We often think of damage on a surface level.
But for plants, much of the important response to an insect bite takes place out of sight. Over minutes and hours, particular plant genes are turned on and off to fight back, translating into changes in its defenses.
In one of the broadest studies of its kind, scientists at the University of Missouri Bond Life Sciences Center recently looked at all plant genes and their response to the enemy.
“There are 28,000 genes in the plant, and we detected 2,778 genes responding, depending on the type of insect,” said Jack Schultz, Bond LSC director and study co-author. “Imagine you only look at a few of these genes, you get a very limited picture and possibly one that doesn’t represent what’s going on at all. This is by far the most comprehensive study of its type, allowing scientists to draw conclusions and get it right.”
Their results showed that the model Arabidopsis plant recognizes and responds differently to four insect species. The insects cause changes on a transcriptional level, triggering proteins that switch on and off plant genes to help defend against more attacks.
The difference in the insect
“It was no surprise that the plant responded differently to having its leaves chewed by a caterpillar or pierced by an aphid’s needle-like mouthparts,” said Heidi Appel, Bond LSC Investigator and lead author of the study. “But we were amazed that the plant responded so differently to insects that feed in the same way.”
Plants fed on by caterpillars – cabbage butterfly and beet armyworms – shared less than a quarter of their changes in gene expression. Likewise, plants fed on by the two species of aphids shared less than 10 percent of their changes in gene expression.
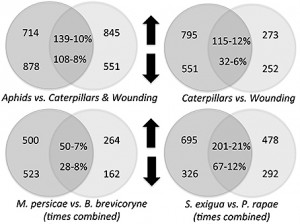
The plant responses to caterpillars were also very different than the plant response to mechanical wounding, sharing only about 10 percent of their gene expression changes. The overlap in plant gene responses between caterpillar and aphid treatments was also only 10 percent.
“The important thing is plants can tell the insects apart and respond in significantly different ways,” Schultz said. “And that’s more than most people give plants credit for.”
A sister study explored this phenomena further, led by former MU doctoral student Erin Rehrig.
It showed feeding of both caterpillars increased jasmonate and ethylene – well-known plant hormones that mediate defense responses. However, plants responded quicker and more strongly when fed on by the beet armyworm than by the cabbage butterfly caterpillar in most cases, indicating again that the plant can tell the two caterpillars apart.
The result is that the plant turns defense genes on earlier for beet armyworm.
In ecological terms, a quick defense response means the caterpillar won’t hang around very long and will move on to a different meal source.
More questions
A study this large has potential to open up a world of questions begging for answers.
“Among the genes changed when insects bite are ones that regulate processes like root growth, water use and other ecologically significant process that plants carefully monitor and control,” Schultz said. “Questions about the cost to the plant if the insect continues to eat would be an interesting follow-up study for doctoral students to explore these deeper genetic interactions.”
Frontiers in Plant Science published the primary study in its November 2014 issue. The sister study can be read here.