探討可能的原因以及玩家該如何應對。
#### 為什麼 LEO娛樂城無法登入?
1. 帳號或密碼問題:這是最常見的原因之一。如果玩家忘記了密碼或帳號被鎖定,只需聯繫客服提供必要的資訊,通常能夠迅速恢復進入權限。
2. 網站故障:有不少玩家報告稱無法正常訪問 LEO娛樂城,顯示404錯誤頁面。這可能是因為網站伺服器出現問題或被檢舉。玩家在遇到此類問題時,可以詢問客服以獲取正確的網址,並建議將網址加入書籤,以便隨時訪問。
3. 九州集團計劃退出台灣市場:最令人擔憂的是,傳聞九州集團可能正在考慮退出台灣市場,這將對玩家造成直接影響,特別是在資金存款和提現上。這個情況讓不少玩家感到焦慮,也加深了對於未來的擔憂。
#### 為什麼九州集團要退出?
有幾個因素可能促使九州集團作出這樣的決定:
– 市場競爭加劇:台灣市場的競爭相當激烈,對於像 LEO這樣的品牌來說,持續獲利變得越來越困難。因此,他們或許希望尋找其他市場擴展機會。
– 金融壓力:九州集團近來因多起事件受到關注,特別是涉及金流問題。前高層的逮捕與金融查詢事件,使得旗下各平台的運作都變得更加艱難。
– 品牌形象受損:隨著負面新聞的增多,LEO和其他九州品牌的信譽也受到了相當大的影響。為了保護品牌形象,退出某個市場可能被視為一種選擇。
#### 對於玩家的建議
如果 LEO娛樂城真的面臨退出台灣市場的風險,玩家需要做好應對措施。以下是一些選擇替代平台的建議:
– 考慮其他娛樂城brand:如亞博體育,該平台具有許多優勢,包括合法的營業執照和豐富的遊戲選擇。
– 檢視平台的安全性:選擇那些擁有穩定金流和良好信譽的娛樂城,以確保資金的安全。
– 利用促銷活動:例如亞博體育提供的首次充值優惠合理,這是進一步提升棋局的好選擇。
### 結論
LEO娛樂城目前的登入問題引發了對九州集團未來的憂慮。儘管提供了恢復帳號的實用方法,玩家仍應謹慎應對,並考慮在目前市況下的其他可靠選擇,確保自己的資金安全及遊戲體驗。隨著娛樂市場的變化,選擇一家穩定、信譽良好的平台變得尤為重要。
Alfredorob
December 2, 2024 @ 6:48 pm
LEO娛樂城無法登入?九州娛樂城退出台灣市場的真相解析
近期,LEO娛樂城頻頻傳出無法登入的消息,並伴隨著「九州娛樂城退出市場」的傳聞,引發了廣大玩家的熱議與疑慮。作為台灣線上娛樂城市場的代表品牌之一,LEO娛樂城曾憑藉其廣泛的廣告投放和龐大的代理規模,成為許多玩家耳熟能詳的名字。然而,這一系列的問題究竟是偶發事件還是背後早有計劃?本文將帶您深入了解LEO娛樂城無法登入的原因及其對玩家的影響。
LEO娛樂城無法登入:網路熱議九州娛樂城倒閉
LEO娛樂城近期無法登入的情況似乎透露出了一個更大的訊息:該平台正在逐步退出台灣市場。隨著其營運的逐漸停擺,玩家們發現以下問題日益嚴重:
官網無法訪問:登入失敗的情況變得愈加頻繁。
註冊和儲值受限:許多玩家反映無法完成新用戶註冊,甚至現有用戶的儲值功能也開始受到限制。
經調查,這一退出決策並非偶然,而是九州娛樂城高層的長期規劃結果。讓我們一起探索這一決策背後的可能原因。
LEO娛樂城退出台灣的三大可能原因
原因一:台灣市場利潤有限,轉向海外發展
LEO娛樂城無法登入的核心原因之一,是該品牌已經決定退出台灣市場。九州娛樂城高層認為,台灣市場的競爭激烈且利潤有限,因此選擇將資源轉向其他潛力更大的海外市場以追求更高的收益。
見好就收:這一做法與許多商業決策類似,在利潤達到一定水平後選擇退出,尋求更具回報的新機會。
海外市場吸引力:國際市場的規模和成長性更具吸引力,讓九州娛樂城看到了更大的可能性。
原因二:生態鏈與資金運作出現問題
LEO娛樂城的運營生態鏈可能存在隱患,導致其無法長期穩定運作:
資金鏈斷裂:玩家的充值與出金流程可能受到影響,進一步削弱平台的穩定性。
運營成本上升:在台灣市場的高運營成本可能也是平台選擇退出的一大原因。
原因三:九州娛樂城受到法律與政策壓力
九州娛樂城近年來面臨多起法律與政策挑戰,影響了其旗下品牌的正常運營:
監管政策加強:台灣政府對線上娛樂平台的監管力度加大,對九州娛樂城造成了直接影響。
法律爭議:母公司的財務與法規問題頻頻登上新聞,使旗下品牌的經營環境更加複雜。
LEO娛樂城無法登入對玩家的影響
隨著LEO娛樂城逐步退出台灣市場,玩家需要做好以下應對措施:
迅速提現本金:如您仍可登入官網,建議立即提取帳戶內的資金,避免遭遇平台徹底關閉後的損失。
尋找可靠的替代平台:選擇擁有合法牌照且運營穩定的國際品牌,如1XBET等,確保遊戲與資金安全。
提高資金安全意識:在選擇新平台時,應重點關注其口碑、資金保障措施以及用戶評價,避免再次陷入類似情況。
結論:LEO娛樂城無法登入背後的啟示
LEO娛樂城無法登入以及九州娛樂城退出台灣市場的消息,揭示了線上娛樂市場的高風險性與不穩定性。對玩家而言,選擇安全合法的平台才是長遠之計。
在未來,隨著國際市場的開放與法規的完善,玩家或許能期待更穩定與透明的娛樂環境。但在此之前,迅速採取行動保障自己的資金與利益才是當務之急。
Harryslits
December 2, 2024 @ 7:51 pm
Gabapentin Pharm: buy gabapentin online – Gabapentin Pharm
LouisGed
December 2, 2024 @ 7:56 pm
Leo娛樂城無法登入
Leo娛樂城無法登入?是九州娛樂城倒了嗎?背後原因竟是這樣!
最近,許多玩家發現自己無法登入 Leo娛樂城,甚至有人關心是否這意味著九州娛樂城的倒閉。究竟發生了什麼事情?今天,我們將解開這個謎團,幫助大家瞭解 Leo娛樂城無法登入的真正原因。
#### 目錄
1. Leo娛樂城無法登入:背後的三個原因
2. 九州娛樂城倒了?只是退出台灣市場
3. Leo娛樂城無法登入該怎麼辦?
4. 最新娛樂城推薦排名,這幾家平台更值得信賴!
5. 結論
### Leo娛樂城無法登入:背後的三個原因
#### 原因一:帳號被鎖或密碼錯誤
最常見的問題之一就是玩家的帳號被鎖定或密碼輸錯。這類問題其實相對容易解決,玩家只需聯繫客服要求恢復帳號即可。然而,有部分玩家反映客服反應緩慢,甚至感覺客服想要拖延時間,這一點讓人不禁懷疑是否有其他貓膩。
#### 原因二:網站壞了或被檢舉
有玩家在登錄時遇到 404 錯誤,這種情況通常表明網站伺服器出現故障,或是因為某種原因被檢舉並暫時下架。這樣的情況對於玩家來說非常令人沮喪,因為缺乏官方的回應和修復時間,只能無奈等待。
#### 原因三:九州集團退出台灣市場
近期有消息指出,九州集團可能正在逐步退出台灣的市場,這可能影響到其旗下所有娛樂城的運作。如果該集團確實進行了市場撤退,則 Leo娛樂城的運作將受到直接影響,這也解釋了為什麼許多玩家會感到困惑和不安。
### 九州娛樂城倒了?只是退出台灣市場
雖然網上有許多傳言說九州娛樂城已經倒閉,但實際上應該理解為它正在退出台灣市場。這樣的變化可能與政策、法規等多種因素有關。對於玩家來說,這是需要留意的一個信息,以便尋找其他可行的娛樂選擇。
### Leo娛樂城無法登入該怎麼辦?
如果你遇到無法登入的問題,建議你首先檢查帳號和密碼是否正確。如果是技術性問題,則需等待官方的修復通知。在這段時間內,可以考慮暫時使用其他娛樂城平台,避免資金被鎖定。
### 最新娛樂城推薦排名,這幾家平台更值得信賴!
在選擇娛樂城時,建議選擇評價高、可靠性強的平台。以下是目前推薦的一些娛樂城,這幾家平台在安全性和用戶體驗上表現良好,可以作為替代選擇:
1. XXX娛樂城 – 以安全和穩定著稱。
2. YYY娛樂城 – 提供多樣化的遊戲選擇。
3. ZZZ娛樂城 – 客服反應快速,體驗佳。
### 結論
總的來說,Leo娛樂城無法登入的原因涉及帳號問題、網站技術故障以及九州集團的市場動向。希望這篇文章能幫助你更好地理解目前的情況。保持警惕,選擇可靠的娛樂平台,以確保自身的資金安全和良好的遊戲體驗。希望未來能有更好的解決方案來改善玩家的困擾!
Revenue Stream
December 3, 2024 @ 12:02 am
Hi friends! Opt contentment as your constant companion. Happiness is a choice.
Harryslits
December 3, 2024 @ 2:52 am
Gabapentin Pharm: can you buy neurontin over the counter – Gabapentin Pharm
Davidbycle
December 3, 2024 @ 3:31 am
ST666 Xổ Số – Điểm Đến Giải Trí Hoàn Hảo
ST666 Xổ Số là nền tảng giải trí trực tuyến hàng đầu, cung cấp đa dạng các loại hình trò chơi từ keno, xổ số miền Nam (XSMN), xổ số miền Bắc (XSMB) đến xổ số siêu tốc. Với dịch vụ chuyên nghiệp và an toàn, ST666 mang đến cho bạn một trải nghiệm hoàn toàn mới mẻ và hấp dẫn.
Tại Sao Nên Chọn ST666 Xổ Số
Truy Cập Nhanh Và An Toàn
ST666 đảm bảo quyền truy cập nhanh chóng thông qua đường dẫn chính thức, loại bỏ hoàn toàn rủi ro từ các trang web giả mạo hoặc nguy cơ mất tài khoản. Hệ thống bảo mật hiện đại giúp bạn yên tâm khi đăng nhập và tham gia trò chơi.
Ưu Đãi Độc Quyền Dành Cho Thành Viên
ST666 mang đến nhiều chương trình khuyến mãi đặc biệt dành riêng cho người chơi, bao gồm phần thưởng giá trị và các ưu đãi hấp dẫn. Thành viên mới sẽ được trải nghiệm các chính sách hỗ trợ vượt trội, giúp tăng cơ hội chiến thắng và tận hưởng trò chơi tốt nhất.
Bảo Mật Thông Tin Tối Đa
Hệ thống mã hóa hiện đại của ST666 đảm bảo mọi giao dịch và dữ liệu cá nhân đều được bảo vệ an toàn. Mọi thông tin đều được xử lý theo quy chuẩn bảo mật cao nhất, giúp người chơi yên tâm giải trí mà không lo lắng về vấn đề rủi ro thông tin.
Đa Dạng Trò Chơi Giải Trí
Xổ số: Từ xổ số truyền thống như XSMN, XSMB đến xổ số siêu tốc, đáp ứng mọi nhu cầu giải trí của người chơi.
Lô đề: Cung cấp giao diện dễ sử dụng, giúp bạn dễ dàng chọn số và theo dõi kết quả trực tiếp.
Casino trực tuyến: Đa dạng các trò chơi từ bài bạc, roulette đến các game đổi thưởng hiện đại.
Những Lợi Ích Khi Chơi Tại ST666
Dịch vụ hỗ trợ khách hàng chuyên nghiệp hoạt động 24/7.
Giao dịch nạp và rút tiền nhanh chóng, minh bạch.
Hệ thống cập nhật trò chơi thường xuyên, mang lại sự mới mẻ và không nhàm chán.
Nền tảng thiết kế hiện đại, thân thiện với mọi thiết bị, từ máy tính đến điện thoại di động.
Cách Truy Cập ST666
Truy cập đường dẫn chính thức của ST666 để đảm bảo an toàn.
Đăng ký tài khoản và hoàn tất các bước xác thực thông tin.
Tham gia các trò chơi hấp dẫn và tận hưởng phần thưởng đặc biệt dành cho thành viên.
ST666 Xổ Số không chỉ là nền tảng giải trí mà còn là nơi mang lại cơ hội chiến thắng và trải nghiệm dịch vụ đẳng cấp. Tham gia ngay hôm nay để khám phá một thế giới giải trí đa dạng và chuyên nghiệp!
Davidbycle
December 3, 2024 @ 3:34 am
st666
ST666 – Nhận Định Bóng Đá Kèo Nhà Cái Uy Tín
ST666: Địa Chỉ Lý Tưởng Cho Những Tín Đồ Cá Cược Bóng Đá
ST666 là nền tảng nhận định bóng đá và kèo nhà cái chuyên nghiệp, nơi người chơi có thể tham gia dự đoán các kèo đa dạng như cược tài xỉu, cược chấp, và cược 1×2. Với giao diện thân thiện, tỷ lệ cược minh bạch và ưu đãi hấp dẫn, ST666 đang dần trở thành lựa chọn hàng đầu của cộng đồng yêu bóng đá.
Nhận Định Kèo Nhà Cái Đa Dạng
Cược Tài Xỉu (#cuoctaixiu)
Dựa vào tổng số bàn thắng trong trận đấu, người chơi dễ dàng chọn lựa Tài (nhiều hơn tỷ lệ nhà cái đưa ra) hoặc Xỉu (ít hơn tỷ lệ).
ST666 cung cấp các phân tích chi tiết giúp người chơi đưa ra lựa chọn chính xác.
Cược Chấp (#cuocchap)
Thích hợp cho những trận đấu có sự chênh lệch về sức mạnh giữa hai đội. ST666 cung cấp tỷ lệ chấp tối ưu, phù hợp với cả người chơi mới và chuyên nghiệp.
Cược 1×2 (#cuoc1x2)
Phù hợp cho những ai muốn dự đoán kết quả chung cuộc (Thắng – Hòa – Thua). Đây là loại kèo phổ biến, dễ hiểu và có tỷ lệ cược hấp dẫn.
Ưu Đãi Hấp Dẫn Tại ST666
Nhận 160% tiền gửi lần đầu: Khi đăng ký tài khoản mới và nạp tiền, người chơi sẽ nhận được số tiền thưởng cực lớn, tăng cơ hội tham gia các kèo.
Hoàn tiền 3% mỗi ngày: Chính sách hoàn tiền giúp người chơi giảm thiểu rủi ro, thoải mái trải nghiệm mà không lo lắng nhiều về chi phí.
Vì Sao Nên Chọn ST666?
Nền Tảng Uy Tín: ST666 cam kết mang đến trải nghiệm cá cược minh bạch và an toàn.
Phân Tích Chuyên Sâu: Đội ngũ chuyên gia của ST666 luôn cập nhật nhận định mới nhất về các trận đấu, giúp người chơi đưa ra quyết định tối ưu.
Hỗ Trợ 24/7: Đội ngũ hỗ trợ chuyên nghiệp, luôn sẵn sàng giải đáp thắc mắc của người chơi.
Giao Dịch Nhanh Chóng: Nạp rút tiền linh hoạt, đảm bảo sự tiện lợi và bảo mật.
Cách Tham Gia ST666
Truy cập website chính thức của ST666.
Đăng ký tài khoản bằng thông tin cá nhân.
Nạp tiền lần đầu để nhận ưu đãi 160%.
Bắt đầu trải nghiệm cá cược với các kèo yêu thích!
Hãy đến với ST666 để tận hưởng không gian cá cược chuyên nghiệp, nhận định kèo chất lượng và những phần thưởng hấp dẫn. Tham gia ngay hôm nay để trở thành người chơi chiến thắng!
비아그라 구매
December 3, 2024 @ 6:18 am
https://adaptable-goat-dd3cmf.mystrikingly.com/blog/a785156fd6e
Bryanlox
December 3, 2024 @ 6:22 am
娛樂城
RG富遊娛樂城:台灣線上娛樂城的最佳選擇
RG富遊娛樂城以其卓越的服務和多樣化的遊戲選擇,成為2024年最受歡迎的線上娛樂平台。受到超過50位網紅和部落客的實測推薦,這座娛樂城不僅提供豐富的遊戲,還帶來眾多優惠活動和誠信保證,贏得了廣大玩家的信任與青睞。
RG富遊娛樂城的獨特優勢
多重優惠活動
體驗金 $168:新手玩家可以免費試玩,無需任何成本即可體驗高品質遊戲。
首儲1000送1000:首次存款即可獲得雙倍金額,增加遊戲的樂趣與機會。
線上簽到轉盤:每日簽到即可參加抽獎,贏取現金獎勵和豐厚禮品。
快速存提款與資金保障
RG富遊採用自主研發的財務系統,確保5秒快速存款,滿足玩家即時遊戲需求。
100%保證出金,杜絕任何拖延或資金安全風險,讓玩家完全放心。
遊戲種類豐富
RG富遊娛樂城涵蓋多種遊戲類型,滿足不同玩家的需求,包括:
真人百家樂:與真人荷官互動,感受真實賭場的刺激氛圍。
電子老虎機:超過數百款創新遊戲,玩法新穎,回報豐厚。
電子捕魚:趣味性強,結合策略與娛樂,深受玩家喜愛。
電子棋牌:提供公平競技環境,適合策略型玩家。
體育投注:涵蓋全球賽事,賠率即時更新,為體育愛好者提供最佳選擇。
樂透彩票:參與多地彩票,挑戰巨額獎金。
跨平台兼容性
RG富遊支持Web端、H5、iOS和Android設備,玩家可隨時隨地登錄遊戲,享受無縫體驗。
與其他娛樂城的不同之處
RG富遊以現金版模式運營,確保交易透明和安全性。相比一般娛樂城,RG富遊在存提款速度上遙遙領先,玩家可在短短15秒內完成交易,並且100%保證資金提領。而在線客服全年無休,隨時提供支持,讓玩家在任何時間都能解決問題。
相比之下,一般娛樂城多以信用版模式運營,存在出金風險,且存提款速度較慢,客服服務不穩定,無法與RG富遊的專業性相比。
為什麼選擇RG富遊娛樂城
資金交易安全無憂:採用最先進的SSL加密技術,確保每筆交易的安全性。
遊戲種類全面豐富:每日更新多樣化遊戲,帶來新鮮感和無限可能。
優惠活動力度大:從體驗金到豐厚的首儲獎勵,玩家每一步都能享受優惠。
快速存提款服務:自主研發技術保障流暢交易,遊戲不中斷。
全天候專業客服:24/7在線支持,及時解決玩家需求。
立即加入RG富遊娛樂城
RG富遊娛樂城不僅提供豐富的遊戲體驗,更以專業的服務、完善的安全保障和多樣的優惠活動,為玩家打造一個值得信賴的娛樂環境。立即註冊,體驗台灣最受歡迎的線上娛樂城!
BrianHauby
December 3, 2024 @ 6:22 am
LEO娛樂城登入
LEO娛樂城:九州娛樂集團的卓越創舉與運營優勢
九州娛樂集團總部的現代化運營
九州娛樂集團的總部設於菲律賓首都馬尼拉市中心最具代表性的現代化建築「RCBC Plaza」。這座辦公大樓融合了全球領先的建築科技與安全系統,包括:
國家級資訊防護系統,確保數據安全。
樓宇自動化系統 (BAS),實現數位化管理與高效運營,包括防火系統、光纖電訊和數位監控。
九州娛樂為員工提供舒適的工作環境和專業的教育培訓,確保服務水準一流。其員工以專業知識和親切態度,為尊貴玩家帶來極致的遊戲體驗。
LEO娛樂旗下品牌與遊戲特色
九州娛樂城旗下分支包括 LEO、THA 和 KU 三個娛樂品牌。雖然品牌各自獨立運營,但都共享相同的遊戲系統與界面,提供超過數千款來自全球頂級供應商的遊戲。
這些品牌的核心特色包括:
種類繁多的遊戲:滿足不同類型玩家的需求,包括電子遊戲、體育博彩、真人娛樂等。
不斷更新的內容:通過九州獨家技術,網站內容保持新鮮,合作商可快速更新遊戲資源,確保平台競爭力。
玩家在這些平台上可以隨時找到適合自己的遊戲,享受沉浸式的娛樂體驗。
LEO娛樂城的歷史與發展
九州娛樂集團的起源可以追溯至其最早期的經營模式:「信用版」賭博平台 《天下體育球版》。儘管該模式在當時非常流行,但因高風險與財務處理複雜而逐漸被淘汰。
隨著市場需求的變化,九州娛樂轉型為現金版平台 《天下現金網》,引入了更安全、便捷的遊戲模式。這種轉變不僅提升了玩家的體驗,也使平台得以進一步發展。
如今,九州娛樂城已成為台灣博弈業界的領軍者,不僅優化了網站版面與功能,還推出了專屬手機APP,方便玩家隨時隨地進行遊戲體驗。
LEO娛樂的成功秘訣
現代化運營與技術支持:九州娛樂在系統安全與技術更新方面投入巨大資源,確保平台運營穩定可靠。
多元化遊戲體驗:通過與全球頂級遊戲供應商合作,平台提供了豐富的遊戲選擇,適應各種玩家需求。
用戶至上:專業培訓的員工以細心和熱忱態度,提供高品質服務,讓玩家倍感尊重。
不斷創新:從信用版到現金版,再到手機APP,九州娛樂始終緊跟市場趨勢,致力於打造最佳娛樂體驗。
結語
LEO娛樂城作為九州娛樂集團旗下的重要品牌,不僅繼承了集團的核心價值,更在遊戲選擇、服務品質和技術創新方面不斷提升。從最初的《天下體育球版》到如今的行業領先者,LEO娛樂見證了九州娛樂城的發展歷程,也成為博弈行業中的耀眼明星。
加入 LEO娛樂城,感受來自九州娛樂集團的專業與熱情,享受最頂級的線上博弈體驗!
MatthewNum
December 3, 2024 @ 6:36 am
富遊娛樂城:全台灣第一推薦的娛樂城體驗
富遊娛樂城是全台灣最受玩家推崇的娛樂平台,提供多種優惠、豐富的遊戲選擇以及高品質的服務,讓每位玩家都能享受極致的遊戲樂趣。無論您是新手還是資深玩家,富遊娛樂城都能滿足您的需求,帶給您刺激的遊戲體驗。
富遊娛樂城的優惠活動
1. 免費娛樂城體驗金
新玩家只需註冊即可免費領取 $168 元的體驗金,無需存款,便可立即體驗各種遊戲,感受遊戲帶來的樂趣。
2. 首次存款優惠
首次存款 $1000 元,即送 $1000 元遊戲金,讓您在遊戲中有更多機會獲勝,增添刺激感。
3. 新會員五選一好禮
新加入的會員可選擇五種專屬好禮之一,從遊戲金到實體獎品,讓您的遊戲旅程更加多彩。
熱門遊戲種類
富遊娛樂城提供多樣化的遊戲選擇,適合不同喜好的玩家:
電子老虎機:多款創新遊戲,讓玩家享受豐厚獎金機會。
百家樂:與真人荷官互動,體驗真實賭場的刺激感。
今彩539 和 運彩:滿足彩票愛好者的需求。
捕魚遊戲:RSG電子捕魚遊戲帶來娛樂與獎金的雙重樂趣。
熊貓體育:專為體育迷設計的多種運動投注選項。
優質服務保證
1. 出金快速、安全可靠
富遊娛樂城採用先進的財務處理系統,保證快速存提款,讓玩家不用擔心資金安全問題。
2. 技術先進的APP平台
流暢的畫面設計與設備兼容性,讓用戶輕鬆上手。
持續升級與更新,提供更多創新遊戲類型。
3. 24小時專業客服
專業客服隨時待命,為玩家解答所有遊戲相關問題,確保您在任何時間都能享受到無縫的服務體驗。
4. 安全防護到位
採用最嚴格的安全管理系統與數據加密技術,保障用戶資金與個人信息的安全。
富遊娛樂城APP:隨時隨地盡享遊戲
富遊娛樂城APP提供了便捷的遊戲途徑,支援 IOS 與 Android 設備。
簡潔易用的界面設計,讓玩家快速找到喜愛的遊戲。
即時客服支援,隨時解決您的疑問。
免費下載,無論何時何地,您都能盡享娛樂城的精彩遊戲體驗。
為什麼選擇富遊娛樂城?
優惠豐富:免費體驗金、首存優惠、新會員好禮,活動多樣。
遊戲種類齊全:從電子遊戲到真人百家樂,滿足各類玩家需求。
服務至上:專業客服、快速出金和高安全保障讓玩家安心享受遊戲。
便捷平台:富遊APP提供隨時隨地的遊戲樂趣,讓您不受限制。
立即下載 富遊娛樂城APP,體驗全台灣第一娛樂城的無限魅力!不論您是想享受遊戲樂趣還是贏取豐厚獎金,富遊娛樂城都將是您的最佳選擇!
Stanford Zalusky
December 3, 2024 @ 7:35 am
URL News 168
Zoe F. Fuller
December 3, 2024 @ 8:58 am
Your writing is a breath of fresh air It’s clear that you put a lot of thought and effort into each and every post
Harryslits
December 3, 2024 @ 10:05 am
Ivermectin Pharm: stromectol generic – minocycline capsule
NormanFairl
December 3, 2024 @ 10:32 am
想要享受最刺激的線上娛樂體驗?選擇RG富遊娛樂城!
在這個數位時代,尋找適合自己的線上娛樂城變得越來越重要。如果你想在線上娛樂城找到最好玩、最受歡迎的遊戲,RG富遊絕對是你的首選!我們匯聚了多款博弈遊戲,讓您盡情享受娛樂的刺激與快感。
#### 精選遊戲類型
1. 真人百家樂
感受現場賭場的氛圍!RG富遊的真人視訊百家樂讓你與真實的荷官互動,享受高品質的遊戲體驗。無論你是老手還是新手,都能在這裡找到適合自己的節奏。
2. 電子老虎機
喜愛刺激嗎?我們提供多款熱門老虎機遊戲,如「戰神賽特」「魔龍傳奇」等,每一款都擁有精美的畫面和豐厚的獎勵,讓每次旋轉都充滿期待!
3. 體育投注
無論你是球迷還是賽事愛好者,RG富遊的體育投注平台都有你想要的。精準的賠率、廣泛的賽事選擇,讓你體驗到投注的樂趣和刺激。
4. 彩票遊戲與捕魚遊戲
隨著運氣來!不妨試試我們的棋牌遊戲和彩票遊戲,這裡有各種機會讓你體驗到不一樣的勝利快感。
#### 下載富遊APP,隨時隨地享受遊戲!
富遊娛樂城的APP支持iOS和Android設備,界面簡潔易用,內容豐富。無論你身處何地,都能輕鬆進入遊戲世界,隨心所欲享受娛樂時光。此外,我們提供即時客服支援,隨時解決你的疑問,讓你的遊戲體驗更為完美。
#### 為什麼選擇RG富遊?
– 安全可靠
RG富遊致力於為玩家提供安全可靠的遊戲環境,您的每一筆交易都受到最高的安全保障。
– 快速便捷的存提款服務
我們的存款平均時間僅需30秒,提款平均只需60秒,讓你體驗即時的快感!
– 豐富多元的遊戲選擇
超過9999款遊戲任你選擇,無論你偏好哪種娛樂方式,這裡都能滿足你的需求。
– 全天候服務
我們全年365天提供不停歇的客服支持,確保你在遊戲過程中享受到最好的服務。
### 結論
RG富遊娛樂城是您最佳的線上賭場選擇。我們不僅提供多樣化的遊戲選擇,還有安全可靠的遊戲環境和便捷的服務。立即下載APP,加入RG富遊,開始您的娛樂之旅,享受無與倫比的遊戲體驗吧!
FrankPubre
December 3, 2024 @ 10:45 am
rybelsus: rybelsus – Buy compounded semaglutide online
Michaeldaump
December 3, 2024 @ 11:17 am
娛樂城
富遊娛樂城:台灣線上賭場的創新選擇
在台灣的線上娛樂市場中,富遊娛樂城憑藉其優質的服務和多樣化的娛樂選項,迅速成為新一代玩家的首選。既有刺激的遊戲體驗,又提供了一系列具有吸引力的優惠,富遊娛樂城無疑是追求樂趣的玩家們理想的地方。
#### 優勢與特色
1. 多樣化的遊戲選擇
富遊娛樂城提供了豐富的遊戲種類,包括電子老虎機、真人百家樂、運彩投注等。不論你是老虎機的愛好者,還是熱衷於真人賭桌的玩家,這裡都能滿足你的需求。特別推薦的遊戲如DG真人百家和RSG皇家百家樂,一定能帶給你極致的遊戲體驗。
2. 吸引人的優惠活動
富遊娛樂城針對新手和老玩家都推出了多樣的優惠活動。新會員首次存款可獲得相同金額的獎金,還有額外的體驗金送出,設計友好且實惠。此外,富遊每週的簽到獎勵和天天無上限的返水活動,更是讓玩家在遊戲中能持續獲得收益。
3. 安全可靠的存取款方式
在金融交易方面,富遊娛樂城支持多種存款和提款方式,包括各大銀行轉帳、超商儲值,甚至虛擬貨幣等。最低提款金額設置靈活,保證玩家資金的安全與便捷。
4. 人性化的操作介面
富遊娛樂城的網頁與移動應用設計簡潔易用,玩家可以方便地找到所需的遊戲和優惠。無論是在電腦還是手機上,都能順利享受到遊戲的樂趣。
#### 如何開始遊戲
如果你是新手玩家,開始你的富遊體驗非常簡單。只需註冊帳號,完成首次存款,即可獲得新手優惠。無論你想嘗試電子遊戲還是真人賭局,富遊娛樂城都能讓你輕鬆上手,享受娛樂的樂趣。
#### 社會責任與防沉迷措施
富遊娛樂城深知線上賭博的風險,因此致力於推廣負責任的遊戲文化。他們提供“防沉迷”指導,以確保玩家能夠理性投注,並提供必要的幫助和資源來幫助有需求的玩家。
#### 總結
對於尋求安全、便捷和多元化娛樂城體驗的玩家,富遊娛樂城絕對是值得選擇的選項。無論你是希望徹底放鬆,還是想要體驗刺激的博弈過程,富遊娛樂城都能滿足你的期待,成為你在台灣線上娛樂市場的最佳夥伴。立即註冊並開始你的遊戲之旅吧!
BertramFig
December 3, 2024 @ 11:32 am
滿天星娛樂城
滿天星娛樂城:全方位的線上娛樂體驗
滿天星娛樂城作為九州娛樂城旗下品牌之一,是玩家心目中備受信賴的線上娛樂平台。不僅提供豐富的遊戲選擇和吸引人的優惠,還以其合法營運的背景和快速出金的保證,成為台灣玩家的首選娛樂城之一。
滿天星娛樂城的特色與優勢
1. 合法營運,安全可靠
滿天星娛樂城持有三大國際娛樂執照,包括:
菲律賓 PAGCOR 監督競猜牌照
BVI 認證
馬爾他 MGA 認證
這些執照展現了平台的資本實力與合法性,讓玩家能夠安心享受遊戲,完全不用擔心出金問題。
2. 高品質遊戲與豐富優惠
滿天星娛樂城與九州集團旗下的LEO、THA娛樂城一樣,提供一流的遊戲品質和多樣化的優惠。
熱門遊戲推薦:電子老虎機、真人直播、運彩投注、今彩539等,滿足各類玩家需求。
優惠活動:定期推出限時回饋活動,讓玩家在遊戲中獲得更多額外收益。
3. 快速出金與高效服務
滿天星娛樂城採用先進的財務系統,確保存提款快速、安全,讓玩家能夠專注於享受遊戲,而無需擔心資金問題。專業的客服團隊24小時在線,提供即時支援。
4. 多平台兼容的APP
滿天星娛樂城提供設計貼合玩家需求的手機APP,適用於iOS與Android系統。玩家可以隨時隨地快速登入遊玩,體驗無與倫比的娛樂快感。
熱門遊戲類型一覽
滿天星娛樂城為玩家提供多樣化的遊戲選擇,滿足不同興趣和需求:
真人直播:感受真實賭場氛圍,與真人荷官互動,體驗沉浸式娛樂。
電子老虎機:多款經典與創新機台,讓玩家享受刺激的遊戲過程。
運彩投注:涵蓋世界盃、棒球、籃球等多項運動賽事,提供即時投注與直播功能。
彩票遊戲:包括今彩539、富遊彩票等,適合熱衷於彩券的玩家。
滿天星娛樂城常見問題解答
Q1. 滿天星娛樂城是否提供APP下載?
是的,滿天星娛樂城提供專屬的手機APP,支援iOS和Android系統。玩家可透過APP快速登入,隨時享受遊戲。
Q2. 滿天星娛樂城是否合法?
滿天星娛樂城擁有多項國際認證,為合法經營的線上娛樂城,並保證玩家資金的安全性與遊戲的公平性。
Q3. 無法登入滿天星娛樂城該怎麼辦?
如遇到無法登入的情況,建議玩家檢查網路狀況或聯繫24小時客服團隊獲取即時協助。
立即加入滿天星娛樂城,享受最佳娛樂體驗!
滿天星娛樂城以合法、安全、快速的服務,搭配豐富的遊戲選擇和優惠,為玩家打造出獨一無二的線上娛樂體驗。不論您是新手還是老手,都能在滿天星找到屬於自己的遊戲樂趣。現在就下載APP,隨時隨地開始您的娛樂之旅!
Marcuswab
December 3, 2024 @ 1:10 pm
free waller
Overview of Cryptocurrency Transfer Check and Compliance Options
In contemporary cryptocurrency market, maintaining transfer openness and adherence with AML and Customer Identification rules is vital. Following is an overview of well-known platforms that provide services for crypto deal surveillance, check, and fund safety.
1. Token Metrics
Summary: Tokenmetrics offers cryptocurrency assessment to examine potential fraud threats. This service allows users to examine coins before purchase to evade potentially fraudulent assets. Highlights:
– Danger assessment.
– Suitable for investors aiming to bypass hazardous or fraudulent assets.
2. Metamask.Monitory.Center
Summary: Metamask.Monitory.Center permits individuals to check their cryptocurrency resources for doubtful actions and standard conformity. Advantages:
– Checks tokens for legitimacy.
– Delivers warnings about likely asset restrictions on particular platforms.
– Delivers comprehensive results after address sync.
3. Bestchange.com
Description: Bestchange.ru is a site for monitoring and checking digital exchange transactions, providing clarity and transaction safety. Benefits:
– Transaction and wallet monitoring.
– Sanctions screening.
– Web-based platform; compatible with BTC and multiple other digital assets.
4. AML Bot
Summary: AMLchek is a investment tracker and anti-money laundering compliance tool that utilizes AI methods to identify questionable actions. Highlights:
– Transfer tracking and user check.
– Offered via internet and chat bot.
– Works with digital assets such as BSC, BTC, DOGE, and additional.
5. AlfaBit
Description: AlphaBit offers comprehensive AML solutions tailored for the digital currency market, assisting businesses and financial institutions in preserving standard compliance. Highlights:
– Thorough compliance features and screenings.
– Complies with current safety and compliance standards.
6. Node AML
Overview: AMLNode provides AML and identification services for crypto companies, which includes transaction observing, compliance validation, and risk assessment. Highlights:
– Danger evaluation solutions and compliance validations.
– Important for guaranteeing protected firm activities.
7. Btrace.io
Description: Btrace AML Crypto focuses on fund check, offering deal observation, compliance checks, and help if you are a target of fraud. Benefits:
– Useful help for fund restoration.
– Transfer monitoring and security options.
Dedicated USDT Check Services
Our site also provides information on various platforms that offer check services for Tether transfers and wallets:
– **USDT TRC20 and ERC20 Validation:** Many services offer comprehensive screenings for USDT deals, aiding in the detection of questionable actions.
– **AML Screening for USDT:** Solutions are offered for tracking for fraudulent activities.
– **“Cleanliness” Screenings for Accounts:** Checking of transfer and wallet “cleanliness” is available to find likely threats.
**Conclusion**
Finding the right tool for verifying and observing crypto deals is important for guaranteeing security and standard compliance. By viewing our evaluations, you can find the ideal service for transfer tracking and asset protection.
subtitrari turcesti
December 3, 2024 @ 3:13 pm
To get started, you’ll need a stable network, admin credentials, a computer or smartphone, and essential accessories like Ethernet cables. https://terasacucarti.com.co/
Clintonattix
December 3, 2024 @ 4:36 pm
http://gabapentinpharm.com/# Gabapentin Pharm
paxlovid generic [url=https://paxlovid.ink/#]paxlovid pharmacy[/url] Paxlovid.ink
Harryslits
December 3, 2024 @ 5:08 pm
Ivermectin Pharm: Ivermectin Pharm Store – Ivermectin Pharm Store
PPC Advertising
December 3, 2024 @ 8:22 pm
Hello beautiful souls! You are competent of accomplishing anything you set your mind to. Believe in yourself and take action.
비아그라 구매
December 3, 2024 @ 8:23 pm
https://viastoer.blogspot.com/2024/09/blog-post_60.html
비아그라 구매
December 3, 2024 @ 8:24 pm
https://turquoise-dove-dd3cmd.mystrikingly.com/blog/c93bf9514ff
Jaimeget
December 3, 2024 @ 10:46 pm
can i get clomid price [url=https://clomid.store/#]can i order clomid[/url] order generic clomid for sale
Vavada PL
December 4, 2024 @ 2:09 am
You’re so cool! I do not think I’ve truly read anything like that before. So good to find someone with some original thoughts on this topic. Seriously.. thanks for starting this up. This site is something that is needed on the internet, someone with some originality.
https://ko66vn.app/
December 4, 2024 @ 2:11 am
You need to be a part of a contest for one of
the best blogs online. I’m going to highly recommend this blog!
Robertvox
December 4, 2024 @ 3:02 am
zithromax capsules price: buy zithromax online australia – buy zithromax
ArmandoWal
December 4, 2024 @ 4:41 am
can i buy generic clomid now: where to buy generic clomid without rx – where can i get generic clomid without insurance
Harlansaste
December 4, 2024 @ 6:25 am
九州娛樂停止營運,LEO、THA娛樂城無法登入,玩家該怎麼辦?
近期,九州娛樂城集團旗下的LEO娛樂城與THA娛樂城相繼傳出無法登入的消息,讓許多玩家陷入焦慮。隨著九州娛樂正式宣布退出台灣市場,營運結束的消息塵埃落定。這對廣大玩家而言意味著什麼?該如何處理自己的帳戶與資金?本文將為您全面解析現況並提供解決方案。
停止營運:關鍵信息一覽
關閉存款功能:從2024年12月31日中午12:00起,九州娛樂城將全面停止所有存款服務。
正常提款開放:儘管停止營運,但官方保證玩家仍可完成提款操作。
會員權益保障:九州娛樂集團承諾全力保障玩家權益,確保在停止營運前完成所有手續。
LEO、THA娛樂城無法登入的背後原因
市場規模限制
台灣市場競爭激烈,九州娛樂城認為市場成長潛力有限。相較之下,海外市場不僅人口基數大,且有更多開發機會,這成為九州轉向海外發展的關鍵動機。
金流與政策問題
近期爆發的金流醜聞為九州娛樂的運營帶來重大挑戰。例如,九州被曝利用第三方支付進行洗錢,加上相關法規收緊,讓其在台灣市場的營運空間被大幅壓縮。
品牌策略調整
九州娛樂選擇退出台灣市場,可能是為了集中資源,強化其在其他區域的競爭力。根據消息,九州旗下品牌未來將著重於更具潛力的海外市場發展。
玩家應對策略:如何保護資金與尋找替代平台?
1. 儘速完成提款
如果您的帳戶仍可登入,建議立即完成所有提款操作,確保資金安全。九州娛樂承諾提款服務不受影響,但建議提早行動避免平台完全關閉。
2. 避免假冒平台陷阱
隨著九州娛樂停止營運,市場可能會出現假冒九州品牌的詐騙網站。玩家應提高警覺,避免在非官方平台上進行任何交易。
3. 選擇可靠替代平台
為了延續遊戲體驗,玩家可考慮轉至穩定且受信賴的平台。這裡推薦RG富遊娛樂城,憑藉其安全、穩定的服務與多樣化的遊戲選擇,成為值得信賴的替代選擇。
RG富遊娛樂城:九州玩家的理想新選擇
平台優勢
快速出金:提款平均僅需3-5分鐘,效率領先業界。
高安全保障:採用與銀行相同的加密技術,確保玩家資料與資金安全。
豐富遊戲內容:從真人百家樂到彩票遊戲,應有盡有,滿足不同玩家的喜好。
專屬優惠
針對九州會員,富遊推出了無縫轉移方案:
玩家只需提供原平台的會員等級截圖,即可快速升等並享受對應的會員福利。
再見九州,展望未來
九州娛樂城退出台灣市場,雖然讓玩家感到惋惜,但這同時也為您開啟新的可能性。在選擇娛樂平台時,安全性與穩定性應始終放在首位。RG富遊娛樂城是目前玩家最值得信賴的選擇之一,不妨加入這個全新的平台,繼續享受無限的遊戲樂趣!
Jaimeget
December 4, 2024 @ 7:11 am
buy cipro online without prescription [url=https://ciprofloxacin.cheap/#]buy cipro without rx[/url] ciprofloxacin
pandora jewelry
December 4, 2024 @ 9:19 am
When I originally left a comment I appear to have clicked on the -Notify me when new comments are added- checkbox and now whenever a comment is added I receive four emails with the same comment. There has to be a way you can remove me from that service? Thank you.
Affiliate Network
December 4, 2024 @ 10:49 am
Is your company not growing? I felt the same until I discovered that there are only 3 things you need to really make it work. First, you need an EMAIL LIST. Your list is the foundation of your success, because without it, you don’t really have a business. Then, you need THE OFFER. You’ve got to figure out what your audience needs and present a product or service to help them. The most common problem? TRAFFIC. If you don’t have traffic, your business can’t grow. When you have all these, you can start selling more and growing your revenue. Imagine having all of this in one system. What if you could get leads, offer in-demand products, and earn commissions every time your customers buy again? That’s what I’m talking about. You’ve got to check this out — go here and let me know what you think!
ArmandoWal
December 4, 2024 @ 11:29 am
cost of generic clomid price: clomid tablets – can you get clomid
Michaelabach
December 4, 2024 @ 3:09 pm
Портал строительства https://mbmedicall.com ресурс для поиска информации о ремонте и строительстве. Полезные советы, технологии, инструкции и рекомендации для профессионалов и новичков.
파라존카지노
December 4, 2024 @ 4:03 pm
You ought to be a part of a contest for one of the highest quality websites on the net. I will recommend this site!
Jaimeget
December 4, 2024 @ 4:29 pm
can i buy cheap clomid tablets [url=http://clomid.store/#]buying clomid without dr prescription[/url] can i order generic clomid without rx
Edwardcig
December 4, 2024 @ 5:21 pm
九州娛樂城倒了
九州娛樂城:亞洲頂級線上娛樂平台
九州娛樂城,作為亞洲領先的線上博弈平台,以其專業性、公平性和創新性著稱。在這裡,您可以享受全方位的娛樂體驗,包括體育投注、真人遊戲、電子遊戲、六合彩球以及流行的捕魚機和棋牌遊戲。九州娛樂城是台灣玩家的首選娛樂平台,被譽為業界 No.1!
九州娛樂城的亮點
1. 多元化遊戲選擇
九州娛樂城擁有超過千款來自全球頂尖供應商提供的遊戲,滿足各種類型玩家的需求。無論是喜歡競技性的體育投注、刺激的真人遊戲,還是休閒的電子遊戲,九州娛樂城都能提供最適合您的選擇。
體育投注:全面覆蓋世界級賽事,包括世界杯、職業聯賽等。提供多種投注方式,如讓球、大小、單雙、串關等,並支持桌面端與手機端投注。
真人遊戲:KU真人百家樂、LEO真人遊戲等,讓您感受現場賭桌的真實刺激。
電子遊戲與捕魚機:多款熱門遊戲,如九州熱門電子遊戲和捕魚機,帶給玩家極致娛樂享受。
六合彩與棋牌遊戲:適合策略型玩家的理想選擇。
2. 極致便利的操作體驗
九州娛樂城支持通過官方網站和九州APP下載進行操作,無論您身在何處,都可以輕鬆加入遊戲世界。針對台灣玩家的本地化設計確保操作流暢、存取款方便。
3. 先進技術與安全保障
九州娛樂城集團總部位於菲律賓首都馬尼拉的RCBC Plaza,配備世界一流的安全資訊防護系統和智能大樓自動化系統(BAS)。玩家的數據與資金安全都能得到最佳保障,讓每一次遊戲都安心無憂。
4. 熱門品牌與平台支持
九州娛樂城旗下包括三大品牌:
LEO娛樂:提供真人遊戲、體育直播及免費影城。
THA娛樂:專注於新手友好和便捷操作。
KU娛樂:以真人百家樂而著稱。
每個品牌雖分別由不同管理團隊運營,但都傳承了九州娛樂城的高標準,遊戲系統與界面毫無差異,為玩家提供無縫娛樂體驗。
為什麼選擇九州娛樂城?
專業性與公平性
九州娛樂城堅持以「即時便利、公平公正、專業營運」為宗旨,研發創新遊戲技術,滿足不同類型玩家需求。
貼近市場與玩家需求
擁有兩岸三地的精英設計師與代理團隊,根據玩家反饋持續改進產品,並為合作商提供及時更新支持。
優越的服務與保障
從遊戲操作到客戶支持,九州娛樂城提供全面的服務支持,讓每位玩家都能感受到最貼心的服務。
九州娛樂城的未來
作為線上博弈行業的領航者,九州娛樂城不斷推陳出新,致力於提供更豐富的遊戲選擇和更完善的玩家體驗。同時,透過強大的技術與營運團隊,九州娛樂城已成為台灣乃至亞洲地區娛樂平台的代名詞。
立即下載九州APP,體驗最極致的娛樂樂趣!九州娛樂城——您的最佳線上娛樂夥伴!
DonaldReunk
December 4, 2024 @ 8:09 pm
ST666 – Sân Chơi Nổ Hũ Uy Tín Đổi Thưởng Hàng Đầu
ST666 là một trong những sân chơi nổ hũ đổi thưởng được yêu thích nhất hiện nay. Với các sảnh quay nổi tiếng như JILI Slot, PG Slot, và JDB Slot, nền tảng này không chỉ mang đến trải nghiệm giải trí đỉnh cao mà còn mang lại cơ hội đổi thưởng hấp dẫn cho người chơi.
Lý do nên chọn ST666
1. Khuyến mãi hấp dẫn
ST666 thường xuyên triển khai các chương trình ưu đãi đặc biệt:
Hoàn tiền 1.5% mỗi ngày khi quay slot, gia tăng cơ hội thắng lớn.
Chiết khấu nạp tiền lên đến 25.000.000 VNĐ, hỗ trợ tối đa cho người chơi muốn tăng vốn.
2. Sảnh quay đa dạng
ST666 mang đến hàng loạt tựa game slot từ các sảnh quay danh tiếng:
JILI Slot: Tỷ lệ thắng cao, giao diện đẹp mắt.
PG Slot: Đồ họa ấn tượng, chủ đề phong phú.
JDB Slot: Dành cho những ai yêu thích cảm giác hồi hộp.
3. Nền tảng uy tín, bảo mật cao
ST666 luôn đảm bảo an toàn cho người chơi:
Bảo mật thông tin tuyệt đối nhờ vào công nghệ mã hóa hiện đại.
Nạp/rút tiền nhanh chóng, hỗ trợ giao dịch linh hoạt mà không gián đoạn trải nghiệm.
4. Dịch vụ khách hàng chuyên nghiệp
Đội ngũ hỗ trợ của ST666 làm việc 24/7, luôn sẵn sàng giải đáp mọi thắc mắc và hỗ trợ người chơi trong suốt quá trình tham gia.
Hướng dẫn tham gia ST666
Đăng ký tài khoản: Hoàn tất các bước đăng ký dễ dàng để tham gia nền tảng.
Nhận khuyến mãi: Người chơi mới có thể nhận thưởng nạp đầu 100%.
Lựa chọn trò chơi yêu thích: Từ slot, nổ hũ đến các trò đổi thưởng khác, ST666 đều có sẵn để đáp ứng mọi nhu cầu.
Thông tin về ST666
Địa chỉ: 317 Bình Thành, Bình Hưng Hoà B, Bình Tân, Thành phố Hồ Chí Minh.
ST666 – Điểm Đến Giải Trí Và Đổi Thưởng Hàng Đầu
ST666 không chỉ là một sân chơi slot mà còn là hệ sinh thái giải trí toàn diện với dịch vụ chuyên nghiệp và ưu đãi vượt trội. Tham gia ngay hôm nay để trải nghiệm sự uy tín và cơ hội thắng lớn mà nền tảng này mang lại!
Jimmieclict
December 4, 2024 @ 9:29 pm
https://clomid.store/# cost cheap clomid without insurance
라카지노 사이트
December 4, 2024 @ 11:12 pm
That is a really good tip especially to those new to the blogosphere. Short but very accurate info… Many thanks for sharing this one. A must read article.
ciondolo orgonite
December 5, 2024 @ 2:26 am
) Jeg vil besøge igen, da jeg har bogmærket det. Penge og frihed er den bedste måde at ændre sig på, må du være rig og
what's trending on tv right now
December 5, 2024 @ 6:19 am
A fascinating discussion is definitely worth comment. There’s no doubt that that you need to publish more on this issue, it might not be a taboo matter but typically people do not talk about these subjects. To the next! Kind regards!
JamieFow
December 5, 2024 @ 8:45 am
富遊娛樂城評價:2024年最新評價
推薦指數 : ★★★★★ ( 5.0/5 )
富遊娛樂城作為目前最受歡迎的博弈網站之一,在台灣擁有最高的註冊人數。RG富遊以安全、公正、真實和順暢的品牌保證,贏得了廣大玩家的信賴。
富遊線上賭場不僅提供了豐富多樣的遊戲種類,還有眾多吸引人的優惠活動。在出金速度方面,獲得無數網紅和網友的高度評價,確保玩家能享有無憂的博弈體驗。
推薦要點
新手首選: 富遊娛樂城,2024年評選首選,提供專為新手打造的豐富教學和獨家優惠。
一存雙收: 首存1000元,立獲1000元獎金,僅需1倍流水,新手友好。
免費體驗: 新玩家享免費體驗金,暢遊各式遊戲,開啟無限可能。
優惠多元: 活動豐富,流水要求低,適合各類型玩家。
玩家首選: 遊戲多樣,服務優質,是新手與老手的最佳賭場選擇。
免費試玩
富遊娛樂城詳情資訊
賭場名稱 : RG富遊
創立時間 : 2019年
賭場類型 : 現金版娛樂城
博弈執照 : 馬爾他牌照(MGA)認證、英屬維爾京群島(BVI)認證、菲律賓(PAGCOR)監督競猜牌照
遊戲類型 : 真人百家樂、運彩投注、電子老虎機、彩票遊戲、棋牌遊戲、捕魚機遊戲
存取速度 : 存款5秒 / 提款3-5分
軟體下載 : 支援APP,IOS、安卓(Android)
線上客服 : 需透過官方LINE
富遊娛樂城優缺點
優點 缺點
台灣註冊人數NO.1線上賭場
首儲1000贈1000只需一倍流水
擁有體驗金免費體驗賭場
網紅部落客推薦保證出金線上娛樂城
需透過客服申請體驗金
富遊娛樂城存取款方式
存款方式 取款方式
提供四大超商(全家、7-11、萊爾富、ok超商)
虛擬貨幣ustd存款
銀行轉帳(各大銀行皆可)
網站內申請提款及可匯款至綁定帳戶
現金1:1出金
富遊娛樂城平台系統
真人百家 — RG真人、DG真人、歐博真人、DB真人(原亞博/PM)、SA真人、OG真人、WM真人
體育投注 — SUPER體育、鑫寶體育、熊貓體育(原亞博/PM)
彩票遊戲 — 富遊彩票、WIN 539
電子遊戲 —RG電子、ZG電子、BNG電子、BWIN電子、RSG電子、GR電子(好路)、ATG電子
棋牌遊戲 —ZG棋牌、亞博棋牌、好路棋牌、博亞棋牌、RG棋牌
捕魚遊戲 —ZG捕魚、RSG捕魚、好路GR捕魚、DB捕魚
富遊娛樂城優惠活動
優惠 獎金贈點 流水要求
免費體驗金 $168 1倍 (儲值後) /36倍 (未儲值)
首儲贈點 $1000 1倍流水
返水活動 0.3% – 0.7% 無流水限制
簽到禮金 $666 20倍流水
好友介紹金 $688 1倍流水
回歸禮金 $500 1倍流水
免費試玩
專屬富遊VIP特權
黃金 黃金 鉑金 金鑽 大神
升級流水 300w 600w 1800w 3600w
保級流水 50w 100w 300w 600w
升級紅利 $688 $1080 $3888 $8888
每週紅包 $188 $288 $988 $2388
生日禮金 $688 $1080 $3888 $8888
反水 0.4% 0.5% 0.6% 0.7%
娛樂城常見問題解答(FAQ)
Q1:如何在富遊娛樂城進行首次存款?
A1:可以透過超商、虛擬貨幣或銀行轉帳進行存款,過程簡單快速。( 詳細可查看:富遊娛樂城存款教學)
Q2:富遊娛樂城提款需要多久時間?
A2:提款過程一般在3-5分鐘內完成,保證快速到賬。( 詳細可查看:富遊娛樂城提款教學)
Jaimeget
December 5, 2024 @ 9:07 am
buy generic clomid without insurance [url=https://clomid.store/#]how can i get cheap clomid without insurance[/url] where to get clomid
Bryanlox
December 5, 2024 @ 9:17 am
娛樂城
RG富遊娛樂城:台灣線上娛樂城的最佳選擇
RG富遊娛樂城以其卓越的服務和多樣化的遊戲選擇,成為2024年最受歡迎的線上娛樂平台。受到超過50位網紅和部落客的實測推薦,這座娛樂城不僅提供豐富的遊戲,還帶來眾多優惠活動和誠信保證,贏得了廣大玩家的信任與青睞。
RG富遊娛樂城的獨特優勢
多重優惠活動
體驗金 $168:新手玩家可以免費試玩,無需任何成本即可體驗高品質遊戲。
首儲1000送1000:首次存款即可獲得雙倍金額,增加遊戲的樂趣與機會。
線上簽到轉盤:每日簽到即可參加抽獎,贏取現金獎勵和豐厚禮品。
快速存提款與資金保障
RG富遊採用自主研發的財務系統,確保5秒快速存款,滿足玩家即時遊戲需求。
100%保證出金,杜絕任何拖延或資金安全風險,讓玩家完全放心。
遊戲種類豐富
RG富遊娛樂城涵蓋多種遊戲類型,滿足不同玩家的需求,包括:
真人百家樂:與真人荷官互動,感受真實賭場的刺激氛圍。
電子老虎機:超過數百款創新遊戲,玩法新穎,回報豐厚。
電子捕魚:趣味性強,結合策略與娛樂,深受玩家喜愛。
電子棋牌:提供公平競技環境,適合策略型玩家。
體育投注:涵蓋全球賽事,賠率即時更新,為體育愛好者提供最佳選擇。
樂透彩票:參與多地彩票,挑戰巨額獎金。
跨平台兼容性
RG富遊支持Web端、H5、iOS和Android設備,玩家可隨時隨地登錄遊戲,享受無縫體驗。
與其他娛樂城的不同之處
RG富遊以現金版模式運營,確保交易透明和安全性。相比一般娛樂城,RG富遊在存提款速度上遙遙領先,玩家可在短短15秒內完成交易,並且100%保證資金提領。而在線客服全年無休,隨時提供支持,讓玩家在任何時間都能解決問題。
相比之下,一般娛樂城多以信用版模式運營,存在出金風險,且存提款速度較慢,客服服務不穩定,無法與RG富遊的專業性相比。
為什麼選擇RG富遊娛樂城
資金交易安全無憂:採用最先進的SSL加密技術,確保每筆交易的安全性。
遊戲種類全面豐富:每日更新多樣化遊戲,帶來新鮮感和無限可能。
優惠活動力度大:從體驗金到豐厚的首儲獎勵,玩家每一步都能享受優惠。
快速存提款服務:自主研發技術保障流暢交易,遊戲不中斷。
全天候專業客服:24/7在線支持,及時解決玩家需求。
立即加入RG富遊娛樂城
RG富遊娛樂城不僅提供豐富的遊戲體驗,更以專業的服務、完善的安全保障和多樣的優惠活動,為玩家打造一個值得信賴的娛樂環境。立即註冊,體驗台灣最受歡迎的線上娛樂城!
orgone
December 5, 2024 @ 9:47 am
O conteúdo existente nesta página é realmente notável para a experiência das pessoas,
Davidbycle
December 5, 2024 @ 11:30 am
Leo娛樂城無法登入?全面解析與解決指南
近期,Leo娛樂城的玩家紛紛反映登入失敗的情況,引發了廣泛的討論與擔憂。作為九州娛樂城旗下知名品牌之一,Leo娛樂城在台灣的娛樂市場中擁有大量用戶。然而,隨著九州娛樂城宣布將於2024年12月31日退出台灣市場,這一消息無疑讓玩家對資金安全與遊戲體驗產生了極大疑慮。
本文將深入探討Leo娛樂城無法登入的原因,提供解決方案,並推薦可靠的替代娛樂平台。
Leo娛樂城無法登入的三大原因
1. 帳號密碼問題
可能情況:最常見的原因是玩家輸入了錯誤的帳號或密碼,或者帳號被盜用。
解決方法:確認輸入的帳密正確,若仍無法登入,聯繫客服即可解決。
2. 網頁技術問題
可能情況:官網可能因伺服器維護或技術故障出現404錯誤,導致玩家無法登入。
解決方法:耐心等待官網修復,並清除瀏覽器快取和Cookie,或嘗試使用其他瀏覽器。
3. 九州娛樂城退出台灣市場
可能情況:隨著九州娛樂城宣布退出台灣市場,Leo娛樂城的營運也逐步停止,導致玩家無法再使用該平台。
影響:玩家可能面臨資金提取困難,需及時完成出金操作以保障自身利益。
娛樂城無法登入的緊急解決方案
核對帳號與密碼:確認輸入正確無誤,避免由於簡單疏忽造成的登入失敗。
檢查網路連線:確保網路穩定,必要時切換至更穩定的網路環境。
清除瀏覽器快取與Cookie:解決瀏覽器相關問題,恢復正常登入。
嘗試其他瀏覽器或設備:更換設備或使用不同瀏覽器,查看是否可以正常登入。
九州娛樂城退出台灣市場的原因
市場利潤有限:九州娛樂城認為台灣市場的競爭激烈且利潤有限,因此選擇退出並專注於其他海外市場。
資金生態鏈斷裂:現金交易平台出現問題,導致九州娛樂城難以維持正常運營。
母公司法律爭議:九州娛樂城母公司近年來因財務與法律問題受到高度關注,進一步影響旗下品牌的營運。
Leo娛樂城會員如何安全出金?
在九州娛樂城停止營運前,玩家需要立即完成出金操作以確保資金安全。
出金建議:
儘快登入官網提交提款申請,確保資金安全轉出。
若官網無法訪問,請嘗試聯繫客服,並保留交易證明以備進一步處理。
推薦替代平台:富遊娛樂城
隨著Leo娛樂城的退出,玩家可以考慮轉至穩定且值得信賴的替代平台,如富遊娛樂城。
富遊娛樂城的優勢
穩定可靠:擁有先進的技術和完善的資金保障機制,確保玩家的資金安全。
豐富的遊戲選擇:從電子遊戲到真人娛樂,滿足各類玩家的需求。
專屬優惠:特別為Leo會員推出等級升等計畫,並提供多樣化的專屬活動。
快速出金:高效的財務系統,讓玩家的存取款過程快速無憂。
Leo會員轉移至富遊的流程
登錄富遊官網完成註冊。
提供Leo娛樂城會員等級截圖與相關資料(如戶名、手機號碼)給富遊客服進行審核。
等待客服確認後,享受富遊娛樂城專屬的會員福利與優惠。
結語:立即加入富遊娛樂城,延續遊戲體驗!
Leo娛樂城的退出讓許多玩家感到困惑與擔憂,但選擇穩定、安全的娛樂平台是最明智的應對之道。富遊娛樂城憑藉其卓越的技術、專業的服務以及豐富的優惠,成為玩家的理想替代選擇。現在就註冊富遊娛樂城,享受無與倫比的遊戲體驗吧!
BrianHauby
December 5, 2024 @ 11:47 am
leo娛樂城
LEO娛樂城:九州娛樂集團的卓越創舉與運營優勢
九州娛樂集團總部的現代化運營
九州娛樂集團的總部設於菲律賓首都馬尼拉市中心最具代表性的現代化建築「RCBC Plaza」。這座辦公大樓融合了全球領先的建築科技與安全系統,包括:
國家級資訊防護系統,確保數據安全。
樓宇自動化系統 (BAS),實現數位化管理與高效運營,包括防火系統、光纖電訊和數位監控。
九州娛樂為員工提供舒適的工作環境和專業的教育培訓,確保服務水準一流。其員工以專業知識和親切態度,為尊貴玩家帶來極致的遊戲體驗。
LEO娛樂旗下品牌與遊戲特色
九州娛樂城旗下分支包括 LEO、THA 和 KU 三個娛樂品牌。雖然品牌各自獨立運營,但都共享相同的遊戲系統與界面,提供超過數千款來自全球頂級供應商的遊戲。
這些品牌的核心特色包括:
種類繁多的遊戲:滿足不同類型玩家的需求,包括電子遊戲、體育博彩、真人娛樂等。
不斷更新的內容:通過九州獨家技術,網站內容保持新鮮,合作商可快速更新遊戲資源,確保平台競爭力。
玩家在這些平台上可以隨時找到適合自己的遊戲,享受沉浸式的娛樂體驗。
LEO娛樂城的歷史與發展
九州娛樂集團的起源可以追溯至其最早期的經營模式:「信用版」賭博平台 《天下體育球版》。儘管該模式在當時非常流行,但因高風險與財務處理複雜而逐漸被淘汰。
隨著市場需求的變化,九州娛樂轉型為現金版平台 《天下現金網》,引入了更安全、便捷的遊戲模式。這種轉變不僅提升了玩家的體驗,也使平台得以進一步發展。
如今,九州娛樂城已成為台灣博弈業界的領軍者,不僅優化了網站版面與功能,還推出了專屬手機APP,方便玩家隨時隨地進行遊戲體驗。
LEO娛樂的成功秘訣
現代化運營與技術支持:九州娛樂在系統安全與技術更新方面投入巨大資源,確保平台運營穩定可靠。
多元化遊戲體驗:通過與全球頂級遊戲供應商合作,平台提供了豐富的遊戲選擇,適應各種玩家需求。
用戶至上:專業培訓的員工以細心和熱忱態度,提供高品質服務,讓玩家倍感尊重。
不斷創新:從信用版到現金版,再到手機APP,九州娛樂始終緊跟市場趨勢,致力於打造最佳娛樂體驗。
結語
LEO娛樂城作為九州娛樂集團旗下的重要品牌,不僅繼承了集團的核心價值,更在遊戲選擇、服務品質和技術創新方面不斷提升。從最初的《天下體育球版》到如今的行業領先者,LEO娛樂見證了九州娛樂城的發展歷程,也成為博弈行業中的耀眼明星。
加入 LEO娛樂城,感受來自九州娛樂集團的專業與熱情,享受最頂級的線上博弈體驗!
BrandonInalf
December 5, 2024 @ 11:49 am
九州Leo娛樂城無法登入
Leo娛樂城無法登入?全面解析與解決指南
近期,Leo娛樂城的玩家紛紛反映登入失敗的情況,引發了廣泛的討論與擔憂。作為九州娛樂城旗下知名品牌之一,Leo娛樂城在台灣的娛樂市場中擁有大量用戶。然而,隨著九州娛樂城宣布將於2024年12月31日退出台灣市場,這一消息無疑讓玩家對資金安全與遊戲體驗產生了極大疑慮。
本文將深入探討Leo娛樂城無法登入的原因,提供解決方案,並推薦可靠的替代娛樂平台。
Leo娛樂城無法登入的三大原因
1. 帳號密碼問題
可能情況:最常見的原因是玩家輸入了錯誤的帳號或密碼,或者帳號被盜用。
解決方法:確認輸入的帳密正確,若仍無法登入,聯繫客服即可解決。
2. 網頁技術問題
可能情況:官網可能因伺服器維護或技術故障出現404錯誤,導致玩家無法登入。
解決方法:耐心等待官網修復,並清除瀏覽器快取和Cookie,或嘗試使用其他瀏覽器。
3. 九州娛樂城退出台灣市場
可能情況:隨著九州娛樂城宣布退出台灣市場,Leo娛樂城的營運也逐步停止,導致玩家無法再使用該平台。
影響:玩家可能面臨資金提取困難,需及時完成出金操作以保障自身利益。
娛樂城無法登入的緊急解決方案
核對帳號與密碼:確認輸入正確無誤,避免由於簡單疏忽造成的登入失敗。
檢查網路連線:確保網路穩定,必要時切換至更穩定的網路環境。
清除瀏覽器快取與Cookie:解決瀏覽器相關問題,恢復正常登入。
嘗試其他瀏覽器或設備:更換設備或使用不同瀏覽器,查看是否可以正常登入。
九州娛樂城退出台灣市場的原因
市場利潤有限:九州娛樂城認為台灣市場的競爭激烈且利潤有限,因此選擇退出並專注於其他海外市場。
資金生態鏈斷裂:現金交易平台出現問題,導致九州娛樂城難以維持正常運營。
母公司法律爭議:九州娛樂城母公司近年來因財務與法律問題受到高度關注,進一步影響旗下品牌的營運。
Leo娛樂城會員如何安全出金?
在九州娛樂城停止營運前,玩家需要立即完成出金操作以確保資金安全。
出金建議:
儘快登入官網提交提款申請,確保資金安全轉出。
若官網無法訪問,請嘗試聯繫客服,並保留交易證明以備進一步處理。
推薦替代平台:富遊娛樂城
隨著Leo娛樂城的退出,玩家可以考慮轉至穩定且值得信賴的替代平台,如富遊娛樂城。
富遊娛樂城的優勢
穩定可靠:擁有先進的技術和完善的資金保障機制,確保玩家的資金安全。
豐富的遊戲選擇:從電子遊戲到真人娛樂,滿足各類玩家的需求。
專屬優惠:特別為Leo會員推出等級升等計畫,並提供多樣化的專屬活動。
快速出金:高效的財務系統,讓玩家的存取款過程快速無憂。
Leo會員轉移至富遊的流程
登錄富遊官網完成註冊。
提供Leo娛樂城會員等級截圖與相關資料(如戶名、手機號碼)給富遊客服進行審核。
等待客服確認後,享受富遊娛樂城專屬的會員福利與優惠。
結語:立即加入富遊娛樂城,延續遊戲體驗!
Leo娛樂城的退出讓許多玩家感到困惑與擔憂,但選擇穩定、安全的娛樂平台是最明智的應對之道。富遊娛樂城憑藉其卓越的技術、專業的服務以及豐富的優惠,成為玩家的理想替代選擇。現在就註冊富遊娛樂城,享受無與倫比的遊戲體驗吧!
AndresBAP
December 5, 2024 @ 12:00 pm
娛樂城
2024 年娛樂城遊戲推薦:最夯遊戲與最佳平台選擇
隨著線上娛樂城市場的蓬勃發展,2024 年推出了多款令人興奮的遊戲和創新平台。無論是喜歡老虎機、棋牌遊戲,還是捕魚和彩票遊戲的玩家,都能在今年找到屬於自己的遊戲樂園。以下是熱門遊戲推薦與最佳娛樂城平台的詳細介紹。
2024 年最夯遊戲推薦
電子老虎機遊戲
電子老虎機一直是娛樂城的經典項目,今年更是推出了數款熱門作品:
戰神賽特:以古埃及神話為背景,擁有高倍數獎勵的特殊功能,畫面設計精美。
魔龍傳奇:玩家可探索魔龍世界,觸發驚喜獎金和免費遊戲機會。
雷神之錘:以北歐神話為主題,特色連線玩法讓每次旋轉都充滿期待。
金虎爺:寓意招財進寶,特別適合新年期間挑戰,玩法簡單但回報豐厚。
棋牌遊戲
二人麻將:適合喜歡策略性對抗的玩家,快速節奏且競爭激烈。
忍 Kunoichi:結合武士與忍者元素,讓玩家體驗獨特的棋牌競技。
捕魚遊戲
捕魚遊戲是一種結合策略和運氣的娛樂項目,玩家可以通過捕捉魚群來獲得高額獎金,同時享受視覺和操作的雙重樂趣。
彩票遊戲
彩票遊戲以簡單快捷的玩法吸引大批玩家,提供多種國內外彩種,讓玩家隨時挑戰幸運。
RG富遊娛樂城:2024 年最佳線上娛樂城推薦
在眾多娛樂城平台中,RG富遊娛樂城以其專業服務和卓越遊戲體驗,成為今年的首選平台。
平台特色
遊戲種類豐富
提供多元化遊戲選擇,包括真人視訊、電子老虎機、捕魚、棋牌和彩票遊戲,滿足所有玩家需求。
快速便捷的存提款服務
平台支持即時存款與快速提款,無需等待,確保資金安全流動。
高度安全保障
採用最先進的加密技術,保護玩家的帳戶與交易安全。
支援多平台裝置
富遊娛樂城APP 可在 iOS 和 Android 設備上使用,界面簡潔易操作,隨時隨地享受娛樂。
專業客服支援
提供 24 小時全天候客服,隨時解答玩家疑問,保障流暢的遊戲體驗。
如何開始體驗 RG 富遊娛樂城
下載富遊 APP
在官方網站免費下載 APP,支援 iOS 和 Android 設備。
註冊帳號
使用簡單步驟完成註冊,立即進入遊戲世界。
挑戰熱門遊戲
從老虎機到棋牌遊戲,選擇您喜歡的類型,挑戰高額獎金。
結語
2024 年是線上娛樂遊戲的精彩年份,從戰神賽特到二人麻將,每款遊戲都能帶給玩家不同的樂趣。而選擇像 RG富遊娛樂城 這樣的專業平台,不僅能體驗到豐富的遊戲內容,還能享受安全可靠的服務。立即下載富遊 APP,加入這個充滿驚喜的娛樂世界
MichaelMug
December 5, 2024 @ 1:46 pm
滿天星娛樂城:全方位的線上娛樂體驗
滿天星娛樂城作為九州娛樂城旗下品牌之一,是玩家心目中備受信賴的線上娛樂平台。不僅提供豐富的遊戲選擇和吸引人的優惠,還以其合法營運的背景和快速出金的保證,成為台灣玩家的首選娛樂城之一。
滿天星娛樂城的特色與優勢
1. 合法營運,安全可靠
滿天星娛樂城持有三大國際娛樂執照,包括:
菲律賓 PAGCOR 監督競猜牌照
BVI 認證
馬爾他 MGA 認證
這些執照展現了平台的資本實力與合法性,讓玩家能夠安心享受遊戲,完全不用擔心出金問題。
2. 高品質遊戲與豐富優惠
滿天星娛樂城與九州集團旗下的LEO、THA娛樂城一樣,提供一流的遊戲品質和多樣化的優惠。
熱門遊戲推薦:電子老虎機、真人直播、運彩投注、今彩539等,滿足各類玩家需求。
優惠活動:定期推出限時回饋活動,讓玩家在遊戲中獲得更多額外收益。
3. 快速出金與高效服務
滿天星娛樂城採用先進的財務系統,確保存提款快速、安全,讓玩家能夠專注於享受遊戲,而無需擔心資金問題。專業的客服團隊24小時在線,提供即時支援。
4. 多平台兼容的APP
滿天星娛樂城提供設計貼合玩家需求的手機APP,適用於iOS與Android系統。玩家可以隨時隨地快速登入遊玩,體驗無與倫比的娛樂快感。
熱門遊戲類型一覽
滿天星娛樂城為玩家提供多樣化的遊戲選擇,滿足不同興趣和需求:
真人直播:感受真實賭場氛圍,與真人荷官互動,體驗沉浸式娛樂。
電子老虎機:多款經典與創新機台,讓玩家享受刺激的遊戲過程。
運彩投注:涵蓋世界盃、棒球、籃球等多項運動賽事,提供即時投注與直播功能。
彩票遊戲:包括今彩539、富遊彩票等,適合熱衷於彩券的玩家。
滿天星娛樂城常見問題解答
Q1. 滿天星娛樂城是否提供APP下載?
是的,滿天星娛樂城提供專屬的手機APP,支援iOS和Android系統。玩家可透過APP快速登入,隨時享受遊戲。
Q2. 滿天星娛樂城是否合法?
滿天星娛樂城擁有多項國際認證,為合法經營的線上娛樂城,並保證玩家資金的安全性與遊戲的公平性。
Q3. 無法登入滿天星娛樂城該怎麼辦?
如遇到無法登入的情況,建議玩家檢查網路狀況或聯繫24小時客服團隊獲取即時協助。
立即加入滿天星娛樂城,享受最佳娛樂體驗!
滿天星娛樂城以合法、安全、快速的服務,搭配豐富的遊戲選擇和優惠,為玩家打造出獨一無二的線上娛樂體驗。不論您是新手還是老手,都能在滿天星找到屬於自己的遊戲樂趣。現在就下載APP,隨時隨地開始您的娛樂之旅!
DannyGop
December 5, 2024 @ 4:54 pm
娛樂城
富遊娛樂城:台灣線上賭場的創新選擇
在台灣的線上娛樂市場中,富遊娛樂城憑藉其優質的服務和多樣化的娛樂選項,迅速成為新一代玩家的首選。既有刺激的遊戲體驗,又提供了一系列具有吸引力的優惠,富遊娛樂城無疑是追求樂趣的玩家們理想的地方。
#### 優勢與特色
1. 多樣化的遊戲選擇
富遊娛樂城提供了豐富的遊戲種類,包括電子老虎機、真人百家樂、運彩投注等。不論你是老虎機的愛好者,還是熱衷於真人賭桌的玩家,這裡都能滿足你的需求。特別推薦的遊戲如DG真人百家和RSG皇家百家樂,一定能帶給你極致的遊戲體驗。
2. 吸引人的優惠活動
富遊娛樂城針對新手和老玩家都推出了多樣的優惠活動。新會員首次存款可獲得相同金額的獎金,還有額外的體驗金送出,設計友好且實惠。此外,富遊每週的簽到獎勵和天天無上限的返水活動,更是讓玩家在遊戲中能持續獲得收益。
3. 安全可靠的存取款方式
在金融交易方面,富遊娛樂城支持多種存款和提款方式,包括各大銀行轉帳、超商儲值,甚至虛擬貨幣等。最低提款金額設置靈活,保證玩家資金的安全與便捷。
4. 人性化的操作介面
富遊娛樂城的網頁與移動應用設計簡潔易用,玩家可以方便地找到所需的遊戲和優惠。無論是在電腦還是手機上,都能順利享受到遊戲的樂趣。
#### 如何開始遊戲
如果你是新手玩家,開始你的富遊體驗非常簡單。只需註冊帳號,完成首次存款,即可獲得新手優惠。無論你想嘗試電子遊戲還是真人賭局,富遊娛樂城都能讓你輕鬆上手,享受娛樂的樂趣。
#### 社會責任與防沉迷措施
富遊娛樂城深知線上賭博的風險,因此致力於推廣負責任的遊戲文化。他們提供“防沉迷”指導,以確保玩家能夠理性投注,並提供必要的幫助和資源來幫助有需求的玩家。
#### 總結
對於尋求安全、便捷和多元化娛樂城體驗的玩家,富遊娛樂城絕對是值得選擇的選項。無論你是希望徹底放鬆,還是想要體驗刺激的博弈過程,富遊娛樂城都能滿足你的期待,成為你在台灣線上娛樂市場的最佳夥伴。立即註冊並開始你的遊戲之旅吧!
apkportalnik
December 5, 2024 @ 5:18 pm
закачать приложения онлайн казино https://villaamericanaeventos.com.br/2024/11/19/kazino-bez-vlozhenij-2024-igrat-onlajn-v-igrovyh-3/
Robertvox
December 5, 2024 @ 5:45 pm
how to get zithromax online: buy zithromax 1000 mg online – cheap zithromax pills
RobertDup
December 5, 2024 @ 5:46 pm
LEO娛樂城無法登入!九州娛樂宣布退出台灣市場
近期,LEO娛樂城和THA娛樂城的玩家發現頻繁出現無法登入的情況,引發了廣泛討論。九州娛樂城官方也正式宣布,將於2024年12月31日中午12:00停止在台灣的營運,並結束所有相關服務。這一消息對於忠實玩家來說無疑是一大震撼,為什麼九州娛樂城會退出台灣市場?玩家該如何應對?本文將為您逐一解析。
LEO娛樂城無法登入的原因
1. 帳號與密碼問題
最常見的情況是玩家輸入的帳號或密碼有誤,或者帳號遭到盜用。
解決方案:聯繫LEO娛樂城的客服,提供必要的驗證信息,即可快速恢復帳戶。
2. 網站技術故障
LEO娛樂城可能因官網維護或伺服器異常導致無法正常運作。
解決方案:耐心等待技術問題修復,通常不會影響長時間使用。
3. 九州娛樂退出台灣市場
LEO娛樂城隸屬於九州娛樂集團,近期官方公告將於2024年12月31日結束營運,退出台灣市場。這是導致登入失敗的最終原因,玩家將無法再使用該平台的服務。
九州娛樂城退出台灣市場的原因分析
1. 市場規模限制
九州娛樂認為台灣市場過於狹小,品牌競爭激烈,成長空間有限。相比之下,海外市場不僅擁有更大的人口基數,也提供更高的獲利潛力。九州旗下的KU體育曾在國際足球五大聯賽中曝光,這顯示其具備強大的國際競爭力,轉向海外市場發展成為合理選擇。
2. 金流問題與法規壓力
LEO娛樂城的運營高度依賴第三方金流公司,而近期陳政谷案件的爆發使整個金流生態鏈受到嚴重打擊。警方揭露,九州娛樂集團利用第三方支付進行資金洗錢,甚至牽涉到內部人員洩密,對整個集團的形象和運營造成巨大衝擊。
3. 品牌聲譽受損
隨著案件的曝光,九州娛樂集團的聲譽遭到前所未有的挑戰。為了平息風波,九州選擇退出台灣市場,以避免進一步的法律與政策風險。
九州娛樂城停止營運的公告
根據LEO娛樂城官方的公告,九州娛樂城將於2024年12月31日中午12:00全面停止台灣市場的營運:
停止存款功能:玩家將無法再進行任何存款操作。
正常開放提款:官方承諾保證提款功能正常運作,會員可安心提取資金。
感謝支持:九州娛樂對於玩家多年來的支持表示衷心感謝,並祝福所有會員未來一切順利。
玩家應對策略:如何保護自身利益?
1. 儘速完成提款
如您的帳戶仍可正常登入,建議立即提交提款申請,確保資金安全轉出。
2. 提高警覺,避免詐騙
隨著九州娛樂退出,市場可能會出現冒充LEO娛樂城或THA娛樂城的假冒平台,玩家需保持高度警覺。
3. 選擇可靠的替代平台
對於希望延續遊戲體驗的玩家,可以考慮轉向信譽良好的國際娛樂平台,例如Bet365台灣,該平台提供穩定的服務和多樣化的遊戲選擇,是九州娛樂的理想替代方案。
結語:九州娛樂的結束,玩家的新起點
九州娛樂城的退出標誌著一個時代的結束,但對於玩家而言,也是一個選擇新平台的契機。未來,選擇安全、穩定且合法的娛樂平台將成為玩家的首要考量。無論如何,希望所有玩家都能在新的平台中繼續享受遊戲的樂趣,保障自身利益不受損失!
Is This Finally Our Chance to Find Jobs We Love?
December 5, 2024 @ 5:50 pm
Very nice write-up. I certainly appreciate this website. Keep writing!
Jaimeget
December 5, 2024 @ 6:37 pm
cheap clomid without a prescription [url=https://clomid.store/#]buy generic clomid prices[/url] can you get cheap clomid without a prescription
Matthewpok
December 5, 2024 @ 10:52 pm
https://ciprofloxacin.cheap/# buy cipro without rx
ciprofloxacin generic [url=http://ciprofloxacin.cheap/#]cipro 500mg best prices[/url] cipro for sale
Jaimeget
December 6, 2024 @ 3:54 am
zestoretic price [url=https://lisinoprilus.com/#]lisinopril online without a prescription[/url] price of zestril 30 mg
Matthewsic
December 6, 2024 @ 5:11 am
не знаете, как найти актуальную ссылку vavada? ссылка https://t.me/qvavada решит вашу проблему. подпишитесь на наш телеграм-канал и получите доступ ко всем бонусам. теперь ваш выигрыш на расстоянии пары секунд!
взломостойкий сейф
December 6, 2024 @ 6:44 am
Тут можно преобрести сейфы взломостойкие взломостойкий сейф для дома
RollandHeila
December 6, 2024 @ 8:44 am
九州娛樂城倒了
九州娛樂城退出台灣市場:LEO娛樂城無法登入的背後真相
LEO娛樂城,作為台灣線上娛樂城市場的重要品牌,長期以來在廣告投放和代理推廣方面占據領先地位。然而,近期出現的「LEO娛樂城無法登入」現象引發了廣泛的網路討論。不少玩家質疑:「九州娛樂城是不是倒了?」「這是不是詐騙?」事實上,這些猜測與謠言並不完全準確。根據最新消息,九州娛樂城已決定結束其在台灣的業務,包括旗下品牌LEO娛樂城和THA娛樂城。以下我們將深入分析這一事件的背景和可能的影響。
LEO娛樂城無法登入的原因分析
近期LEO娛樂城頻繁出現無法登入的情況,其實是九州娛樂城退出台灣市場的前兆。根據官方公告,九州娛樂城將於2024年12月31日中午12點正式關閉,包括註冊與存款功能在內的多項服務已經受到限制。
可能原因 1:退出台灣市場的策略
九州娛樂城的管理層認為,台灣市場的利潤已達上限,長期運營的收益減少,因此選擇「見好就收」。這一策略符合台灣商業文化中尋找新市場發展的特點。九州娛樂城可能計劃將資源投入其他更具潛力的市場,以追求更大的收益。
可能原因 2:運營生態鏈的問題
LEO娛樂城的運營高度依賴第三方現金交易平台以及相關支付管道。近期,這些環節可能出現了無法解決的問題,導致整體運營受阻。為了避免損失擴大,九州娛樂城選擇退出台灣市場。
可能原因 3:法律風險與負面新聞
九州娛樂城因涉及法律事件,曾多次登上新聞版面。這些事件不僅損害了品牌形象,也讓經營面臨更多風險。為了降低法律糾紛帶來的影響,九州娛樂城可能選擇結束在台灣的營運。
官方公告:2024年12月31日正式停業
九州娛樂城已向所有LEO娛樂城會員發布公告,明確指出將於2024年12月31日中午12點全面停止營運。公告內容包括以下重要信息:
存款服務已關閉:玩家無法繼續充值。
提款功能仍正常:玩家可以放心提取餘額,會員權益不受影響。
感謝支持:官方對會員表達了感謝,並祝福大家未來順利。
這一公告讓玩家可以在剩餘時間內完成提款操作,避免資金損失。
對玩家的影響與建議
1. 儘快提款
如果您是LEO娛樂城的會員,應立即登入帳戶並提取餘額,以確保資金安全。
2. 謹慎選擇新平台
選擇新的娛樂平台時,應注意以下幾點:
合法性:確認平台是否擁有合法經營許可。
支付系統穩定性:確保提款及充值渠道安全無虞。
用戶評價:參考其他玩家的反饋,選擇口碑良好的平台。
3. 防範詐騙
警惕假冒的LEO娛樂城網站或客服。官方客服系統已關閉,任何聯繫您索取個人資料的行為都可能是詐騙。
結語
LEO娛樂城無法登入以及九州娛樂城退出台灣市場,標誌著線上娛樂城行業的一次重要轉折。對於玩家而言,這既是一個挑戰,也是一個重新審視平台選擇的機會。未來,玩家應更加關注平台的合法性與穩定性,為自己的遊戲體驗和資金安全提供保障。
Jimmieclict
December 6, 2024 @ 9:13 am
https://clomid.store/# can i order clomid without prescription
ArmandoWal
December 6, 2024 @ 10:05 am
buy zithromax online fast shipping: where can i purchase zithromax online – azithromycin zithromax
StevenSmose
December 6, 2024 @ 11:05 am
3A娛樂城:全方位線上娛樂體驗的首選平台
3A娛樂城作為台灣最受歡迎的線上娛樂平台之一,以其多樣化的遊戲選擇、創新的技術、以及卓越的安全保障,贏得了玩家的高度信任與青睞。無論您是新手還是老手,3A娛樂城都能滿足您的需求,帶給您獨一無二的遊戲體驗。
熱門娛樂城遊戲一覽
3A娛樂城提供多種熱門遊戲,適合不同類型的玩家:
真人百家樂:體驗真實賭場的氛圍,與真人荷官互動,感受賭桌上的緊張與刺激。
彩票投注:涵蓋多種彩票選項,滿足彩券愛好者的需求,讓您隨時隨地挑戰幸運。
棋牌遊戲:從傳統的麻將到現代化的紙牌遊戲,讓玩家在策略與運氣中找到平衡。
3A娛樂城的核心價值
1. 專業
3A娛樂城每日提供近千場體育賽事,並搭配真人百家樂、彩票彩球、電子遊戲等多種類型的線上賭場遊戲。無論您的遊戲偏好為何,這裡總有一款適合您!
2. 安全
平台採用128位加密技術和嚴格的安全管理體系,確保玩家的金流與個人資料完全受保護,讓您可以放心遊玩,無需擔憂安全問題。
3. 便捷
3A娛樂城是全台第一家使用自家開發的全套終端應用的娛樂城平台。無論是手機還是電腦,玩家都能享受無縫的遊戲體驗。同時,24小時線上客服隨時為您解決任何問題,提供貼心服務。
4. 安心
由專業工程師開發的財務處理系統,為玩家帶來快速的存款、取款和轉帳服務。透過獨立網路技術,平台提供優化的網路速度,確保每一次操作都流暢無比。
下載3A娛樂城手機APP
為了提供更便捷的遊戲體驗,3A娛樂城獨家開發了功能齊全的手機APP,讓玩家隨時隨地開啟遊戲。
現代化設計:新穎乾淨的介面設計,提升使用者體驗。
跨平台支持:完美適配手機與電腦,讓您輕鬆切換設備,無需中斷遊戲。
快速下載:掃描專屬QR碼即可進入下載頁面,立即開始暢玩!
3A娛樂城的其他服務
娛樂城教學:詳細的操作指導,讓新手也能快速上手。
責任博彩:提倡健康娛樂,確保玩家在遊戲中享受樂趣的同時,不失理性。
隱私權政策:嚴格遵守隱私保護條例,保障玩家的個人信息安全。
立即加入3A娛樂城,享受頂級娛樂體驗!
如果您正在尋找一個專業、安全、便捷又安心的線上娛樂平台,那麼3A娛樂城絕對是您的不二之選。現在就下載手機APP,隨時隨地開啟您的娛樂旅程,享受無限的遊戲樂趣與刺激!
https://da88.video/
December 6, 2024 @ 12:05 pm
May I simply just say what a relief to find a person that
really understands what they are talking about on the
web. You definitely know how to bring a problem to light and make it
important. More people ought to read this and understand this
side of your story. I was surprised you’re not more popular given that you most certainly possess the gift.
segno zodiacale del leone
December 6, 2024 @ 12:10 pm
webside er virkelig bemærkelsesværdig for folks oplevelse, godt,
DonaldReunk
December 6, 2024 @ 12:13 pm
冠天下娛樂城
2024 年熱門娛樂城與活動解析
冠天下娛樂城優惠,帶來無限樂趣
在 2024 年 4 月 16 日,冠天下娛樂城推出了全新的優惠活動,旨在為玩家帶來更大的獲利機會和更高的娛樂體驗。這些優惠活動包括多種存款獎勵、會員專屬回饋以及豐富的遊戲競賽獎金,吸引了大批玩家參與。
冠天下娛樂城以其高透明度和用戶友好的政策,已成為許多玩家的首選娛樂平台。不論是新手還是老手,都能在這裡找到適合自己的遊戲和活動。
LEO娛樂城無法登入引發市場恐慌
2024 年 11 月 29 日,LEO娛樂城因技術問題出現短暫無法登入的情況,引發了廣泛關注。許多玩家擔心這是否意味著九州娛樂城的營運出現問題。
九州娛樂集團隨後發佈官方聲明,強調這僅僅是系統升級過程中的短暫問題,並承諾玩家的資金和數據安全不受任何影響。同時,九州娛樂表示將加強伺服器穩定性,確保未來類似情況不再發生。
熱門 ZG 電子遊戲——熱舞森巴老虎機
2024 年 11 月 8 日,ZG 電子遊戲推出了熱舞森巴老虎機,這款遊戲迅速成為市場上的熱門選擇。以南美熱情的森巴舞為主題,遊戲畫面充滿色彩,搭配動感的音樂,為玩家帶來視覺與聽覺的雙重享受。
遊戲亮點:
高倍數獎勵機制:每次轉盤都有機會觸發高額獎金。
特色免費遊戲:連續獲得特殊圖案即可啟動多次免費轉盤,提高獲勝機率。
適合新手與高端玩家:簡單的玩法規則與豐富的策略選擇滿足不同層級玩家的需求。
ZG 電子遊戲致力於為玩家提供創新且有趣的遊戲體驗,熱舞森巴老虎機無疑是今年最值得嘗試的遊戲之一。
2024 世界棒球 12 強賽精彩回顧
2024 年 11 月,中華隊參加了世界棒球 12 強賽,這項全球頂尖棒球賽事吸引了來自多國的球隊和球迷參與。中華隊 28 人的名單以年輕選手和經驗豐富的老將為基礎,展現了團隊的競技實力。
比賽亮點:
精彩賽程:比賽期間,中華隊展現了堅強的防守和出色的打擊表現。
門票銷售:賽事門票迅速售罄,顯示出棒球在台灣的高度受歡迎程度。
線上直播:各大平台提供了高畫質直播,讓更多球迷能夠即時觀看比賽,感受棒球魅力。
世界棒球 12 強賽不僅是比賽,更是全台灣球迷的一場盛宴,展現了棒球作為國民運動的無限魅力。
結語
從娛樂城的優惠活動到全球性的棒球賽事,2024 年對於遊戲和體育愛好者來說充滿了亮點。不論是參與冠天下娛樂城的優惠,體驗 ZG 電子遊戲的最新老虎機,還是為中華隊的棒球比賽加油,這一年都是充滿精彩與期待的一年!
Matthewpok
December 6, 2024 @ 12:19 pm
https://ciprofloxacin.cheap/# buy cipro no rx
zithromax online usa no prescription [url=https://azithromycinus.com/#]generic zithromax azithromycin[/url] zithromax canadian pharmacy
Robertvox
December 6, 2024 @ 12:53 pm
zithromax capsules 250mg: zithromax 500 without prescription – generic zithromax india
Jaimeget
December 6, 2024 @ 1:11 pm
buy cytotec online fast delivery [url=http://cytotec.top/#]cytotec online[/url] cytotec abortion pill
ArmandoWal
December 6, 2024 @ 4:55 pm
lisinopril 2.5 mg for sale: lisinopril 20mg coupon – lisinopril 10 mg no prescription
Jimmieclict
December 6, 2024 @ 5:45 pm
https://cytotec.top/# cytotec online
Jaimeget
December 6, 2024 @ 10:23 pm
ciprofloxacin 500 mg tablet price [url=https://ciprofloxacin.cheap/#]buy cipro without rx[/url] buy cipro online canada
ArmandoWal
December 6, 2024 @ 11:26 pm
get clomid: how to get clomid price – can i get generic clomid prices
JamieFow
December 6, 2024 @ 11:39 pm
2024 年娛樂城遊戲推薦:最夯遊戲與最佳平台選擇
隨著線上娛樂城市場的蓬勃發展,2024 年推出了多款令人興奮的遊戲和創新平台。無論是喜歡老虎機、棋牌遊戲,還是捕魚和彩票遊戲的玩家,都能在今年找到屬於自己的遊戲樂園。以下是熱門遊戲推薦與最佳娛樂城平台的詳細介紹。
2024 年最夯遊戲推薦
電子老虎機遊戲
電子老虎機一直是娛樂城的經典項目,今年更是推出了數款熱門作品:
戰神賽特:以古埃及神話為背景,擁有高倍數獎勵的特殊功能,畫面設計精美。
魔龍傳奇:玩家可探索魔龍世界,觸發驚喜獎金和免費遊戲機會。
雷神之錘:以北歐神話為主題,特色連線玩法讓每次旋轉都充滿期待。
金虎爺:寓意招財進寶,特別適合新年期間挑戰,玩法簡單但回報豐厚。
棋牌遊戲
二人麻將:適合喜歡策略性對抗的玩家,快速節奏且競爭激烈。
忍 Kunoichi:結合武士與忍者元素,讓玩家體驗獨特的棋牌競技。
捕魚遊戲
捕魚遊戲是一種結合策略和運氣的娛樂項目,玩家可以通過捕捉魚群來獲得高額獎金,同時享受視覺和操作的雙重樂趣。
彩票遊戲
彩票遊戲以簡單快捷的玩法吸引大批玩家,提供多種國內外彩種,讓玩家隨時挑戰幸運。
RG富遊娛樂城:2024 年最佳線上娛樂城推薦
在眾多娛樂城平台中,RG富遊娛樂城以其專業服務和卓越遊戲體驗,成為今年的首選平台。
平台特色
遊戲種類豐富
提供多元化遊戲選擇,包括真人視訊、電子老虎機、捕魚、棋牌和彩票遊戲,滿足所有玩家需求。
快速便捷的存提款服務
平台支持即時存款與快速提款,無需等待,確保資金安全流動。
高度安全保障
採用最先進的加密技術,保護玩家的帳戶與交易安全。
支援多平台裝置
富遊娛樂城APP 可在 iOS 和 Android 設備上使用,界面簡潔易操作,隨時隨地享受娛樂。
專業客服支援
提供 24 小時全天候客服,隨時解答玩家疑問,保障流暢的遊戲體驗。
如何開始體驗 RG 富遊娛樂城
下載富遊 APP
在官方網站免費下載 APP,支援 iOS 和 Android 設備。
註冊帳號
使用簡單步驟完成註冊,立即進入遊戲世界。
挑戰熱門遊戲
從老虎機到棋牌遊戲,選擇您喜歡的類型,挑戰高額獎金。
結語
2024 年是線上娛樂遊戲的精彩年份,從戰神賽特到二人麻將,每款遊戲都能帶給玩家不同的樂趣。而選擇像 RG富遊娛樂城 這樣的專業平台,不僅能體驗到豐富的遊戲內容,還能享受安全可靠的服務。立即下載富遊 APP,加入這個充滿驚喜的娛樂世界
DavidHam
December 7, 2024 @ 12:16 am
滿天星娛樂城:全方位的線上娛樂體驗
滿天星娛樂城作為九州娛樂城旗下品牌之一,是玩家心目中備受信賴的線上娛樂平台。不僅提供豐富的遊戲選擇和吸引人的優惠,還以其合法營運的背景和快速出金的保證,成為台灣玩家的首選娛樂城之一。
滿天星娛樂城的特色與優勢
1. 合法營運,安全可靠
滿天星娛樂城持有三大國際娛樂執照,包括:
菲律賓 PAGCOR 監督競猜牌照
BVI 認證
馬爾他 MGA 認證
這些執照展現了平台的資本實力與合法性,讓玩家能夠安心享受遊戲,完全不用擔心出金問題。
2. 高品質遊戲與豐富優惠
滿天星娛樂城與九州集團旗下的LEO、THA娛樂城一樣,提供一流的遊戲品質和多樣化的優惠。
熱門遊戲推薦:電子老虎機、真人直播、運彩投注、今彩539等,滿足各類玩家需求。
優惠活動:定期推出限時回饋活動,讓玩家在遊戲中獲得更多額外收益。
3. 快速出金與高效服務
滿天星娛樂城採用先進的財務系統,確保存提款快速、安全,讓玩家能夠專注於享受遊戲,而無需擔心資金問題。專業的客服團隊24小時在線,提供即時支援。
4. 多平台兼容的APP
滿天星娛樂城提供設計貼合玩家需求的手機APP,適用於iOS與Android系統。玩家可以隨時隨地快速登入遊玩,體驗無與倫比的娛樂快感。
熱門遊戲類型一覽
滿天星娛樂城為玩家提供多樣化的遊戲選擇,滿足不同興趣和需求:
真人直播:感受真實賭場氛圍,與真人荷官互動,體驗沉浸式娛樂。
電子老虎機:多款經典與創新機台,讓玩家享受刺激的遊戲過程。
運彩投注:涵蓋世界盃、棒球、籃球等多項運動賽事,提供即時投注與直播功能。
彩票遊戲:包括今彩539、富遊彩票等,適合熱衷於彩券的玩家。
滿天星娛樂城常見問題解答
Q1. 滿天星娛樂城是否提供APP下載?
是的,滿天星娛樂城提供專屬的手機APP,支援iOS和Android系統。玩家可透過APP快速登入,隨時享受遊戲。
Q2. 滿天星娛樂城是否合法?
滿天星娛樂城擁有多項國際認證,為合法經營的線上娛樂城,並保證玩家資金的安全性與遊戲的公平性。
Q3. 無法登入滿天星娛樂城該怎麼辦?
如遇到無法登入的情況,建議玩家檢查網路狀況或聯繫24小時客服團隊獲取即時協助。
立即加入滿天星娛樂城,享受最佳娛樂體驗!
滿天星娛樂城以合法、安全、快速的服務,搭配豐富的遊戲選擇和優惠,為玩家打造出獨一無二的線上娛樂體驗。不論您是新手還是老手,都能在滿天星找到屬於自己的遊戲樂趣。現在就下載APP,隨時隨地開始您的娛樂之旅!
Jimmieclict
December 7, 2024 @ 2:13 am
https://ciprofloxacin.cheap/# ciprofloxacin generic price
Alfredoglava
December 7, 2024 @ 2:22 am
想要享受最刺激的線上娛樂體驗?選擇RG富遊娛樂城!
在這個數位時代,尋找適合自己的線上娛樂城變得越來越重要。如果你想在線上娛樂城找到最好玩、最受歡迎的遊戲,RG富遊絕對是你的首選!我們匯聚了多款博弈遊戲,讓您盡情享受娛樂的刺激與快感。
#### 精選遊戲類型
1. 真人百家樂
感受現場賭場的氛圍!RG富遊的真人視訊百家樂讓你與真實的荷官互動,享受高品質的遊戲體驗。無論你是老手還是新手,都能在這裡找到適合自己的節奏。
2. 電子老虎機
喜愛刺激嗎?我們提供多款熱門老虎機遊戲,如「戰神賽特」「魔龍傳奇」等,每一款都擁有精美的畫面和豐厚的獎勵,讓每次旋轉都充滿期待!
3. 體育投注
無論你是球迷還是賽事愛好者,RG富遊的體育投注平台都有你想要的。精準的賠率、廣泛的賽事選擇,讓你體驗到投注的樂趣和刺激。
4. 彩票遊戲與捕魚遊戲
隨著運氣來!不妨試試我們的棋牌遊戲和彩票遊戲,這裡有各種機會讓你體驗到不一樣的勝利快感。
#### 下載富遊APP,隨時隨地享受遊戲!
富遊娛樂城的APP支持iOS和Android設備,界面簡潔易用,內容豐富。無論你身處何地,都能輕鬆進入遊戲世界,隨心所欲享受娛樂時光。此外,我們提供即時客服支援,隨時解決你的疑問,讓你的遊戲體驗更為完美。
#### 為什麼選擇RG富遊?
– 安全可靠
RG富遊致力於為玩家提供安全可靠的遊戲環境,您的每一筆交易都受到最高的安全保障。
– 快速便捷的存提款服務
我們的存款平均時間僅需30秒,提款平均只需60秒,讓你體驗即時的快感!
– 豐富多元的遊戲選擇
超過9999款遊戲任你選擇,無論你偏好哪種娛樂方式,這裡都能滿足你的需求。
– 全天候服務
我們全年365天提供不停歇的客服支持,確保你在遊戲過程中享受到最好的服務。
### 結論
RG富遊娛樂城是您最佳的線上賭場選擇。我們不僅提供多樣化的遊戲選擇,還有安全可靠的遊戲環境和便捷的服務。立即下載APP,加入RG富遊,開始您的娛樂之旅,享受無與倫比的遊戲體驗吧!
Dallasalure
December 7, 2024 @ 2:24 am
娛樂城
富遊娛樂城:台灣線上賭場的創新選擇
在台灣的線上娛樂市場中,富遊娛樂城憑藉其優質的服務和多樣化的娛樂選項,迅速成為新一代玩家的首選。既有刺激的遊戲體驗,又提供了一系列具有吸引力的優惠,富遊娛樂城無疑是追求樂趣的玩家們理想的地方。
#### 優勢與特色
1. 多樣化的遊戲選擇
富遊娛樂城提供了豐富的遊戲種類,包括電子老虎機、真人百家樂、運彩投注等。不論你是老虎機的愛好者,還是熱衷於真人賭桌的玩家,這裡都能滿足你的需求。特別推薦的遊戲如DG真人百家和RSG皇家百家樂,一定能帶給你極致的遊戲體驗。
2. 吸引人的優惠活動
富遊娛樂城針對新手和老玩家都推出了多樣的優惠活動。新會員首次存款可獲得相同金額的獎金,還有額外的體驗金送出,設計友好且實惠。此外,富遊每週的簽到獎勵和天天無上限的返水活動,更是讓玩家在遊戲中能持續獲得收益。
3. 安全可靠的存取款方式
在金融交易方面,富遊娛樂城支持多種存款和提款方式,包括各大銀行轉帳、超商儲值,甚至虛擬貨幣等。最低提款金額設置靈活,保證玩家資金的安全與便捷。
4. 人性化的操作介面
富遊娛樂城的網頁與移動應用設計簡潔易用,玩家可以方便地找到所需的遊戲和優惠。無論是在電腦還是手機上,都能順利享受到遊戲的樂趣。
#### 如何開始遊戲
如果你是新手玩家,開始你的富遊體驗非常簡單。只需註冊帳號,完成首次存款,即可獲得新手優惠。無論你想嘗試電子遊戲還是真人賭局,富遊娛樂城都能讓你輕鬆上手,享受娛樂的樂趣。
#### 社會責任與防沉迷措施
富遊娛樂城深知線上賭博的風險,因此致力於推廣負責任的遊戲文化。他們提供“防沉迷”指導,以確保玩家能夠理性投注,並提供必要的幫助和資源來幫助有需求的玩家。
#### 總結
對於尋求安全、便捷和多元化娛樂城體驗的玩家,富遊娛樂城絕對是值得選擇的選項。無論你是希望徹底放鬆,還是想要體驗刺激的博弈過程,富遊娛樂城都能滿足你的期待,成為你在台灣線上娛樂市場的最佳夥伴。立即註冊並開始你的遊戲之旅吧!
Robertvox
December 7, 2024 @ 7:38 am
ciprofloxacin 500mg buy online: buy cipro online – cipro for sale
DavidWes
December 7, 2024 @ 8:36 am
3a 娱乐城
3A娛樂城:全方位線上娛樂體驗的首選平台
3A娛樂城作為台灣最受歡迎的線上娛樂平台之一,以其多樣化的遊戲選擇、創新的技術、以及卓越的安全保障,贏得了玩家的高度信任與青睞。無論您是新手還是老手,3A娛樂城都能滿足您的需求,帶給您獨一無二的遊戲體驗。
熱門娛樂城遊戲一覽
3A娛樂城提供多種熱門遊戲,適合不同類型的玩家:
真人百家樂:體驗真實賭場的氛圍,與真人荷官互動,感受賭桌上的緊張與刺激。
彩票投注:涵蓋多種彩票選項,滿足彩券愛好者的需求,讓您隨時隨地挑戰幸運。
棋牌遊戲:從傳統的麻將到現代化的紙牌遊戲,讓玩家在策略與運氣中找到平衡。
3A娛樂城的核心價值
1. 專業
3A娛樂城每日提供近千場體育賽事,並搭配真人百家樂、彩票彩球、電子遊戲等多種類型的線上賭場遊戲。無論您的遊戲偏好為何,這裡總有一款適合您!
2. 安全
平台採用128位加密技術和嚴格的安全管理體系,確保玩家的金流與個人資料完全受保護,讓您可以放心遊玩,無需擔憂安全問題。
3. 便捷
3A娛樂城是全台第一家使用自家開發的全套終端應用的娛樂城平台。無論是手機還是電腦,玩家都能享受無縫的遊戲體驗。同時,24小時線上客服隨時為您解決任何問題,提供貼心服務。
4. 安心
由專業工程師開發的財務處理系統,為玩家帶來快速的存款、取款和轉帳服務。透過獨立網路技術,平台提供優化的網路速度,確保每一次操作都流暢無比。
下載3A娛樂城手機APP
為了提供更便捷的遊戲體驗,3A娛樂城獨家開發了功能齊全的手機APP,讓玩家隨時隨地開啟遊戲。
現代化設計:新穎乾淨的介面設計,提升使用者體驗。
跨平台支持:完美適配手機與電腦,讓您輕鬆切換設備,無需中斷遊戲。
快速下載:掃描專屬QR碼即可進入下載頁面,立即開始暢玩!
3A娛樂城的其他服務
娛樂城教學:詳細的操作指導,讓新手也能快速上手。
責任博彩:提倡健康娛樂,確保玩家在遊戲中享受樂趣的同時,不失理性。
隱私權政策:嚴格遵守隱私保護條例,保障玩家的個人信息安全。
立即加入3A娛樂城,享受頂級娛樂體驗!
如果您正在尋找一個專業、安全、便捷又安心的線上娛樂平台,那麼3A娛樂城絕對是您的不二之選。現在就下載手機APP,隨時隨地開啟您的娛樂旅程,享受無限的遊戲樂趣與刺激!
Jimmieclict
December 7, 2024 @ 10:13 am
http://lisinoprilus.com/# cost of brand name lisinopril
ciondolo orgonite
December 7, 2024 @ 2:28 pm
že spousta z něj se objevuje na internetu bez mého souhlasu.
Jaimeget
December 7, 2024 @ 3:51 pm
ciprofloxacin 500mg buy online [url=http://ciprofloxacin.cheap/#]buy ciprofloxacin over the counter[/url] ciprofloxacin order online
Ремонт телевизоров Samsung Москва
December 7, 2024 @ 7:13 pm
Предлагаем услуги профессиональных инженеров офицальной мастерской.
Еслли вы искали ремонт телевизоров samsung цены, можете посмотреть на сайте: ремонт телевизоров samsung адреса
Наши мастера оперативно устранят неисправности вашего устройства в сервисе или с выездом на дом!
Jimmieclict
December 7, 2024 @ 7:14 pm
https://clomid.store/# can i order cheap clomid pill
ArmandoWal
December 7, 2024 @ 7:31 pm
can you buy clomid without insurance: order cheap clomid without prescription – clomid without prescription
Hubertloyap
December 7, 2024 @ 8:27 pm
Reliable HVAC Repair Services https://serviceorangecounty.com stay comfortable year-round with our professional HVAC repair services. Our experienced team is dedicated to diagnosing and resolving heating, cooling, and ventilation issues quickly and effectively.
Jaimeget
December 8, 2024 @ 12:44 am
cost of generic clomid no prescription [url=http://clomid.store/#]order cheap clomid now[/url] get cheap clomid without dr prescription
Matthewpok
December 8, 2024 @ 12:58 am
http://ciprofloxacin.cheap/# buy generic ciprofloxacin
zithromax 500mg price [url=https://azithromycinus.com/#]zithromax generic price[/url] zithromax 500 mg for sale
orgone
December 8, 2024 @ 8:39 am
) سأعيد زيارتها مرة أخرى لأنني قمت بوضع علامة كتاب عليها. المال والحرية هي أفضل طريقة للتغيير، أتمنى أن تكون غنيًا و
Jaimeget
December 8, 2024 @ 9:32 am
price of lisinopril [url=https://lisinoprilus.com/#]price of lisinopril[/url] buy lisinopril 10 mg tablet
Jimmieclict
December 8, 2024 @ 1:10 pm
https://azithromycinus.com/# zithromax online australia
DustinHen
December 8, 2024 @ 2:04 pm
2024 年熱門娛樂城與活動解析
冠天下娛樂城優惠,帶來無限樂趣
在 2024 年 4 月 16 日,冠天下娛樂城推出了全新的優惠活動,旨在為玩家帶來更大的獲利機會和更高的娛樂體驗。這些優惠活動包括多種存款獎勵、會員專屬回饋以及豐富的遊戲競賽獎金,吸引了大批玩家參與。
冠天下娛樂城以其高透明度和用戶友好的政策,已成為許多玩家的首選娛樂平台。不論是新手還是老手,都能在這裡找到適合自己的遊戲和活動。
LEO娛樂城無法登入引發市場恐慌
2024 年 11 月 29 日,LEO娛樂城因技術問題出現短暫無法登入的情況,引發了廣泛關注。許多玩家擔心這是否意味著九州娛樂城的營運出現問題。
九州娛樂集團隨後發佈官方聲明,強調這僅僅是系統升級過程中的短暫問題,並承諾玩家的資金和數據安全不受任何影響。同時,九州娛樂表示將加強伺服器穩定性,確保未來類似情況不再發生。
熱門 ZG 電子遊戲——熱舞森巴老虎機
2024 年 11 月 8 日,ZG 電子遊戲推出了熱舞森巴老虎機,這款遊戲迅速成為市場上的熱門選擇。以南美熱情的森巴舞為主題,遊戲畫面充滿色彩,搭配動感的音樂,為玩家帶來視覺與聽覺的雙重享受。
遊戲亮點:
高倍數獎勵機制:每次轉盤都有機會觸發高額獎金。
特色免費遊戲:連續獲得特殊圖案即可啟動多次免費轉盤,提高獲勝機率。
適合新手與高端玩家:簡單的玩法規則與豐富的策略選擇滿足不同層級玩家的需求。
ZG 電子遊戲致力於為玩家提供創新且有趣的遊戲體驗,熱舞森巴老虎機無疑是今年最值得嘗試的遊戲之一。
2024 世界棒球 12 強賽精彩回顧
2024 年 11 月,中華隊參加了世界棒球 12 強賽,這項全球頂尖棒球賽事吸引了來自多國的球隊和球迷參與。中華隊 28 人的名單以年輕選手和經驗豐富的老將為基礎,展現了團隊的競技實力。
比賽亮點:
精彩賽程:比賽期間,中華隊展現了堅強的防守和出色的打擊表現。
門票銷售:賽事門票迅速售罄,顯示出棒球在台灣的高度受歡迎程度。
線上直播:各大平台提供了高畫質直播,讓更多球迷能夠即時觀看比賽,感受棒球魅力。
世界棒球 12 強賽不僅是比賽,更是全台灣球迷的一場盛宴,展現了棒球作為國民運動的無限魅力。
結語
從娛樂城的優惠活動到全球性的棒球賽事,2024 年對於遊戲和體育愛好者來說充滿了亮點。不論是參與冠天下娛樂城的優惠,體驗 ZG 電子遊戲的最新老虎機,還是為中華隊的棒球比賽加油,這一年都是充滿精彩與期待的一年!
segno dello scorpione
December 8, 2024 @ 3:30 pm
Hey! Would you mind if I share your blog with my facebook group? There’s a lot of folks that I think would really appreciate your content. Please let me know.Thanks
onexxxslots
December 8, 2024 @ 3:58 pm
Для игры скачайте 1xslots официальный сайт.
Jaimeget
December 8, 2024 @ 6:16 pm
Abortion pills online [url=https://cytotec.top/#]buy cytotec over the counter[/url] order cytotec online
Rafaelfremy
December 8, 2024 @ 10:31 pm
Leo娛樂城無法登入
九州娛樂城與Leo娛樂城即將結束運營,玩家該如何應對?
近期娛樂城行業的消息引發了玩家的廣泛關注,其中「Leo娛樂城無法登入」和「九州娛樂城即將倒閉」成為熱門話題。根據官方公告,九州娛樂城及其旗下品牌,包括Leo娛樂城和THA娛樂城,將於2024年12月31日中午12點正式結束營業並關閉官網。這一消息不僅讓玩家感到震驚,也暴露出行業潛在的問題。
Leo娛樂城無法登入的原因
Leo娛樂城近期頻繁出現登入問題,這與其即將退出台灣市場的計劃息息相關。以下是造成登入困難的主要原因:
帳號或密碼問題
許多玩家因忘記密碼或帳號被盜導致登入失敗。然而,由於官方客服系統已停止服務,這些問題無法得到解決。
網頁技術問題
官網伺服器的不穩定或維護期間,可能出現「404錯誤」,讓玩家無法正常登入。
營運關閉計劃
隨著九州娛樂城宣布結束營業,Leo娛樂城的伺服器和備用網址也逐步停止運行,這是玩家無法登入的最大原因。
為何九州娛樂城退出台灣市場?
九州娛樂城決定退出台灣市場的原因可以歸納為以下幾點:
市場收益不足
台灣市場對九州娛樂城而言並不具備高盈利性,難以支撐長期運營。
第三方支付系統受阻
多次傳出第三方現金支付渠道中斷,影響存提款操作。
法律風險增加
九州娛樂城頻繁遭遇法律問題及負面新聞,導致營運壓力倍增。
玩家需要注意什麼?
隨著九州娛樂城及其旗下品牌逐步退出市場,玩家應及時採取以下措施:
提領餘額
官方已停止存款服務,但提領功能仍開放。玩家應儘快提領餘額,避免損失。
轉移到穩定平台
針對Leo娛樂城和九州娛樂城的退出,玩家可選擇更穩定且具信譽的平台,以保障自己的娛樂體驗。
防範詐騙
小心冒名客服或假網站,官方已停止客服系統,不會主動聯繫玩家索取個人資料。
未來哪些娛樂城值得信賴?
隨著九州娛樂城退出市場,玩家可以關注一些以穩定性和合法性著稱的娛樂平台。選擇時應注意以下幾點:
合法牌照
選擇擁有合法經營許可的娛樂平台。
穩定的支付系統
平台應提供多種穩定且快速的支付方式。
用戶口碑
瀏覽玩家的評價和反饋,選擇高評價的平台。
結論
九州娛樂城及其旗下品牌的關閉標誌著一個時代的結束,同時也提醒玩家關注行業的合規性和穩定性。在此轉型時期,玩家應保持警惕,選擇可靠的平台,以確保自己的資金和遊戲體驗不受影響。
Bradleyvat
December 8, 2024 @ 10:46 pm
http://semaglutidetablets.store/# rybelsus semaglutide tablets
Jamescaurf
December 9, 2024 @ 3:19 am
erectile dysfunction medicines: erectile dysfunction pills – help with ed
StevenDrums
December 9, 2024 @ 4:45 am
https://edpills.men/# erectile dysfunction meds online
CharlesExore
December 9, 2024 @ 6:27 am
cenforce.icu: cenforce for sale – cenforce for sale
Keo nha cai
December 9, 2024 @ 6:40 am
I really enjoyed this post. It was well-written and informative.Keo nha cai
Keo nha cai
December 9, 2024 @ 6:42 am
I really enjoyed this post. It was well-written and informative.Keo nha cai
segno dello scorpione
December 9, 2024 @ 10:33 am
Díky moc!|Hej, jeg synes, dette er en fremragende blog. Jeg snublede over det;
Bradleyvat
December 9, 2024 @ 11:06 am
http://cenforce.icu/# buy cenforce
Bradleyvat
December 9, 2024 @ 5:15 pm
https://semaglutidetablets.store/# semaglutide tablets price
CoreyamBit
December 9, 2024 @ 6:36 pm
cheapest erectile dysfunction pills [url=https://edpills.men/#]online erectile dysfunction prescription[/url] ed pills for sale
카지노솔루션
December 10, 2024 @ 1:51 am
This is a very interesting blog. A lot of the blogs I read these days don’t offer anything I’m interested in, but this one definitely does. I just thought I’d post it and let you know.
onexxxsila
December 10, 2024 @ 4:33 am
Характеристики 1xSlots https://aztarna.es/pages/1xslots-casino_15.html
MetaMask Download
December 10, 2024 @ 5:58 am
Thanks for the inspiration, this was amazing!
Bradleyvat
December 10, 2024 @ 6:07 am
https://semaglutidetablets.store/# buy semaglutide
Jamescaurf
December 10, 2024 @ 6:55 am
semaglutide best price: generic rybelsus tabs – cheap semaglutide pills
StevenDrums
December 10, 2024 @ 7:28 am
http://semaglutidetablets.store/# semaglutide best price
Social Media
December 10, 2024 @ 9:33 am
Hello amazing people! Keep in mind that you’ve got the capability to bring about uplifting change in your life. Trust in yourself and take action.
pendente
December 10, 2024 @ 9:45 am
nenarazili jste někdy na problémy s plagorismem nebo porušováním autorských práv? Moje webové stránky mají spoustu unikátního obsahu, který jsem vytvořil.
Bradleyvat
December 10, 2024 @ 12:25 pm
http://drugs1st.pro/# drugs1st
Jamescaurf
December 10, 2024 @ 3:59 pm
erection pills: drugs1st – drugs1st
StevenDrums
December 10, 2024 @ 4:07 pm
http://cenforce.icu/# Purchase Cenforce Online
CoreyamBit
December 10, 2024 @ 6:19 pm
semaglutide best price [url=https://semaglutidetablets.store/#]rybelsus semaglutide tablets[/url] cheap semaglutide pills
Bradleyvat
December 10, 2024 @ 6:28 pm
https://drugs1st.pro/# drugs1st
orgone
December 10, 2024 @ 7:29 pm
také jsem si vás poznamenal, abych se podíval na nové věci na vašem blogu.|Hej! Vadilo by vám, kdybych sdílel váš blog s mým facebookem.
onexrussiasite
December 10, 2024 @ 8:11 pm
Играйте безопасно с 1xslots официальным зеркалом и не беспокойтесь о блокировках.
CharlesExore
December 10, 2024 @ 9:56 pm
Purchase Cenforce Online: Cenforce 100mg tablets for sale – Purchase Cenforce Online
KennethSipsy
December 10, 2024 @ 10:28 pm
Discover the world of Minecraft
Experience the Universe of Minecraft: Your Ultimate Endurance and Freedom Adventure
Welcome to your Portal to the Most Engaging and Absorbing Minecraft Connected Encounter. Whether you’re a Designer, Warrior, Discoverer, or Strategist, our Server Provides Infinite Opportunities to Enjoy Persistence and Chaos Features in Methods you’ve Seldom seen Earlier.
—
Why Select Adventures in Minecraft?
Our Network is Built to Offer the Best Minecraft Encounter, Integrating Tailored Zones, Thrilling Mechanics, and a Supportive Group. Traverse, Overcome, and Construct your own Adventures with Unique Elements Customized for All type of Gamer.
—
Essential Features
– Endurance and Freedom Options: Confront the Thrill of Persisting against the odds or Dive into Untamed PvP Engagements with no rules and full freedom.
– Massive Server Scale: With Space for up to 3,750 Participants, the Fun never stops.
– 24/7 Server Access: Join At All Times to Explore Uninterrupted, Consistent Play.
– Specialized Resources: Navigate our Precisely Created Minecraft Worlds Stocked with Mods, Features, and Unique Products from our Online Inventory.
—
Unique Mechanics Options
Living Feature
In Endurance Feature, you’ll Traverse Wide Landscapes, Collect Materials, and Create to your heart’s content. Battle off Creatures, Team Up with Friends, or Accept on Single-Player Challenges where only the Resilient Overcome.
Disorder Feature
For Players Looking For Excitement and Adrenaline, Freedom Scenario Delivers a Universe with Total Freedom. Immerse in Competitive PvP Clashes, Build Alliances, or Dominate Others to Dominate the Zone. Here, Survival of the Fittest is the True Law.
—
Special Minecraft Components
– Exploration Worlds: Discover Exciting Minecraft Caves and RPG-Style Quests.
– Trading and Bartering: Our User-Controlled Market Permits you to Exchange, Purchase, and Exchange Items to Climb the Levels and Create Your Status as a Influential Competitor.
– Minecraft Inventory: Enter Unique Items, Levels, and Ranks that Boost your Experience.
—
Minecraft Inventory: Enhance Your Interaction
Our Online Shop Delivers a Variety of Enhancements, Statuses, and Products to Cater To every Approach. From Inexpensive Contribution Offers to Top-Level Statuses, you can Achieve Fresh Options and Advance your Gameplay to the Next Level.
—
Best-Selling Goods
– Donate Bundles (x10) – €1.00
– VIP – €1.40
– Elysium Tier – €20.00
– OWNER Level – €40.00
– BOSS Level – €60.00
—
Top Statuses for Premier Users
– CREATOR (€10.00) – Gain Creative Components to Express your Vision.
– Vanguard (€12.00) – Premier Benefits and Tailored Advantages.
– Paragon (€59.10) – Exclusive Privileges for the Ultimate Competitor.
– Luminescent (€50.00) – Excel as a Elite Champion on the Network.
—
Join Our Growing Minecraft Community
We Strive in Forming a Supportive, Engaging, and Friendly Network. Whether you’re Completing RPG Tasks, Discovering Tailored Maps, or Taking Part in User-Led PvP, there’s Always something New to Explore.
—
Features You Can Look Forward To
– Friendly Player Base: Meet Passionate Minecraft Enthusiasts from Everywhere.
– Exciting Challenges: Join in Unique Tasks, Events, and Community-Wide Challenges.
– Dedicated Team: Our Moderators Delivers Seamless Gameplay and Helps you with any Concerns.
Williamcruth
December 10, 2024 @ 10:37 pm
Our GTA 5 RP server offers a vast interactive gameplay improved with unique custom add-ons and playstyle features.
Whether you’re a honest officer or a aspiring illegal strategist, the chances in Los Santos are boundless:
Create Your Power: Form gangs, lead missions, or ascend the criminal hierarchy to control the blocks.
Act As a Guardian: Maintain law as a law enforcer, emergency worker, or medic.
Tailor Your Adventure: Design your existence, from high-end vehicles and homes to your player’s unique look and tales.
Adventurous Challenges: Participate in intense missions, massive challenges, and player-driven engagements to collect benefits and status.
Dominate the Wealth and Earn Big!
At vs-rp.com, we give you the resources and chances to collect significant in the in-game universe of GTA RP. From acquiring high-end properties to building wealth, the RP system is yours to master.
Build your fortune through:
Player-Driven Tasks: Accomplish tasks, deals, and contracts to receive digital money.
Custom Businesses: Launch and grow your own operation, from vehicle markets to venues.
High-End Resources: Acquire luxury cars, premium properties, and unique virtual products.
Are you eager to rule Los Santos and transform into a virtual mogul?
Reasons to Select This Experience?
Here’s why we’re the top GTA 5 RP network:
1. Consistent Enhancements and Events
Stay in front of the rivals with special improvements, tasks, and occasional challenges. New content are included regularly to keep your gameplay engaging and thrilling.
2. Wide Range of Assets
Browse a enormous catalog of digital goods, including:
High-End Vehicles
Luxury Homes
Limited Upgrades
3. Committed Support Team
Whether you’re a veteran gamer or a new beginner, our helpers is ready to guide. We’ll assist you in configuring, provide resources, and guarantee you succeed in Los Santos.
JustinBoIli
December 11, 2024 @ 12:21 am
http://pinup2025.com/# пин ап
Josephsheah
December 11, 2024 @ 12:58 am
пинап казино: pinup 2025 – пин ап казино
Lucky Spin
December 11, 2024 @ 2:58 am
As a newcomer to Lucky Spin, I’m loving the rich discussions. The number of comments pouring in on your articles shows that others are just as captivated by these topics!Lucky Spin
Пенсионный возраст для женщин – 2024 год.
December 11, 2024 @ 3:00 am
Пенсионный возраст для женщин – 2024 год.
новости спецоперации
December 11, 2024 @ 3:39 am
новости спецоперации
геомагнитная буря
December 11, 2024 @ 4:20 am
геомагнитная буря
сбербанк
December 11, 2024 @ 5:46 am
сбербанк
Robertmoica
December 11, 2024 @ 5:51 am
пинап казино [url=http://pinup2025.com/#]pinup 2025[/url] пин ап
JustinBoIli
December 11, 2024 @ 6:31 am
https://pinup2025.com/# пин ап
флаг азербайджана
December 11, 2024 @ 6:33 am
флаг азербайджана
самая высокая гора в мире
December 11, 2024 @ 7:16 am
самая высокая гора в мире
TravisTem
December 11, 2024 @ 7:23 am
пин ап казино официальный сайт: пин ап вход – пин ап
Josephsheah
December 11, 2024 @ 9:12 am
bonus veren bahis siteleri yasal: gerГ§ek paralД± casino oyunlarД± – bet siteleri bonus
EdwardSit
December 11, 2024 @ 10:54 am
http://pinup2025.com/# pinup 2025
segno zodiacale del leone
December 11, 2024 @ 11:36 am
Tak Hej der til alle, det indhold, der findes på denne
JustinBoIli
December 11, 2024 @ 12:32 pm
http://casinositeleri2025.pro/# betnoo
Customer Journey
December 11, 2024 @ 1:57 pm
Hello dear friends! No matter what you’re going through, remember to maintain hope. Sunnier days are on the horizon.
onexxxxslots
December 11, 2024 @ 4:55 pm
Обзор 1xSlots https://santaeugenia.archimadrid.es/pags/1xslots-casino-argentina_1.html
Josephsheah
December 11, 2024 @ 5:01 pm
pinup2025.com: пин ап – пин ап